Junning Hu, Marianna Jocas, Jordanne Nichols, and Tanushree Patil ▫ Western Reserve Academy, Hudson, Ohio, United States
Reviewed on 6 May 2023; Accepted on 13 July 2023; Published on 16 October 2023
With help from the 2023 BioTreks Production Team.
Vitamin D deficiency is a global problem that is linked to a wide range of chronic conditions such as osteoporosis, muscle weakness, and fatigue. Few foods naturally produce adequate vitamin D concentrations and vitamin D supplements do not fully alleviate the symptoms of the deficiency. In recent years, scientists have used biofortification to enhance vitamin levels in food. Our team looked towards a group of scientists who biofortified the tomato to increase its vitamin D concentration levels. In tomatoes, the 7-DR2 gene product transforms the 7 Dehydrocholesterol (7-DHC) into cholesterol as a part of the SGA synthesis. The tomato and the bell pepper are part of the Solanaceae family, which share the same genomic pathways. Bell peppers are a commonly consumed vegetable, easily accessible, and are a good source of vitamins A and C, potassium, folic acid, and fiber, making them an ideal candidate for genetic modification. Using Agrobacterium-mediated transformation, a genome editing technology, we intend to introduce CRISPR-Cas9 into the bell pepper and knock out the 7-DR2 gene. 7-DR2 turns 7-DHC, the precursor to vitamin D, into cholesterol if not changed or blocked. In doing this, vitamin D would accumulate in the vegetable. Hence, the vitamin D concentration in the bell pepper will increase and become a natural supplement for vitamin D deficiency.
Keywords: Vitamin D deficiency, biofortification, 7-DR2, 7-Dehydrocholesterol, CRISPR-Cas9
Authors are listed in alphabetical order. Beth Pethel from Western Reserve Academy, Hudson, Ohio, United States mentored the group. Please direct all correspondence to .
Approximately one billion people are affected by vitamin D deficiency worldwide, and 50% of the world’s population is vitamin D insufficient (Siddiqee, 2021). Vitamin D restores and maintains calcium in bones and improves phosphorus absorption in the body (Vitamin D, 2022). One of the most common forms of vitamin D is vitamin D3, which is produced upon exposure to the sun’s ultraviolet rays. Insufficient amounts of vitamin D or vitamin D3 can cause fatigue, mood changes, muscle weakness, and dizziness (Mayo Clinic, 2022). In some cases, osteoporosis develops, where bones are so delicate that mild stresses such as bending over or coughing can cause a fracture (De Martines et al., 2021).
Several countries report vitamin D deficiency within their population. India, Pakistan, and Iran’s populations are most deficient, with more than 20% of the population having less than 12 ng/ml in their diet. In India alone, it has been estimated that 490 million people are deficient (Cashman et al., 2016). This is caused by the low concentrations of vitamin D in natural foods and because many countries are stifled without resources, unable to get sufficient amounts of vitamin D. Communities without widespread access to vitamin supplements require large quantities of fish and eggs to meet adequate vitamin D concentrations (Calvo et al., 2005, p. 312). However, even diets that are primarily meat and egg-based can only account for 2.7–5.3 micrograms (𝜇g)/day of vitamin D. The recommended intake is 20 𝜇g/day, leaving people around 14.7 𝜇g/day deficient (Vitamin D – Health Professional Fact Sheet, 2022). Additionally, as veganism becomes increasingly popular, with approximately 79 million people worldwide who identify, it poses a risk, as most sources rich in natural vitamin D include meat and dairy products (Vegan and Plant-Based, 2022). This shift towards veganism leaves more than one percent of the world’s population with a severe vitamin D deficiency.
These issues are most commonly attempted to be solved through vitamin D supplements. However, recent meta-analyses show that supplements are not significant in preventing the effects of vitamin D deficiency, such as bone fragility (LeBoff et al., 2022). The lack of vitamin D naturally occurring in foods and supplements highlights the need for a natural, accessible, and inclusive food supplement that helps target the deficiency.
In recent years, scientists have used biofortification to enhance vitamin levels in food. Biofortification is the process of breeding crops to contain higher levels of vitamins, minerals, and proteins. Some examples include rice, which has been biofortified to address iron deficiency, and sweet potatoes, which have had their antioxidant properties increased (Garg, et al., 2018). As a basis for our project, we followed the scientists who biofortified tomatoes —a fruit which is a part of the Solanaceous family— to target vitamin D deficiency. In this study, scientists used the gene-editing technology, CRISPR-Cas 9, which modifies, deletes, or corrects specific DNA regions, to alter 7-DHC, the precursor to vitamin D, in tomatoes to increase vitamin D levels (Guardian News, 2022). They altered the 7-DR2 gene because it turns 7-DHC into cholesterol if it is not altered or blocked, which allows vitamin D3 to accumulate in the leaves (Medline Plus, 2022). Once modified, the plants were exposed to sunlight to convert the vitamin levels stored in the leaves into active vitamins (Guardian News, 2022).
While the scientist’s research solely focused on tomatoes, this method could be applied to other foods in the Solanaceous family, like peppers, chilies, and potatoes. All Solanaceae food pathways are similar, but the one closest to the tomato is the bell pepper. The bell pepper, Capsicum annuum, are also low in calories and high in vitamins A and C, making them a perfect addition to a healthy diet (Arnarson, 2023). They are greatly accessible as they are inexpensive, readily available, and easily grown in various climates.
Within the United States and the global market, bell peppers account for 70.51% of all vegetable production as of 2020 (Karst, 2022). From 2000 to 2021, more than 70% of the world’s bell peppers were produced in Asia (Biswas et al., 2018). However, this is closely followed by Mexico, Spain, Indonesia, and the United States as fellow large producers (Biswas et al., 2018).
The generic price of a bell pepper is 0.55 USD per kg, allowing it to be bought in mass amounts at low prices (2022 fresh, 2022). Consequently, with the global scale of bell pepper production and availability in various different climates, the bell pepper is the most necessary vegetable to target for vitamin D experimentation. Therefore, with this project, the 1.94-billion-dollar industry can expand economically and develop genetically (2022 fresh, 2022).
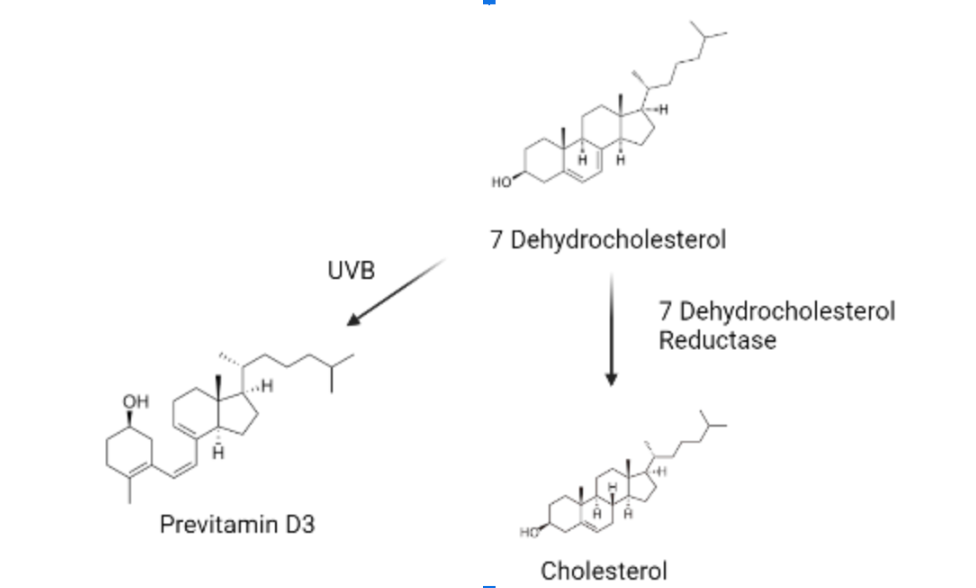
As referenced in Figure 1, 7 Dehydrocholesterol (7-DHC) is the intermediate product of the steroidal glycoalkaloid (SGA) synthesis, which is a pathogen defense mechanism developed by the Solanaceous species (Cárdenas et al., 2016, p. 1). In the pathway of SGA synthesis, 7-DHC is first transformed into cholesterol and then into the SGA. In this process, an enzyme called 7-dehydrocholesterol reductase 2 (7-DR2) makes the transformation reaction occur and controls the reaction rate (Sonawane et al., 2016, p. 2). Wavelengths between 290–315 nm are necessary for the UVB ray in sunlight to induce the photolysis of 7-DHC and transform it into 7-DR2 instead of cholesterol (Figure 1) (Jäpelt & Jakobsen, 2013, p. 2; Li et al., 2022).
The CRISPR-Cas 9 complex, shown in Figure 2, comprises the Cas-9 enzyme and single guide RNA (sgRNA). The sgRNA combines with the complementary target sequence by matching the paired nucleotide bases. The Cas-9 enzyme is activated by the combined structure between sgRNA and the target sequence and then cuts the target sequence, breaking the hydrogen bond between nucleotides so that the gene sequence is not transcribed and expressed (Ran et al., 2013, p. 2).
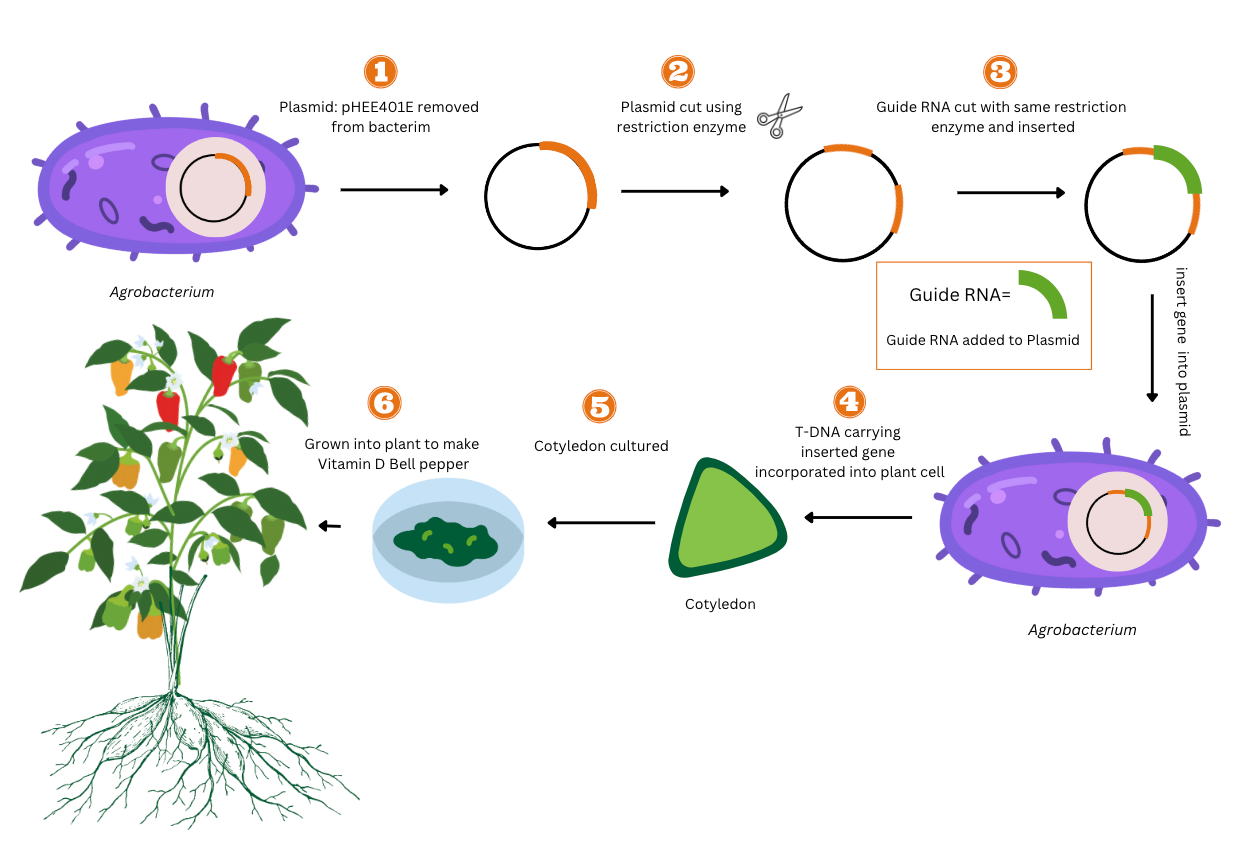
Agrobacterium-mediated transformation is a common technique for scientists to edit a plant’s gene. Agrobacterium is a bacterium that induces tumors on infected plants (Nester, 2008.). Scientists insert the desired gene into the Agrobacterium plasmid and then infect the target plant with Agrobacterium. Hence, the Agrobacterium will incorporate the desired gene into the plant gene and achieve gene editing (Song et al., 2019, p. 8).
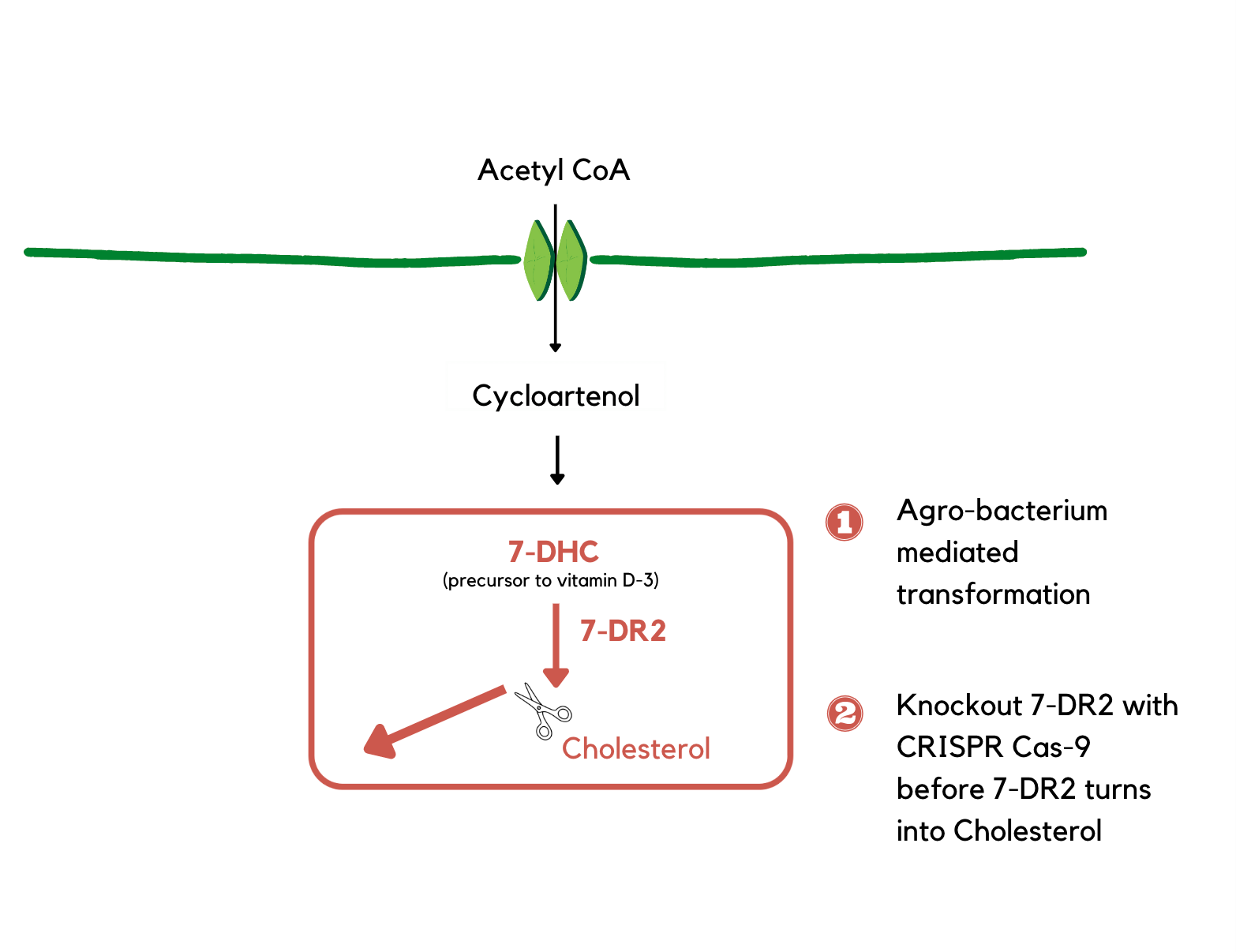
Our team aims to use Agrobacterium-mediated transformation to insert the plasmid expressing CRISPR-Cas 9 and sgRNA into a bell pepper to knock out the gene of the 7-DR2 enzyme. This would lead to the downregulation of the 7-DR2 enzyme, which prevents 7-DHC from transforming into cholesterol and causes 7-DHC to accumulate in the bell pepper. The knockout can also decrease the synthesis of SGA in bell pepper, reducing the toxicity of the bell pepper leaves to humans. When accumulated, 7-DHC is exposed to UVB and transformed into pre-vitamin D3. This process would enrich the concentration of pre-vitamin D3 in the bell pepper. Concentration levels will be measured through plant mass spectrometry to ensure the safety of consumption. Once determined to be within healthy ranges, the bell pepper will be made accessible. Thus, people who consume peppers can supplement the vitamin D concentration in their bodies more naturally.
Systems level
By knocking out the 7-DR2 enzyme with CRISPR-Cas 9, 7-DHC is prevented from transforming into cholesterol and causes vitamin D to accumulate in the bell pepper.
Device level
Agrobacterium is a pathogenic soil bacterium that infects the host plant to develop a large tumor (Kroemer, n.d.). The tumor-inducing properties carry the virulence (vir) genes to encode the enzyme (Kroemer, n.d.). Once the Agrobacterium detects the plant cells, it activates its chromosomal genes to attach to the host plant cell. The vir genes attach to the cleavage and transfer T-DNA (Kroemer, n.d.). The enzymes in the T-DNA synthesize amino acids and provide the Agrobacterium with its essential nutrients (Kroemer, n.d.). Once Agrobacterium is inserted, it incubates the cells and infects the host with bacteria. This infection wounds the plant surface and allows for gene-mediated delivery.
In our project, GV3101, with a disarmed C58 chromosomal backbone, is inserted into the cotyledon stage of the bell pepper. A functional T-binary system, the method in which two vectors transform plants, will be built with the GV3101 strain. The T-complex is a molecular chaperone complex that assists with the folding of proteins upon ATP hydrolysis. The VirD2 and VirE2 will constitute the ‘T Complex’ with the T strand. The T-strand will channel the bacterial membrane to penetrate the plant’s DNA. (Gelvin, 2006).
Due to T-DNA’s missing region, the Ti plasmid is deleted, allowing for the transfer of genetic material into the bell peppers genome. Agrobacterium will export the T-DNA into eukaryotic cells as a single-strand molecule called the T-strand. Within the Agrobacterium, VirA and VirG proteins function as sensory signals in the regularity system and sense phenolic compounds (Gelvin, 2003). These compounds are metabolites produced in the acid of plants through phenylpropanoid metabolization. In an appropriate phenolic compound and sugar concentration, the viral gene will autophosphorylation, a biochemical process where phosphate is added to a protein kinase. Then the VirG gene will transphosphophorylate, which is the process of a phosphate group transferring between receptors and the substrates. The VirG gene will then attach to the vir box and activate vir gene. This activates the VirD4 and all 11 VirB proteins to transfer the T-DNA and link the promoter. The VirB proteins will form a membrane channel and export the DNA for molecular transfer. Once inside the bell pepper, VirD2 will direct the attached T-DNA to the plant nucleus. This fuses the vitro T-complex with the nuclei and starts the temperature-regulated transformation. Thus, the T-binary system will transgenically express the plasmid in the bell pepper’s nuclei.
Parts level
We will gather the sequence of bell pepper from EnsemblPlants and find SGA pathway similar to tomatoes. In the SGA synthesis, the 7-Dehydrocholesterol (7-DHC) will be transformed by the 7-Dehydroreductase 2 (7-DR2) first into cholesterol then α-solanine, a chemical produced by bell pepper to protect itself from pests. An example of the bell pepper gene sequence is shown in Figure 3.

| Figure 3. The figure shows a part of the 7-DR2 gene sequence. |
|---|
Our group chose Hygromycin as the selectable marker. It is an aminocyclitol antibiotic inhibiting the growth of both prokaryotes and eukaryotes, which makes it a desired selectable marker for Agrobacterium-mediated transformation (Zheng et al., 1991). The Hygromycin-resistant gene is integrated into the T-DNA of the plasmid.
The gene editing technology that we are going to use is CRISPR-Cas9. This technology was adapted from the gene editing system used by bacteria as an immune defense. CRISPR arrays are segments created when the bacteria capture pieces of the virus’s DNA and inserts them into their own DNA in a pattern. These arrays allow the bacteria to remember the viruses and produce RNA segments that attach to the viruses’ DNA when they recognize the virus again (Medline Plus, 2022). The Cas9 enzyme then segments the DNA, which disables the virus. In our project, the CRISPR-Cas 9 complex comprises the Cas-9 enzyme and a single guide RNA (sgRNA). The guide RNA is a small piece of the RNA sequence with a short ‘guide,’ which is a sequence that binds to a specific target sequence in a cell’s DNA. The guide RNA recognizes the DNA sequence when inserted in a cell, and then the Cas9 enzyme cuts the DNA at the target location. The cell’s DNA repair machinery can be used to add, remove or edit genetic material and so after the DNA is cut the machinery is used to replace an existing sequence with a customized DNA sequence. Next, the sgRNA combines with the complementary target sequence by matching the paired nucleotide bases. Then, the Cas-9 enzyme is activated by the combined structure between sgRNA and the target sequence and then cuts the target sequence, breaking the hydrogen bond between nucleotides so that the gene sequence is not transcribed and expressed (Ran et al., 2013, p. 2).
We plan to genetically modify green bell peppers (Capsicum annuum) to increase their vitamin D levels, as the pathways are similar to tomatoes. The 7-DHC levels are also higher in green bell peppers, which means they will produce more vitamin D as it is its precursor (Figure 4).
Safety
Genetically modified plants are classified as transgenic plants. Therefore, the bell pepper is transgenic because of the Agrobacterium transformation and CRISPR CAS-9 technology. While transgenic plants are beneficial in improving health and lowering costs, they pose a risk to the surrounding environment. Exacerbated weed problems, reduced diversity, and indigenous displacement are potential hazards created by the proximity of transgenic plants (Bauer-Panskus et al., 2020). However, our bell peppers will be grown in spatiotemporally controlled environments to combat this. The raised beds that grow bell peppers will be spatially separated to minimize cross-contamination. Additionally, staggered planting dates and physical barriers will be added to limit gene flow.
As for safe consumption, we plan to use a Polymerase Chain Reaction test (PCR test) and Sanger DNA Sequencing to test the genetically engineered bell pepper. The PCR test will detect foreign DNA if inconsumable. Whereas Sanger DNA Sequencing will identify the mutations, and it will sequence the DNA.
Discussions
To prevent the overproduction of vitamin D in bell pepper, further research will be needed to ensure the safe consumption of vitamin D-fortified bell pepper. An overdose of vitamin D can cause nausea, vomiting, and frequent urination.
Moreover, the knockout of the 7-DR2 gene interferes with the SGA synthesis, a defensive mechanism of the bell pepper to protect itself from pests. The removal of SGA from bell pepper could possibly lower the resistance of bell pepper to a pathogen or pest. We may need to study the impact of removing the 7-DR2 gene from the bell pepper and find a possible replacement.
Our group hopes to help the development of a natural nutrient supplementation to offer a more effective treatment for vitamin D deficiency with better taste. Our biofortification method can be employed in many other plants in the Solanaceae family, such as the potato, eggplant, and tomato. These plants can also be developed as alternatives to meat diets to offer a reliable but low-cost treatment for impoverished people with vitamin D deficiency.
Next steps
Moving forward, the next step is to begin the project’s experiments. Materials such as agar, Petri dishes, green bell pepper, incubator, PCR machine, Crispr-Cas9, and Agrobacterium are needed in the lab. After understanding lab safety and having mastered skills such as streaking, the experiment can begin. Then the genetically modified bell pepper’s safety for consumption would need to be checked. After the experiment is successfully completed, the “Results and Conclusions” section should be written and included in the paper along with images and figures.
Author contributions
J.H. worked on the background research required for the project. J.H. was responsible for the sections: 7-D2 gene, and CRISPR-cas9, and worked with T.P. on parts. M.J. created the graphs and figures and edited the paper. Additionally, M.J. was behind the creation of the video and worked with T.P. on the abstract. J.N. wrote the introduction and device level and worked with T.P. on safety. J.N. also created and checked the references. T.P. worked on writing the vitamin D section, Abstract, and Parts Level and focused on the revisions and editing of the paper. While all members engaged in the background.
Acknowledgments
We would like to give our most gracious thanks to our mentor Dr. Pethel who gave us plans for the development of the project and advice on the revision of the manuscript. We would also like to thank Dylan Wu for helping us initiate the preliminary research of the project.
References
Arnarson, A. (2023, February 27). Bell peppers 101: Nutrition facts and health benefits. Healthline. https://www.healthline.com/nutrition/foods/bell-peppers#downsides
Bauer-Panskus, A., Miyazaki, J., Kawall, K., & Then, C. (2020). Risk assessment of genetically engineered plants that can persist and propagate in the environment. Environmental Sciences Europe, 32(1), 1-15. https://doi.org/10.1186/s12302-020-00301-0
Biswas, T., Guan, Z., & Wu, F. (2018). An Overview of the US bell pepper industry (Publication No. FE1028. UF/IFAS. https://edis.ifas.ufl.edu/publication/FE1028
Calvo, M. S., Whiting, S. J., & Barton, C. N. (2005). Vitamin D intake: A global perspective of current status. The Journal of Nutrition, 135(2), 310-316. https://doi.org/10.1093/jn/135.2.310
Cárdenas, P. D., Sonawane, P. D., Pollier, J., Vanden Bossche, R., Dewangan, V., Weithorn, E. ,Tal, L., Meir, S., Rogachev, I., Malitsky, S., Giri, A. P., Goossens, A., Burdman S., & Aharoni, A. (2016). GAME9 regulates the biosynthesis of steroidal alkaloids and upstream isoprenoids in the plant mevalonate pathway. Nature Communications, 7(1), 10654. https://doi.org/10.1038/ncomms10654
Cashman, K. D., Dowling, K. G., Škrabáková, Z., Gonzalez-Gross, M., Valtueña, J., De Henauw, S., Moreno, L., Damsgaard, C. T., Michaelsen, K. F., Mølgaard, C., Jorde, R., Grimnes, G., Moschonis, G., Mavrogianni, C., Manios, Y., Thamm, M., Bm Mensink, G., Rabenberg, M., Busch, M. A., …Kiely, M. (2016). Vitamin D deficiency in Europe: pandemic?. The American Journal of Clinical Nutrition, 103(4), 1033-1044. https://doi.org/10.3945/ajcn.115.120873
De Martinis, M., Allegra, A., Sirufo, M. M., Tonacci, A., Pioggia, G., Raggiunti, M., GinaIdi, L., & Gangemi, S. (2021). Vitamin D deficiency, osteoporosis and effect on autoimmune diseases and hematopoiesis: A review. International Journal of Molecular Sciences, 22(16), 8855. https://doi.org/10.3390/ijms22168855
FoodChain Testing. (n.d.). GMO testing options. (n.d.). https://gmotesting.com/testing-options/
Garg, M., Sharma, N., Sharma, S., Kapoor, P., Kumar, A., Chunduri, V., & Arora, P. (2018). Biofortified crops generated by breeding, agronomy, and transgenic approaches are improving lives of millions of people around the world. Frontiers in Nutrition, 5, 12. https://doi.org/10.3389/fnut.2018.00012
Geddes. L. (2022, May 23). Scientists create tomatoes genetically edited to bolster vitamin D Levels. The Guardian. https://www.theguardian.com/science/2022/may/23/scientists-create-tomatoes-genetically-edited-boost-vitamin-d-levels
Gelvin, S. B. (2003). Agrobacterium-mediated plant transformation: The biology behind the “gene-jockeying” tool. Microbiology and Molecular Biology Reviews, 67(1), 16-37. https://doi.org/10.1128/MMBR.67.1.16-37.2003
Gelvin, S. B. (2006). Agrobacterium virulence gene induction. In K. Wang (Ed.), Agrobacterium Protocols (pp. 77-85). Springer. https://doi.org/10.1385/1-59745-130-4:77
Gelvin, S. B. (2012). Traversing the cell: Agrobacterium T-DNA’s journey to the host genome. Frontiers in Plant Science, 3, 52 https://doi.org/10.3389/fpls.2012.00052
Jäpelt, R. B., & Jakobsen, J. (2013). Vitamin D in plants: A review of occurrence, analysis, and biosynthesis. Frontiers in Plant Science, 4, 136 https://doi.org/10.3389/fpls.2013.00136
Karst, T. (2022, May 27). Fresh bell pepper per-capita consumption keeps climbing. The Packer. https://www.thepacker.com/news/produce-crops/fresh-bell-pepper-capita-consumption-keeps-climbing
Kroemer, T. (n.d.). A quick overview of Agrobacterium for plant transformation. GoldBio. https://goldbio.com/articles/article/a-quick-overview-of-agrobacterium-for-plant-transformation
LeBoff, M. S., Chou, S. H., Ratliff, K. A., Cook, N. R., Khurana, B., Kim, E., Cawthon, P. M., Bauer, D. C., Black, D., Gallagher, J. C., Lee, I., Min., Buring, J. E., & Manson, J. E. (2022). Supplemental Vitamin D and incident fractures in midlife and older adults. New England Journal of Medicine, 387(4), 299–309. https://doi.org/10.1056/nejmoa2202106
Li, J., Scarano, A., Gonzalez, N. M., D’Orso, F., Yue, Y., Nemeth, K., Saalbach, G., Hill, L., Martins, C. D., Moran, R., Santino, A., & Martin, C. (2022). Biofortified tomatoes provide a new route to vitamin D sufficiency. Nature Plants, 8(6), 611-616. https://doi.org/10.1038/s41477-022-01154-6
Mayo Clinic. (2022, January 18). Vitamin deficiency anemia – Symptoms and causes. https://www.mayoclinic.org/diseases-conditions/vitamin-deficiency-anemia/symptoms-causes/syc-20355025
Medline Plus. (2022, March 2022). What are genome editing and CRISPR-Cas9?. https://medlineplus.gov/genetics/understanding/genomicresearch/genomeediting/
Nester, E. (2008). Agrobacterium: The natural genetic engineer 100 years later. American Phytopathological Society. https://doi.org/10.1094/APSnetFeatures-2008-0608
Ran, F. A., Hsu, P. D., Wright, J., Agarwala, V., Scott, D. A., & Zhang, F. (2013). Genome engineering using the CRISPR-Cas9 system. Nature Protocols, 8(11), 2281-2308. https://doi.org/10.1038/nprot.2013.143
Siddiqee, M. H., Bhattacharjee, B., Siddiqi, U. R., & MeshbahurRahman, M. (2021). High prevalence of vitamin D deficiency among the South Asian adults: A systematic review and meta-analysis. BMC Public Health, 21, 1-18. https://doi.org/10.1186/s12889-021-11888-1
Sonawane, P. D., Pollier, J., Panda, S., Szymanski, J., Massalha, H., Yona, M., Unger, T., Malitsky, S., Arendt, P., Pauwels, L., Almekias-Siegl, E., Rogachev, I., Meir, S., Cárdenas, P. D., Masri, A., Petrikov, M., Schaller, H., Schaffer, A. A., Kamble, A.,…Aharoni, A. (2017). Plant cholesterol biosynthetic pathway overlaps with phytosterol metabolism. Nature Plants, 3(1), 1-13. https://doi.org/10.1038/nplants.2016.205
Song, G. Q., Prieto, H., & Orbovic, V. (2019). Agrobacterium-mediated transformation of tree fruit crops: Methods, progress, and challenges. Frontiers in Plant Science, 10, 226. https://doi.org/10.3389/fpls.2019.00226
Tridge. (2022). 2022 fresh bell pepper global market overview Retrieved January .17, 2023, from https://www.tridge.com/intelligences/bell-pepper
Soylent. (n.d.). 2023 Vegan and plant-based diet statistics. Retrieved May 9, 2023, from https://soylent.com/pages/vegan-statistics
Sizar, O., Khare, S., Goyal, A., Amy Givler, A. (2022). Vitamin D deficiency. StatPearls Publishing. Zheng, Z., Hayashimoto, A., Li, Z., & Murai, N. (1991). Hygromycin resistance gene cassettes for vector construction and selection of transformed rice protoplasts. Plant Physiology, 97(2), 832-835. https://doi.org/10.1104/pp.97.2.832