Logan Amos, Andrea Belen, Miki Fok, Yihan (Mariana) Ma, Annie Nguyen, Yuejiao (Vivian) Zhou ▫ Western Reserve Academy, Hudson OH
Reviewed on 6 May 2023; Accepted on 13 July 2023; Published on 16 October 2023
With help from the 2023 BioTreks Production Team.
Ultraviolet radiation plays a critical role in the development of skin cancer. The sun emits electromagnetic waves in the form of ultraviolet radiation which penetrates human skin layers and contributes to skin burning, wrinkling, and aging. Overexposure to UV radiation can also produce reactive oxygen species (ROS), byproducts of oxidative metabolism, which cause untreatable cell damage and increase the risk of skin cancer. Sunscreen blocks UV rays from the skin. Even low-protection sunscreen reduces non-melanoma skin cancer by about 40% and lowers the risk of melanoma skin cancer by 50%. However, many active ingredients in sunscreen are harsh chemicals that further skin damage. In 2021, fourteen of the sixteen FDA-allowed sunscreen chemicals were identified and scrutinized for their lack of effectiveness and safety concerns. For example, two chemicals, oxybenzone and octinoxate, are associated with coral reef bleaching and ultimately kill the coral. Dramatic reduction in coral reef populations can disrupt the ecological balance and seriously impact the ecosystem. Our study presents an innovative method to extract two sunscreen pigments from marine bacteria, cyanobacteria, offering the opportunity to replace the traditional active ingredients in chemical sunscreens with nonhazardous pigments. This photosynthetic microbe produces two environmentally friendly pigments, scytonemin and mycosporine-like amino acids (MAAs), shielding the cell from 90% of UV-A light and absorbing UV-B radiation, respectively. We propose to engineer Escherichia coli to overexpress genes required for biosynthesis of two pigments, scytonemin (scyA-F) and mycosporine-glycine (MysC). Combining these pigments with inactive but beneficial ingredients commonly found in other sunscreens produces an environmentally friendly sunscreen that will benefit both the ecosystem and humans.
Keywords: Cyanobacteria, sunscreen, scytonemin, mycosporine-like amino acids, ultraviolet radiation
Authors are listed in alphabetical order. Beth Pethel from Western Reserve Academy, Hudson OH mentored the group. Please direct all correspondence to pethelb@wra.net.
Ultraviolet radiation (UV) is a form of electromagnetic radiation emitted from the sun that can penetrate the layers of human skin (Hölzle & Hönigsmann, 2005). Skin is composed of two main layers. Epidermis is the visible outermost layer of the skin (“Epidermis,” 2017). Underneath is the dermis, which is involved in thermoregulation and sensation (Brown & Krishnamurthy, 2022). UV radiation can reach different skin layers depending on its wavelength (Figure 1). UV radiation is divided into three types based on the variations in wavelength. UV-A has the longest wavelength amongst the three types of UV radiation. With a range of 310-400 nm, most UV-A radiation can reach the Earth’s surface and hit the dermis, contributing to skin burning and wrinkling (Radiation: Ultraviolet, 2016). Medium wavelength UV-B ranges between 280-315 nm and is responsible for tanning and thickening of the epidermis.
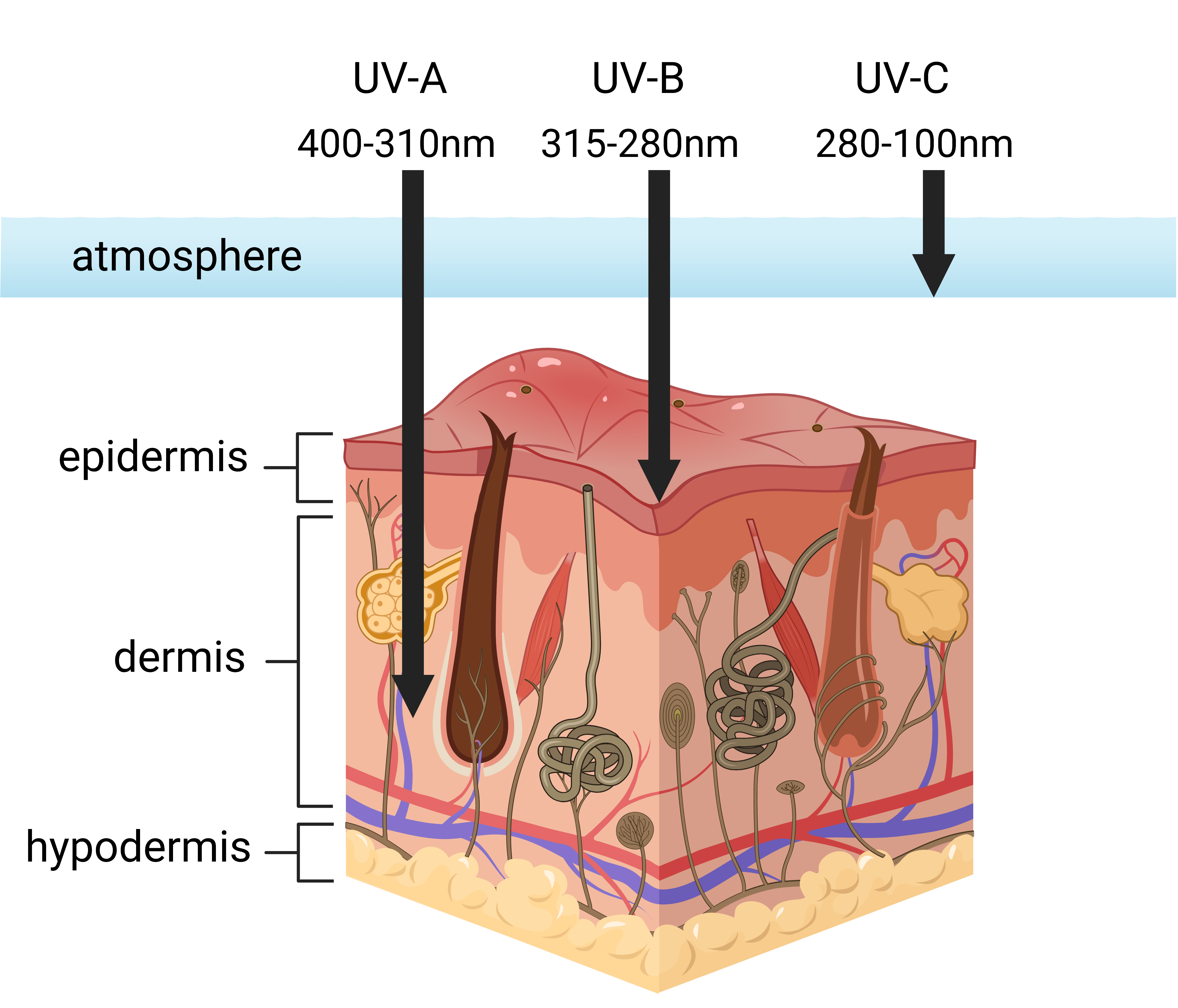
| Figure 1. Different types of UV rays penetrating the skin |
|---|
Though UV-B radiation does not penetrate the skin layers as deep as UV-A, it can still promote skin aging and increases the chance of developing skin cancer. UV-C, with a wavelength of 100-280 nm, is the shortest wavelength among the three and the most harmful type of UV radiation that can cause severe burns and eye injuries (Radiation: Ultraviolet, 2016). Fortunately, UV-C is completely filtered by the ozone layer in the Earth’s atmosphere (Ultraviolet (UV), 2020). Since UV-C and 90-95% of UV-B radiation are absorbed by the stratospheric ozone layer and do not reach the ground, the UV radiations humans come in contact with are mainly UV-A and a small amount of UV-B (Singh et al., 2021). All three types of UV radiation cause unstable oxygen molecules to form in the skin which damage the elastin fibers that are responsible for skin elasticity and resilience (Better Health Channel, 2021).
Ozone depletion, which occurs due to the release of gaseous chlorine and bromine from industry, increases the levels of UV radiation that reaches Earth (Barnes et al., 2019). Excessive exposure to UV radiation can lead to the production of reactive oxygen species (ROS), byproducts of oxidative metabolism. This excess accumulation of ROS results in oxidative stress, which is a state of imbalance in the production rate of ROS and the detoxification rate of these products resulting in an overabundance of free radicals (Ray et al., 2012). ROS are highly reactive chemicals that damage basic building blocks of the cell including DNA, proteins, and lipids which can expedite the effects of aging and increase the risk of cancer (Shields et al., 2021). The damage, if unrepaired, may eventually result in cancer and spontaneous mutation (Rosic, 2019).
Due to UV radiation, skin cancer is the most common cancer in the United States. One out of five Americans will suffer from skin cancer in their lifetime (Skin Cancer,” n.d.). There are two types of skin cancer including melanoma and non-melanoma. Skin cancer typically begins in the epidermis. This layer of skin contains three main types of cells: melanocytes, squamous cells, and basal cells. Melanocytes are the cells that produce melanin, a pigment which gives color to the skin. Squamous cells are the flat cells on the top layer of the epidermis. Basal cells are the cells in the lower part of the epidermis (Figure 2) (American Cancer Society, n.d.). Melanoma develops when melanocytes grow uncontrollably, potentially spreading to the rest of the body if not treated early (Melanoma, 2022). Basal cell carcinoma (BCC) and squamous cell carcinoma (SCC) are the two most common types of non-melanoma skin cancer. BCC forms in the basal cells while SCC develops in squamous cells (“Nonmelanoma Skin,” n.d.). Though BCC and SCC are less likely to spread and easier to treat than melanoma, they are generally more common. Approximately two to three million people suffer from non-melanoma skin cancer each year. In contrast, only 132,000 people are affected by melanoma skin cancer annually (Radiation: Ultraviolet, 2017). From 1976 to 1984, BCC cases increased by 145% and SCC cases increased by 236%, highlighting an increase in skin cancer diagnoses (“Skin Cancer,” n.d.).
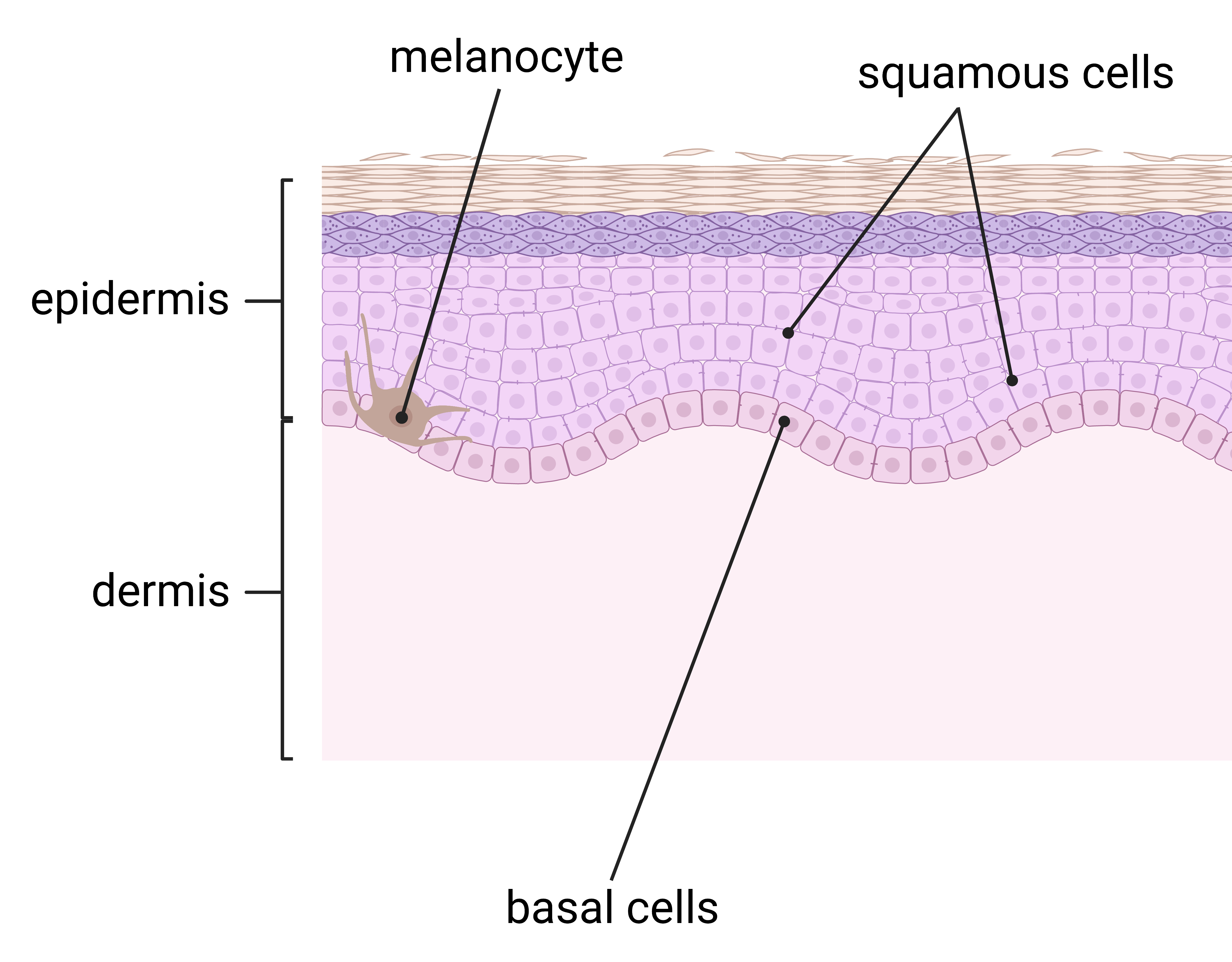
| Figure 2: Three major cells that make up the epidermis |
|---|
Sunscreen can help prevent sunburn, skin cancer, and premature aging (Sunscreen and Your, n.d.). There are two types of sunscreen: physical blockers (e.g., mineral sunscreen) and chemical absorbers. Physical blockers such as zinc oxide and titanium dioxide are small mineral particles that reflect UV rays away from your skin. Chemical absorbers create a thin film over the skin that absorbs UV radiation (Bramlet, 2016). Mineral sunscreen remains on the skin’s surface, acting as a shield, while chemical sunscreen sinks into skin. Each sunscreen has a sun protection factor (SPF) level which measures the amount of solar energy needed to produce a sunburn on protected skin in relation to how much solar energy is needed to produce a sunburn on unprotected skin (“Sun Protection,” n.d.). SPF can be grouped into four categories: (1) low protection (SPF<15), (2) medium protection (SPF 15-29), (3) high protection (30-49), and (4) very high protection (SPF>50). SPF 15 blocks 93% of UV-B rays. Regular use of SPF 15 can reduce non-melanoma skin cancer by about 40% and reduce the risk of melanoma skin cancer by 50% (Autier, 2000).
According to current research, another cause of skin damage is from the harsh chemicals present in modern-day sunscreen (Ruszkiewicz et al., 2017). In 2021, the FDA reviewed previously approved sunscreen ingredients after fourteen of the sixteen allowable chemicals were scrutinized for their lack of effectiveness and safety concerns. Of the ingredients listed, aminobenzoic acid (PABA) and trolamine salicylate were banned in the EU due to their harmful effect(s) on skin (FDA Proposes, n.d.). The only known harmless ingredients are zinc oxide and titanium dioxide (FDA, 2019). While these harmless ingredients were not banned, the allowed concentrations and combinations were altered to ensure sunscreen effectiveness. The FDA recommendations on the safety of those ingredients are ambiguous, requiring more research on the agency’s behalf (FDA, 2019). There is also an environmental concern regarding the use of these chemicals. Although there are sixteen ingredients allowed by the FDA, only eight are regularly used by American sunscreen manufacturers: avobenzone, homosalate, octinoxate, octisalate, octocrylene, oxybenzone, titanium dioxide, and zinc oxide (Boerner, 2021). However, oxybenzone and octinoxate were banned in several regions including Hawaii, Florida’s Key West, and the western Pacific nation of Palau due to their harmful effects on coral reefs and marine life (“Coalition Petitions,” 2021). When oxybenzone and octinoxate enter the marine ecosystem, the coral’s ability to defend itself from bleaching is inhibited (Coral Bleaching, 2015; Is Your, 2018). Coral reef bleaching results in the death of coral tissue algae, which act as a pH balancer, food source, and solar radiation protector. This bleaching ultimately damages its DNA and development (Coral Bleaching, 2015; Is Your, 2018). Studies also show that octocrylene may cause DNA damage in a similar manner (Pandika, 2019). In addition to limiting corals’ defense against bleaching, the chemicals have also been found to kill coral larvae and create phototoxins when absorbed by mature coral (Bennett, 2022).
In light of these issues, this study will rely on the use of marine bacteria, cyanobacteria, to create our final sunscreen product. Also referred to as blue-green algae, cyanobacteria are photosynthetic microbes which flourish near the surface of inland waters (Vincent, 2009). Due to their location on the surface of lakes and ponds, cyanobacteria are exposed to harsh sunlight during their lifetime. Many strains of cyanobacteria produce two pigments: scytonemin and mycosporine-like amino acids (MAAs), which protect the bacteria from UV rays. Unique to cyanobacteria, the butterfly-shaped yellowish-brown scytonemin pigment shields the cell from 90% of UV-A light (Figure 3) (Wada et al., 2013; Lewis, 2020; Assunção et al., 2022). Once isolated and placed in a solvent, scytonemin absorbs radiation at a maximum wavelength of 384 nm (Wada et al., 2013). Mycosporine-like amino acids are water-soluble pigments produced by cyanobacteria and algae (Figure 4) (Wada et al., 2015). MAAs, such as shinorine and mycosporine-glycine, are shown to possess increased absorption of UV-B radiation while having maximum absorption of 334 nm and 331 nm, respectively (Shick et al., 1992; Whitehead & Vernet, 2000). They also harmlessly release this energy as heat. Abiotic stressors such as pH and temperature also have a limited effect on MAAs which provide versatility and durability (Kogej et al., 2006). Scytonemin and mycosporine-like amino acids protect biological molecules from oxidative damage caused by ROS (Wada et al., 2013).
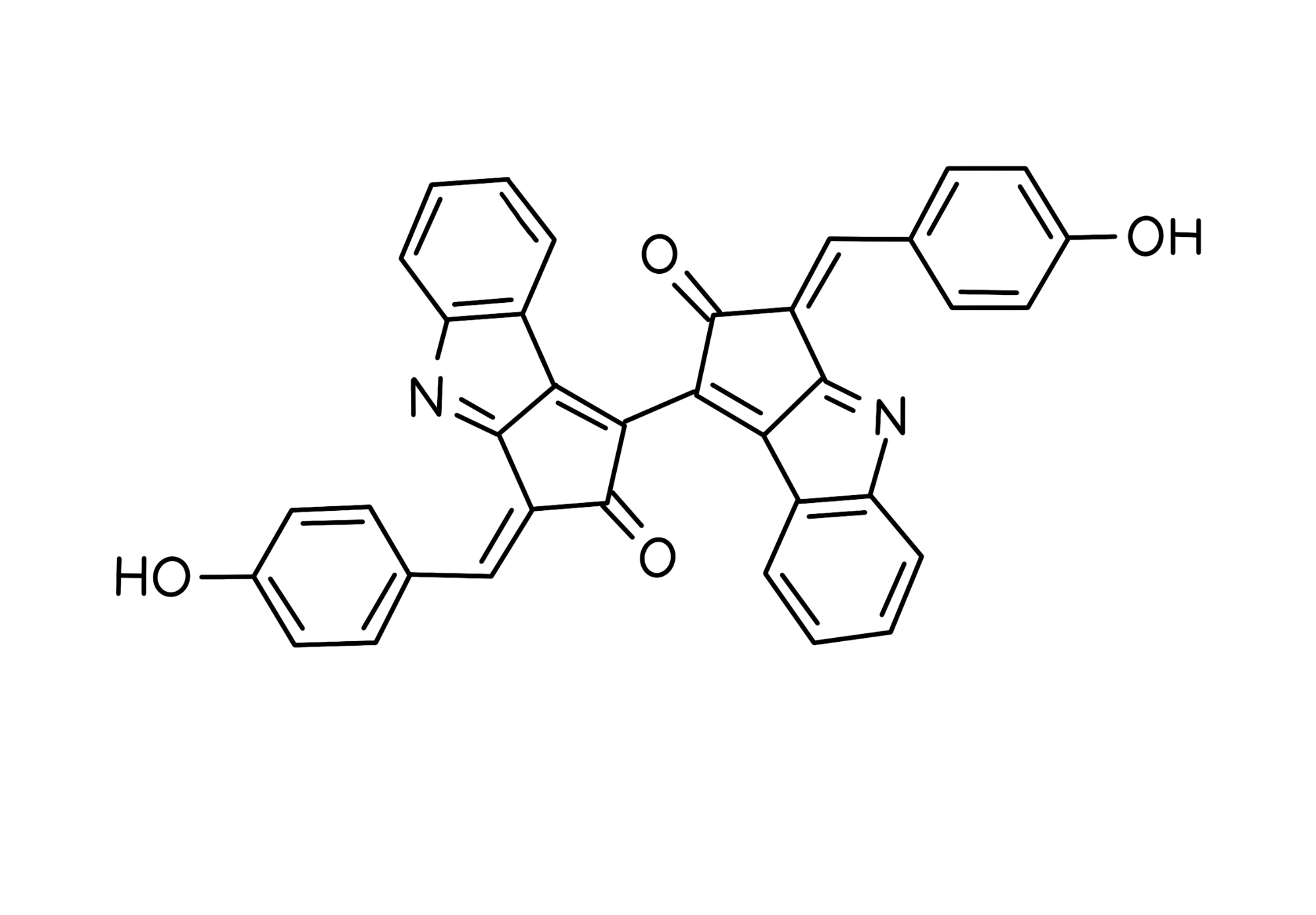
| Figure 3. Chemical Structure of Scytonemin |
|---|
The antioxidative properties of scytonemin and MAAs are necessary to protect biological molecules against oxidative damages induced by UV radiation, along with their harmless effect on the marine environment (Klisch & Häder, 2008). Our study aims to design a safe sunscreen for the consumer and the marine environment. We will insert a synthesized scytonemin biosynthetic gene cluster, scyA-F, into one pET-28a(+) plasmid, while inserting the synthesized mycosporine-glycine biosynthetic gene, MysC, into a second pET-28a(+) plasmid via restriction enzymes and ligase.
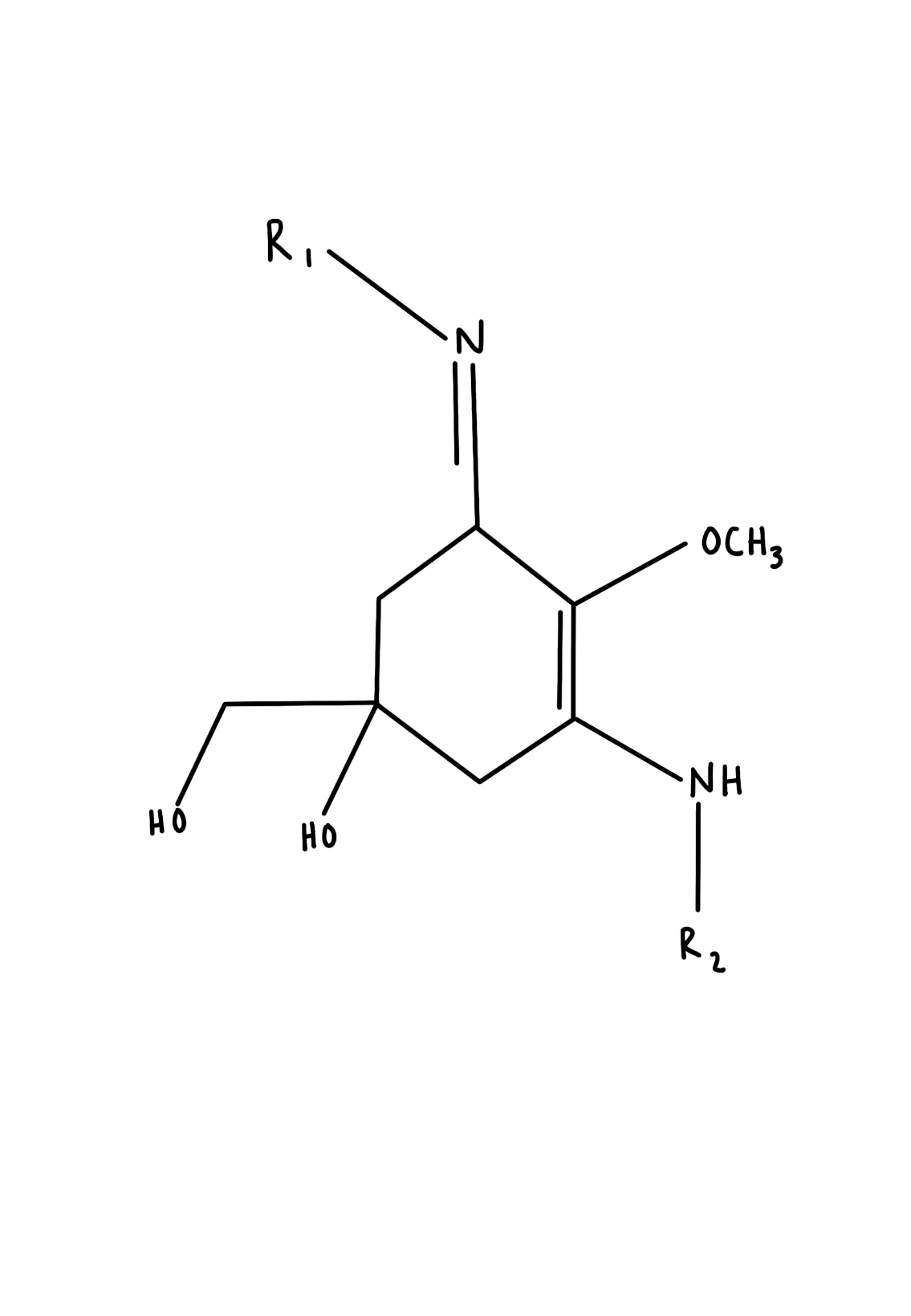
| Figure 4. Chemical Structure of MAA |
|---|
We will use the plasmid pET-28a(+), which contains a T7 promoter, a ribosomal binding site, a T7 terminator, and a gene encoding for kanamycin resistance (Novagen). The bacteria will be plated on nutrient agar with kanamycin in separate petri dishes. Since the kanamycin resistance gene is chosen as a selectable marker, only successfully transformed bacteria will grow on the plate.
We plan to use separate plasmids for each gene due to different purification methods. After the target genes are placed within their respective plasmids, the recombinant plasmids will be transformed via heat shock into separate cultures of competent T7 Express E. coli, which is a strain engineered for high efficiency protein production.
The E. coli containing the scyC-encoding plasmid will undergo a methanol wash protocol, which is known to be successful in extracting metabolites. The media in the E. coli cell culture dish will be removed, purified by methanol wash with water at 4°C, centrifuged, and evaporated. The MAAs purification process will involve High Performance Liquid Chromatography, HPLC, a technique with a high efficiency of separating the amino acids from the culture mixture (Koester, 2016). Once extracted, the MAAs and the scytonemin will be combined with inactive but beneficial ingredients commonly found in other sunscreen products, such as niacinamide, a form of Vitamin B3 that calms irritated skin, and glycerin, a natural moisturizer (Figure 5) (What to Know, n.d.). With this novel approach, we are determined to design an environmentally friendly sunscreen that will benefit both the ecosystem and humans.
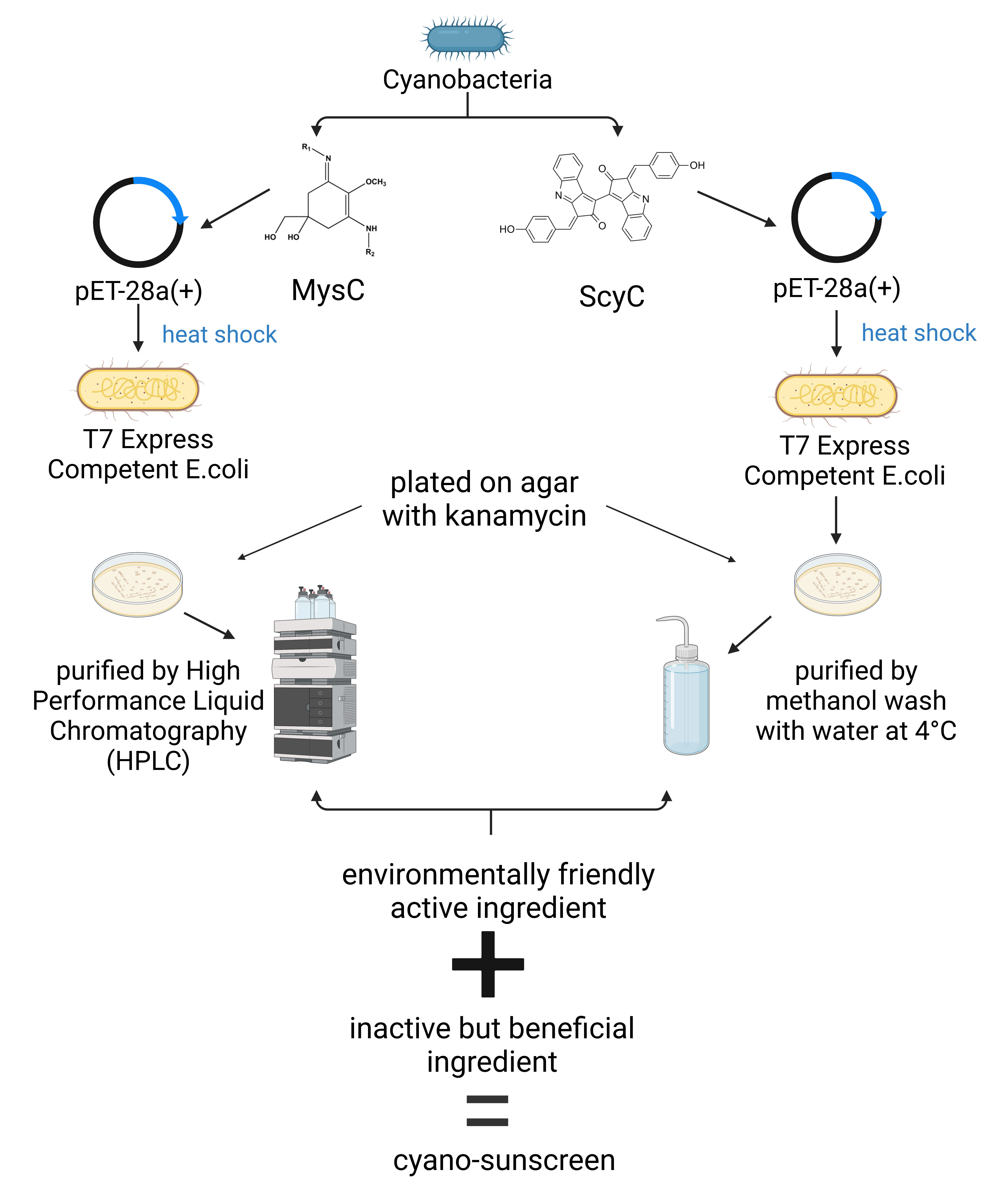
| Figure 5: Cyano-Sunscreen synthesis |
|---|
Systems level
Our design utilizes pigments found in cyanobacteria, one of the largest and most diverse groups of bacteria populating inland waters, to substitute active ingredients in modern chemical sunscreens. The chosen system involves the insertion of genes involved in biosynthesis of two cyanobacterial pigments, mycosporine amino acids and scytonemin, into T7 Express E. coli by subcloning them into the pET-28a(+) plasmid and subsequent transformation. The pET-28a(+) plasmid contains a T7 promoter, a ribosome binding site, T7 terminator, kanamycin resistance, an N-terminal his-tag, and a thrombin tag. There will be 2 unique plasmids, one containing the mycosporine-like amino acid gene (MysC) and the other containing the scytonemin gene (Scy C), along with the gene cluster (Scy A-F). The gene cluster is vital because the production of scytonemin requires each gene for complete protein production. Each plasmid will be transformed into separate T7 Express E. coli competent cells and grown on LB agar plates with kanamycin. The need for two unique plasmids and cell cultures is due to the unique extraction methods necessary to effectively purify the pigments given that one is a protein and the other is a metabolite. After initial growth, a larger vessel will be inoculated with isopropyl-β-d-thiogalactopyranoside (IPTG) which will induce the T7 promoter, allowing for high production. We will use HPLC and a methanol wash method to extract the mycosporine amino acids and scytonemin, respectively. To undergo HPLC, the E. coli will be grown for 15 days, harvested by centrifugation, and lyophilized.
The extraction solution for the cells consists of a 0.2% (v/v) formic acid solution and 2 mM ammonium formate. After adding the extraction solution, the sample is mixed by vortex, stored for 1 hour at room temperature and centrifuged to remove cell debris. The supernatant is then purified by a Shimadzu Prominence High-Performance Liquid Chromatography.A methanol wash method is the preferred method to extract scytonemin from E. coli. The plates with the cells will be immediately placed onto ice (4°C) after incubation at 37°C. After collecting 20 μl of growth media, the plates will be washed with 2 ml H2O, cooled on dry ice, then placed in a -80°C freezer. The solution is then centrifuged, and the supernatant contains the resultant scytonemin. Once extracted, the scytonemin and MAAs will be tested in future experiments for ideal concentrations and mixed with active and beneficial ingredients for the skin.
Device level
The E. coli strain acts as a chassis to aid in producing our target protein and metabolite. T7 Express E. coli was chosen due to its suitability for expression from the T7 promoter found in our plasmid and for the protein production. The production of scytonemin requires the use of a gene cluster consisting of ScyA, B, C, D, E, F. Each gene is necessary for complete protein production. These genes originated from the Nodularia spumigena (strain: UHCC 0039), a type of cyanobacteria. The MysC gene was synthesized from the Cyanobacterium sp. HL-69 bacteria. Our plasmid, pET-28a(+) includes a T7 promoter, ribosomal binding site, a T7 terminator, kanamycin resistance, an N-terminal his-tag, and a thrombin tag. A T7 is a strong promoter with a high transcription rate optimal for protein production.
Parts level
Our team chose a plasmid backbone pET-28a(+) because of its high expression level and its wide application to a range of bacterial expression systems. pET-28(+) is a bacterial expression vector with a T7 promoter, a ribosomal binding site (RBS), a T7 terminator, and a gene coding for kanamycin resistance. The T7 promoter along with an inducer enables the user to have control over the clone of the genes of interest with high selectivity. The RBS is a segment of an mRNA that is responsible for the accuracy during the initiation of protein synthesis. The T7 terminator is a natural occurring terminator that initiates the release of the newly formed RNA. The gene that codes for kanamycin resistance acts as a selectable marker for the users to discern the successfully transformed bacteria since only the desired bacteria containing the plasmid will survive on the agar with kanamycin. The MAA and scytonemin biosynthetic genes are inserted into two separate plasmids using the HindIII (ScyA-F) and EcoRI (MysC) restriction enzyme sites. HindIII and EcoRI were chosen because their corresponding restriction sites exist within the plasmid’s multiple cloning site but would not cut their respective target sequences.
Safety
E. coli has been a primary model organism in scientific research for many decades. T7 Express E. coli is a Biosafety Level 1 organism. In order to ensure safety, personal protective equipment will be required to be worn during experiments, and proper protocol should be followed according to the guidelines set out by the manufacturer.
Cyanobacteria represent a large and diverse group of bacteria, each with varying biohazard safety considerations. In general, small amounts of Cyanobacteria exposure is safe, but overgrowth of cyanobacteria is also known to cause harmful algal blooms (HABs). These algal blooms release toxins into the environment which cannot be consumed by humans and cause the deaths of marine life. It is important that cyanobacteria cultures are properly disposed of when used in a lab to prevent them from affecting local waterways. Although concerns exist regarding the safety of its use, the use of any species of cyanobacteria is not necessary within our study design since the study only requires cyanobacteria pigments.
Our design includes the use of MAA gene MysC and scytonemin gene cluster ScyA-F. Through a variety of studies, MAA and scytonemin have been shown to be ecologically and dermatologically safe. Additional research would have to be done to determine proper concentrations necessary for effective sunscreen treatment while remaining safe.
This study utilizes two purification methods, methanol wash and HPLC, to produce the environmentally active ingredients for the final product cyano-sunscreen. The main concern regarding the methanol wash lies in the use of methanol which is not safe for the skin (Centers for Disease Control and Prevention, 2011). Tests will be performed to find a safe and effective method of separating our target protein and metabolite from methanol that aligns with Environmental Protection Agency guidelines. Generally, methanol can be safely evaporated at room temperature (21°C) under a fume hood to ensure safety. Methanol will be used with caution, including proper PPE as instructed by the manufacturer.
It is critical to dispose of HPLC waste safely as it is regulated by the Environmental Protection Agency as hazardous waste. This is done by using a container or cap designed for HPLC waste collection. The diameter of the existing cap will align with the diameter of the waste line, ensuring a tight fit. The hole will be designed to be suitable for any exhaust filter or air valve tube that may be needed. Solvent reservoirs and waste containers will be maintained airtight. Vapors from HPLC will be avoided in the room at all times as solutions will be prepared in a fume hood. Throughout the experiment, PPE will be worn to ensure lab safety.
Discussions
In future experiments, it will be necessary to determine the specific type of MAA required, as well as the appropriate inactive ingredients. The final product will be a sunscreen that combines scytonemin and MAAs as active ingredients, glycerin (used as a natural moisturizer), dimethicone (which smooths the skin and forms a protective barrier), niacinamide (used to treat acne and protect from environmental stress), and dicaprylyl carbonate (which helps the absorption of other chemicals on the skin) as inactive ingredients. The absorption range of MAAs varies between 310 nm to 362 nm, depending on the specific type of MAA used, as different types of MAAs have varying UV radiation absorption abilities. The ability of MAAs to absorb different wavelengths of UV radiation depends on the extent of amino acid substitution, which is the replacement of one or more amino acids in a protein with another amino acid (Wada et al., 2013). Since MAAs are hydrophilic compounds, water-based formulation ingredients in sunscreen may lead to the degradation of pigments and cause them to lose their absorption efficiency (Klisch & Häder, 2008). In the experimental test section, animal testing will be replaced with silicon models and in vitro methods to examine the effectiveness of the pigments’ radiation-absorbing ability, which will be compared to industry-leading sunscreens. Furthermore, there will be additional tests to determine the environmental safety of the sunscreen. If the experimental results demonstrate the success of our design, one possible application of this project would be to collaborate with skincare brands to promote the use of environmentally friendly sunscreen and mass-produce the “cyano” sunscreen to replace the current, environmentally harmful sunscreens on the market. The produced scytonemin and MAAs could also be applied in other skincare industries beyond sunscreen.
Next steps
The current state of our study is purely theoretical, and we have yet to produce a physical prototype. While it is imperative that we examine the environmental impact of the final product, due to the limitations of our high school synthetic biology lab, we are unable to conduct a comprehensive environmental impact test at this stage. However, we will be able to partially test the sunscreen’s UV protection ability by comparing it to similar sunscreens using UV detection stickers. Additionally, we need to conduct further research on the specific sunscreen ingredients and their concentrations, including both active and inactive ingredients. Moving forward, our immediate focus will be on conducting the intended design and extracting necessary data to evaluate the success of our methods.
Author contributions
Y.M. proposed the original idea and began introductory research. L. A., A. B., M.F., Y.M., and A.N. conducted the early research process. L.A., A.B., M.F., Y.M., A.N., and Y.Z. contributed to the writing and proofreading of the paper. L. A., A. B., M.F., Y.M., A.N., and Y.Z. designed the images and graphics for this project and worked on the video.
Acknowledgements
Our gratitude goes to Western Reserve Academy and their support of our program, including resources and time to pursue our invention. A special acknowledgement is given to our mentor, Dr. Beth Pethel, who we appreciate for introducing us to synthetic biology and cultivating our love for science.
References
American Cancer Society. (n.d.). What is melanoma skin cancer? https://www.cancer.org/cancer/types/melanoma-skin-cancer/about/what-is-melanoma.html
Assunção, J., Amaro, H. M., Malcata, F. X., & Guedes, A. C. (2022). Cyanobacterial pigments: Photosynthetic function and biotechnological purposes. In G. Lopes, M. Silva, & V. Vasconcelos (Eds.), The pharmacological potential of cyanobacteria (pp. 201-256). Academic Press. https://doi.org/10.1016/B978-0-12-821491-6.00008-9
Autier, P. (2000). Do high factor Sunscreens offer protection from melanoma ? The Western Journal of Medicine, 173(1). https://doi.org/10.1136/ewjm.173.1.58
Barnes, P. W., Williamson, C. E., Lucas, R. M., Robinson, S. A., Madronich, S., Paul, N. D., Bornman, J. F., Bais, A. F., Sulzberger, B., Wilson, S. R., Andrady, A. L., McKenzie, R. L., Neale, P. J., Austin, A. T., Bernhard, G. H., Solomon, K. R., Neale, R. E., Young, P. J., Norval, M., . . . Zepp, R. G. (2019). Ozone depletion, ultraviolet radiation, climate change and prospects for a sustainable future. Nature Sustainability, 2(7), 569-579. https://doi.org/10.1038/s41893-019-0314-2
Bennett, P. (2022, May 13). Scientists uncover how sunscreen chemicals become toxic to corals. World Economic Forum. https://www.weforum.org/agenda/2022/05/scientists-uncover-how-sunscreen-chemicals-become-toxic-to-corals/
Better Health Channel. (2021, August 23). Wrinkles. https://www.betterhealth.vic.gov.au/health/conditionsandtreatments/wrinkles
Boerner, L. K. (2021, July 21). What’s in sunscreen, and how does it protect your skin from the sun’s rays?. C&EN. https://cen.acs.org/business/consumer-products/What-in-sunscreen-and-how-does-it-protect-your-skin-from-the-sun-rays/99/i27
Bramlet, K. (2016, June). How does sunscreen work?. MD Anderson Cancer Center. https://www.mdanderson.org/publications/focused-on-health/how-sunscreen-works.h27Z1590624.html
Brown, T. M., & Krishnamurthy, K. (2022). Histology, dermis. StatPearls Publishing.
Center for Biological Diversity. (2021, September 9). Coalition petitions federal government to ban coral-Killing chemicals in sunscreens push for nationwide ban follows Hawai’i law outlawing toxic sunscreens. https://biologicaldiversity.org/w/news/press-releases/coalition-petitions-federal-government-to-ban-coral-killing-chemicals-in-sunscreens-2021-09-09/
Centers for Disease Control and Prevention. (2011, May 12). Methanol: Systemic agent. https://www.cdc.gov/niosh/ershdb/emergencyresponsecard_29750029.html
FDA. (2019, February) .FDA proposes sunscreen regulation changes https://www.fda.gov/media/124654/download
Griffin, L. L., Ali, F. R., & Lear, J. T. (2016). Non-melanoma skin cancer. Clinical Medicine Journal, 16(1), 62-65. https://doi.org/10.7861%2Fclinmedicine.16-1-62
Hölzle, E., & Hönigsmann, H. (2005). UV‐radiation–Sources, Wavelength, Environment. Journal of the German Society of Dermatology, 3(S2), S3-S10. https://doi.org/10.1111/j.1610-0387.2005.04392.x
Johns Hopkins Medicine. (n.d.). Sunscreen and your morning routine. https://www.hopkinsmedicine.org/health/wellness-and-prevention/sunscreen-and-your-morning-routine
Kaur, H. (2019, February 6). Key West bans certain sunscreens to protect coral reef. CNN. https://www.cnn.com/2019/02/06/us/key-west-bans-sunscreens-to-protect-reef/index.html
Klisch, M., & Häder, D. P. (2008). Mycosporine-like amino acids and marine toxins-The common and the different. Marine Drugs, 6(2), 147-163. https://doi.org/10.3390/md6020147
Koester, V. (2016, June 20). What is HPLC?. ChemistryViews. https://www.chemistryviews.org/details/education/9464911/What_is_HPLC/
Kogej, T., Gostinčar, C., Volkmann, M., Gorbushina, A. A., & Gunde-Cimerman, N. (2006). Mycosporines in extremophilic fungi—Novel complementary osmolytes?. Environmental Chemistry, 3(2), 105-110. https://doi.org/10.1071/EN06012
Lewis, C. (2020, January 10). Just follow the ‘butterfly’. ASU research team uses scientific sleuthing skills to uncover a biology-backed timeline of the earth’s great oxygenation event. ASU. https://news.asu.edu/20200110-just-follow-butterfly
Markman, M. (2022, July 20). Melanoma. City of Hope. https://www.cancercenter.com/cancer-types/melanoma
Muzic, J. G., Schmitt, A. R., Wright, A. C., Alniemi, D. T., Zubair, A. S., Lourido, J. M. O., Seda, I. M. S., Weaver, A. L., & Baum, C. L. (2017). Incidence and trends of basal cell carcinoma and cutaneous squamous cell carcinoma. Mayo Clinic Proceedings. 92(6), 890-898. https://doi.org/10.1016/j.mayocp.2017.02.015
National Oceanic and Atmospheric Administration. (2015, July 6). Coral bleaching threat increasing in Western Atlantic and Pacific Oceans. https://www.noaa.gov/media-release/coral-bleaching-threat-increasing-in-western-atlantic-and-pacific-oceans
Pandika, M. (2019, January 22). Common sunscreen ingredient octocrylene might be harmful to coral. Axial. https://axial.acs.org/analytical-chemistry/common-sunscreen-ingredient-octocrylene-might-be-harmful-to-coral
Ray, P. D., Huang, B. W., & Tsuji, Y. (2012). Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cellular Signalling, 24(5), 981-990. https://doi.org/10.1016/j.cellsig.2012.01.008
Richard, E. G. (2022, July 1). All about sunscreen. Why you need it: How it works for you. Skin Cancer Foundation. https://www.skincancer.org/skin-cancer-prevention/sun-protection/sunscreen
Rosic, N. N. (2019). Mycosporine-like amino acids: Making the foundation for organic personalised sunscreens. Marine drugs, 17(11), 638. https://doi.org/10.3390/md17110638
Ruszkiewicz, J. A., Pinkas, A., Ferrer, B., Peres, T. V., Tsatsakis, A., & Aschner, M. (2017). Neurotoxic effect of active ingredients in sunscreen products, a contemporary review. Toxicology Reports, 4, 245-259. https://doi.org/10.1016/j.toxrep.2017.05.006
Shields, H. J., Traa, A., & Van Raamsdonk, J. M. (2021). Beneficial and detrimental effects of reactive oxygen species on lifespan: A comprehensive review of comparative and experimental studies. Frontiers in Cell and Developmental Biology, 9, 181. https://doi.org/10.3389/fcell.2021.628157
Shick, J. M., Dunlap, W. C., Chalker, B. E., Banaszak, A. T., & Rosenzweig, T. K. (1992). Survey of ultraviolet radiation-absorbing mycosporine-like amino acids in organs of coral reef holothuroids. Marine Ecology Progress Series, 90(2), 139-148.
Sigma-Aldrich. (n.d.). pET-28a(+) DNA-Novagen. Merck. https://www.emdmillipore.com/US/en/product/pET-28a-DNA-Novagen,EMD_BIO-69864
Smith, A. (2018, May 24). Is your sunscreen killing the coral reef?. Ocean Conservancy https://oceanconservancy.org/blog/2018/05/24/sunscreen-killing-coral-reef/
Singh, A., Čížková, M., Bišová, K., & Vitova, M. (2021). Exploring mycosporine-like amino acids (MAAs) as safe and natural protective agents against UV-induced skin damage. Antioxidants, 10(5), 683. https://doi.org/10.3390/antiox10050683
Stern, R. S. (2010). Prevalence of a history of skin cancer in 2007: Results of an incidence-based model. Archives of Dermatology, 146(3), 279-282. https://doi.org/10.1001/archdermatol.2010.4
U.S. Food & Drug Administration. (2020, August 19). Ultraviolet (UV) Radiation. https://www.fda.gov/radiation-emitting-products/tanning/ultraviolet-uv-radiation
Vincent, W. F. (2009). Cyanobacteria. In G. E. Likens (Ed.), Encyclopedia of inland waters (pp. 226-232). Academic Press. https://doi.org/10.1016/B978-012370626-3.00127-7
Wada, N., Sakamoto, T., & Matsugo, S. (2015). Mycosporine-like amino acids and their derivatives as natural antioxidants. Antioxidants, 4(3), 603-646. https://doi.org/10.3390/antiox4030603
Wada, N., Sakamoto, T., & Matsugo, S. (2013). Multiple roles of photosynthetic and sunscreen pigments in cyanobacteria focusing on the oxidative stress. Metabolites, 3(2), 463-483. https://doi.org/10.3390/metabo3020463
WedMD. (n.d.). What to know about niacinamide skin care. Retrieved January 19, 2023, from https://www.webmd.com/beauty/what-to-know-about-niacinamide-skin-care
Whitehead, K., & Vernet, M. (2000). Influence of mycosporine‐like amino acids (MAAs) on UV absorption by particulate and dissolved organic matter in La Jolla Bay. Limnology and Oceanography, 45(8), 1788-1796. https://doi.org/10.4319/lo.2000.45.8.1788
World Health Organization. (2017, October 16). Radiation: Ultraviolet (UV) radiation. https://www.who.int/news-room/questions-and-answers/item/radiation-ultraviolet-(uv) Worldwide Cancer Research. (2022, March 1). How does the sun cause cancer?. https://www.worldwidecancerresearch.org/news-opinion/2022/march/how-does-the-sun-cause-skin-cancer/


