Danyan Li▫ Western Reserve Academy, Hudson Ohio, United States
Reviewed on 6 May 2023; Accepted on 13 July 2023; Published on 16 October 2023
With help from the 2023 BioTreks Production Team.
Presbyopia, or age-related farsightedness, is the most common eye disease. It is a hindrance to the day-to-day activities of more than 1.8 billion people around the globe. Presbyopia is caused by a decrease in the concentration of alpha-crystallin and the accumulation of oxidative stress in the lens. This oxidative stress results in the hardening of the lens of the eye, resulting in difficulty focusing on objects nearby. The daily intake of antioxidants—specifically, bioflavonoids—prevents the hardening of the lens. However, humans do not produce flavonoids naturally. Quercetin, a type of bioflavonoid, is recognized for its antioxidant, anti-inflammatory, and anti-carcinogenic properties. Quercetin produced by Bifidobacterium in the gut may be able to travel from the gut to the eye, forming an in situ flavonoid production and delivery system, inhibiting the accumulation of oxidative stress and preventing lens hardening. This project explores a permanent, cost-effective, nonsurgical treatment for presbyopia by genetically engineering Bifidobacterium, a probiotic ubiquitous in the human gastrointestinal tract, using the start-stop assembly method to construct a plasmid designed to generate quercetin. We studied the best growth conditions needed for bacterial plasmid propagation of quercetin and compared the efficiency of DNA transformation between Bifidobacterium and Staphylococcus epidermidis, a gram-positive aerobic probiotic that could be an alternative option for flavonoid production. Here, we report on the first step of construction and propose a workflow to help other researchers avoid possible obstacles in the genetic engineering process.
Keywords: Presbyopia, genetic engineering, flavonoids, Bifidobacteria, quercetin
Authors are listed in alphabetical order. Beth Pethel from Western Reserve Academy, Hudson Ohio, United States mentored the group. Please direct all correspondence to .
Human eyes can perceive and process 30 to 35 frames of image per second due to more than 125 million light-sensing cells in each retina. These light-sensing cells make up about 70% of all sensory receptors in the human body (Brookshire & Saey, 2020; Larson, 2020). More than half of the sensory receptors of the neocortex solely process visual information. This allows the brain higher accuracy and efficiency in processing visual information than other sensory information (Hutmacher, 2019). As a result, humans rely heavily on their vision for perception which means that conditions can significantly affect one’s quality of life, independence, and mobility.
Compared to congenital vision loss, the deterioration of existing eyesight requires constant increasing medical expenses and time from caregivers. The consequential change challenges patients’ physical, social, and mental states. Presbyopia, a prevalent eye condition that typically becomes noticeable around the age of 40 and continues to worsen for around 25 years, makes daily activities challenging. Patients with presbyopia struggle to focus on nearby objects. About a quarter of the world’s population is affected by presbyopia (Fricke et al., 2018). Presbyopia (Figure 1) is a result of lens hardening due to the accumulation of oxidative stress (Eye Doctors of Washington, n.d.). Both the cornea and the lens contribute to the focusing ability of the eyes. Unlike the cornea, the lens is surrounded by a circular muscle that relaxes and constricts to focus on objects. The circular muscle consists of tightly packed fiber cells filled with highly concentrated crystalline proteins that ensure lens transparency, a state that must be maintained to avoid visual impairment (Wride, 2011). Alpha-crystallin is an insoluble chaperone protein responsible for shaping proteins. Without it, proteins will gradually build up within the lens as we age, making it harder for the lens to reshape and focus on nearby objects (Pescosolido et al., 2016).
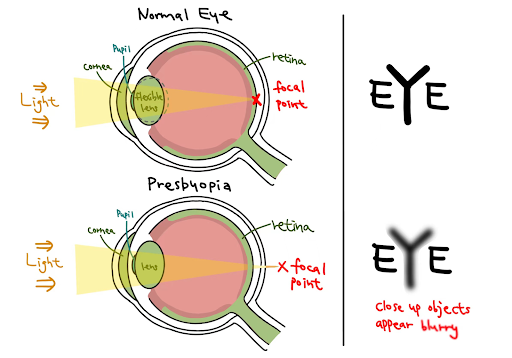
| Figure 1. Normal eye vs. presbyopia. Eyes with presbyopia have inflexible lenses that render the focal point falls behind the retina. |
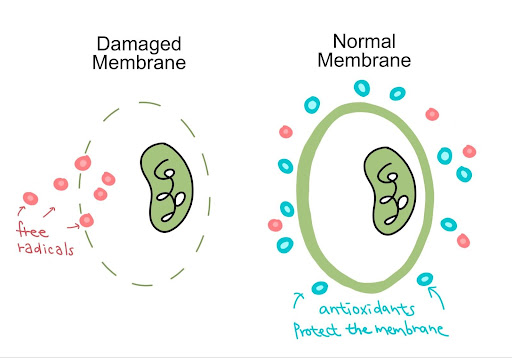
Another major cause of lens hardening is oxidative stress. Unstable oxygen molecules, also known as reactive oxygen species (ROS), are generated through digestion, metabolism, and as a by-product of environmental stressors like radiation and pollution. Healthy bodies can balance ROS with antioxidants. However, when humans reach the age of 40, antioxidants in the body typically decrease (Black, 2020). Thus, age-related eye diseases like cataracts and presbyopia happen more frequently among older adults. Without the interference of antioxidants, ROS accumulates and induces oxidative stress in the eyes. Oxidative stress causes proteins in the lens to cross-link and aggregate, rendering them heavier and less elastic, and therefore unable to adjust to focus on nearby objects (Figure 2).
Common treatments of presbyopia include laser treatments, surgeries such as conductive keratoplasty and corneal inlays, and wearing eyeglasses or contact lenses (Mayo Clinic, 2021; Mukamal, 2022). Nonetheless, studies examining the effectiveness of these treatments stress that restoring natural accommodation or finding an equivalent solution remains difficult to achieve and that symptoms tend to come back within a short period (Wolffsohn & Davies, 2018). In addition, laser surgery is costly at $2,000 or more per eye. Our team provides a more permanent solution to this vision problem: genetically engineering gut bacteria common in human bodies to produce quercetin, a dietary flavonoid abundant in nature (David et al., 2016).
Flavonoids are polyphenolic secondary metabolites that act as antioxidants in fruits and vegetables (Tungmunnithum et al., 2018). Quercetin is a well-studied flavonoid in the field of medicine, known for its antioxidative properties and its ability to efficiently scavenge ROS. Quercetin inactivates ROS by providing active hydrogen (Phaniendra et al., 2015).
Quercetin is “effective even after the [retinal] cells [are] exposed to oxidative stress, but before cell death [occurs]” (Majumdar & Srirangam, 2010). Quercetin is not only capable of preventing the impacts of aging but also of eliminating the existing level of oxidation. By engineering Bifidobacteria to produce quercetin, we hope to prevent presbyopia. In the pharmaceutical industry, quercetin is well-known for its positive effects as an antioxidant and the ability to control ocular blood pressure and maintain lens elasticity. However, it is not yet widely employed in medicine. Once the Bifidobacterium project is scaled up, we expect it to be a relatively inexpensive, more permanent, nonsurgical treatment for presbyopia.
Bifidobacterium, a bacterium ubiquitous in mammalian gastrointestinal tract microbiota, is used as a chassis. This probiotic is recognized for its ability to survive antimicrobial molecules and colonize the gut (Ruiz et al., 2012). Bifidobacterium undergoes several steps in the biosynthetic pathway to simulate the natural production of flavonoids in plants (Figure 3). There are many steps in biosynthetic pathways involved in the production of complex products. (Libretexts, n.d.). The production of quercetin is one such complex biosynthetic pathway. First, TAL and 4CL convert L-tyrosine into p-coumaroyl-coenzyme A (CoA). The product is subsequently catalyzed by CHS, CHI, F3H, FLS, and CPR into producing quercetin (Stahlhut et al., 2015).
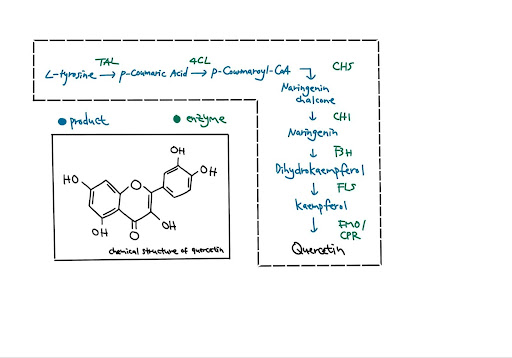
| Figure 3. Biosynthetic pathway and chemical structure of quercetin |
We will transform the eight genes needed for the production of quercetin into a plasmid using the start-stop assembly method for scarless combinatorial assembly, which can have up to 15 coding sequences in one vector. As Bifidobacterium replicates in human guts, it will consistently produce quercetin. Quercetin will be absorbed into the bloodstream via passive transport. Quercetin is harmless when consumed orally: safe for most healthy adults in doses of 1 g every day over 12 weeks. However, as Bifidobacterium colonizes and continues replication with the quercetin-producing function in the gut area, the possibility of overdose increases.
To reduce the risk of kidney damage as a result of overdosing, the majority of plasmids that we selected contain a low-copy number replicon, which produces low DNA yields (Wood et al., 2016). Yet, due to health concerns, the mechanism still needs further research and tests. A possible solution is utilizing a chemical-responsive switch and a chemical- and temperature-responsive switch to terminate unexpected massive replication (Rottinghaus et al., 2022). With our lab resources, temperature control would be the most feasible mechanism. In addition to the genes that will be inserted into the vector, we will also implant an aTc gene, which will start to produce toxins to induce cell death when temperature drops below 22℃.
We obtained our chassis, Bifidobacterium longum 105-A, from RIKEN-JCM(Japan). Due to its high transformation efficiency, we predict that it is the best host for rapid culturing bacteria. Yet, to support this hypothesis, we also grew S. epidermidis to perform contrast experiments in genetic engineering. To boost the Bifidobacteria growth, we plan to test different living conditions, including oxygen level, type of media, and growth period. With maximizing production, we will utilize transformation to insert genes from the biosynthetic pathway of quercetin. This new method to massively produce bioflavonoids will aid us in the production of quercetin.
Materials and methods
Production of antibiotics solution
DNA transformation requires isolated colonies. To ensure the efficiency of colony isolation, we need to prevent the plasmids that we chose, pKO403_lacZ_Cm and pRSFDuet-v2 RsTAL At4CL, from being contaminated. As a result, antibiotics were made to combat the possible contamination and could be used in both media and the stab culture of plasmids (Figure 4).
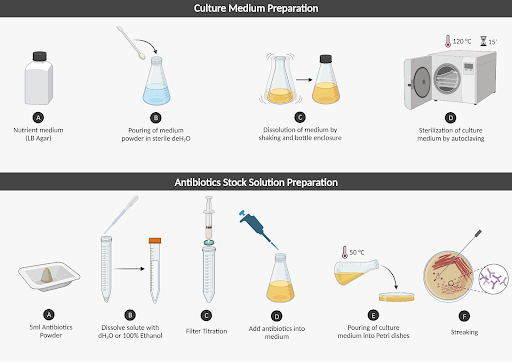
| Figure 4. Complete procedure with detailed steps. |
For the first step of DNA transformation of our project according to the biosynthetic pathway of quercetin, we purchased two plasmids: an empty backbone of Bifidobacterium and the plasmid that contains the first two genes needed, TAL and 4CL (Addgene). They each require different antibiotics: chloramphenicol and kanamycin (Goldbio).
We dissolved 0.5 g chloramphenicol using 10 mL 100% ethanol, and 0.5 g kanamycin using 5 mL distilled water. The stock solution of chloramphenicol is 50000 μg/ml, and 100000 μg/mL for kanamycin. We used a medical syringe and syringe filter to complete the filter titration of solutions (Figure 5). To set up the titration, we fixed the syringe and its filter (BD) using a luer lock to prevent leaking during the process. A new conical tube was set under the syringe’s tip. We took out the plunger and used a pipette to transfer the solution into the barrel of the syringe, before pressing the plunger to provide pressure to the system until the filtered solution all entered the cube down below. The purpose of filter titration is to prevent disparate concentration and clamps when using antibiotics.

| Figure 5. Design of titration system. |
The antibiotic solution needs to be diluted to certain concentration before being pipetted into the bacteria stab culture and BSM media (Carolina). The volume was calculated according to the following formula:
C1V1= C2V2
C1 represents the concentration of our stock solution, while C2 denotes the target concentration for bacteria resistance, and V2 denotes the final volume of diluted solution, which is 2.1 mL for both antibiotics.
C1 = 100000 μg/mL
V1 = unknown = 10.05 μL kanamycin
C2 = 50 μg/mL
V2 = 2.1 mL
(2.1 mL * 50 μg/mL) / 100000 μg/mL = 10.05 μL kanamycin
C1 = 50000 μg/mL
V1 = unknown = 10.05 μL chloramphenicol
C2 = 25 μg/mL
V2 = 2.1 mL
(2.1 mL * 25 μg/mL) / 50000 μg/mL = 10.05 μL chloramphenicol
As a result, 10.05 μL of kanamycin and chloramphenicol is added into the Bifidus selective media (BSM) and stab culture of plasmids from Merck to prevent contamination.
Bifidobacterium growth control
To test the growth of Bifidobacterium under different oxygen concentrations and temperatures, we conducted control experiments.
We made 200 mL media with antibiotics and 200 mL media without antibiotics. Then we streaked 4 plates of Bifidobacterium and separated them into two categories: anaerobic and aerobic. Two aerobic plates were placed at room temperature for two days, allowing bacteria to grow. Chloramphenicol was added only into the media of one plate. The two anaerobic plates were kept in a gas-pak system (Figure 6), one with antibiotics and one without. Plates were set next to each other in a gas-pak pouch (BD). Then, we soaked a piece of paper towel with distilled water and put it under the CO2 generator (BD) in the pouch. The pouch was quickly sealed and placed on the table under room temperature for two days.

| Figure 6. Gas-pak system. |
|---|
Another trial tests the impacts of different temperatures on Bifidobacterium growth. We streaked four plates of the plasmid that contains the Bifidobacterium backbone; two plates were grown under room temperature for two days, while other two plates were grown in the 37℃ incubator for the same amount of time.
Laboratory safety and precaution
Bifidobacterium is a type of probiotic that normally exists and settles in the intestines. According to guidelines in Biosafety in Microbiological and Biomedical Laboratories, Bifidobacterium is classified as level-one biosafety contamination. The microbe poses a minor-to-no threat of infection in healthy adults. S. epidermidis is also classified as level-one biosafety bacteria that is part of normal human flora. The evolution of the bacteria allows it to become non-pathogenic and maintains a benign relationship with its host (Otto M., 2009). However, people with weakened immune systems, including those with HIV/AIDS, cancer, or on medications in preparation for organ transplants, are more susceptible to blood infections caused by either Bifidobacterium and S. epidermidis. To prevent potential infections, we suggest following lab protocol and wearing personal protective equipment, including gloves, lab coats, and goggles. All the materials that were exposed to the bacteria, including inoculating loops and plates, were autoclaved and disposed of properly.
Antibiotics that are used to control contamination also need to be handled with caution. The antibiotics used for the growth of S. epidermidis, chloramphenicol is categorized as hazardous. Chloramphenicol can cause skin, eye, and respiratory irritation. 100% ethanol is the suggested solvent for chloramphenicol. Although ethanol does not generate toxins, the chemical is flammable and needs to be kept away from heat, sparks, and flames to prevent fire (Fisher Scientific, 2003). When working with ethanol, the lab door will be opened to ensure unobstructed airflow. Kanamycin is the other antibiotic that we used for the growth the of the bacteria with plasmids TAL and 4CL. Kanamycin may cause allergic reactions when in contact with skin or inhaled by researchers (Thermo Fisher Scientific, 2021). As a result, all antibiotics were stored in a locked 4℃ fridge in closed-lid bottles. Masks are not required but recommended in order to avoid harm to the respiratory system.
Results
Antibiotics results
Chloramphenicol functions well with the Bifidobacterium. We originally expected only desired bacteria colonies growing even without antibiotics due to our experience of growing S. epidermidis and E. coli. However, the first trial that ran without antibiotics was unsuccessful. All the plates were contaminated with a different kind of bacterium. As we observed, successful colony isolation and growth of our target plasmid were small transparent-to-white dots distributed in a pattern. The unknown bacteria, on the other hand, grew scattered and appeared as larger white dots. Either adding antibiotics into the media or directly into the stab culture prevented the plates from being contaminated. However, kanamycin did not help much with the 4CL and TAL genes. Although the unwanted bacteria population did stop growing on the plates, the target plasmid also vanished. Both plates ultimately had no bacterial colonies.
Oxygen level results
We compared the growth of bacteria under different oxygen levels by counting individual colonies. After two days, plates that were in the pouch had many fewer colonies than the ones that were not. Therefore, we concluded that Bifidobacterium will have a faster growth under aerobic conditions.
Temperature results
We compared the growth of bacteria under different temperatures by counting individual colonies. After two days, plasmids that were growing in the incubator seem to have a close amount of bacteria to those that were left outside. There might be a minor difference, yet from observation, distinction is not obvious. As a result, we concluded that temperature does not have a significant effect on growth.
Discussion
The growth of the bacteria with the plasmid that contains the Bifidobacterium backbone was unexpected and was against our predictions. According to our theoretical research, the best growth condition for Bifidobacterium should be 37℃ and an oxygen level under 5% (Kawasaki et al., 2006). We reviewed our experimental process and realized that there are a few factors that could cause this opposite result.
First, we cannot accurately control the oxygen level to under 5%. Since a vacuum pump is not utilized, the sealed bag still contains a certain amount of oxygen. According to research, Bifidobacterium will stop growing at an oxygen level below 10%, but once the oxygen level reaches 5% and lower, it instead stimulates bacterial growth. We chose Bifidobacterium as our chassis for its ability to survive antimicrobial molecules and colonize the gut as a bacterium ubiquitous in mammalian gastrointestinal tract microbiota (Ruiz et al., 2012). However, we do not have an anaerobic chamber or anaerobic jar that can accurately reduce the level of oxygen for this type of anaerobic bacteria to grow. Therefore, we are unable to obtain the specific level of oxygen.
We did take this fact into consideration before we started our experiment, which is the reason why we added two packs of CO2 generators into the pouch. Yet, it is possible that we lowered the oxygen level to under 10% yet above 5%, thus resulting in slow growth of the bacteria. To improve our experimental design, we should conduct the same experiment with more than two packs of CO2 generators. We also lack a method to test the effectiveness of those CO2 generators based on our results, thus there is a need to purchase an oxygen level detector for us to monitor the oxygen level change. Another possible explanation of the situation could be the specialty of the plasmid. The plasmid does contain a Bifidobacterium backbone, but the vector also contains parts from E. coli, which is an aerobic bacterium.
Second, although the temperature did not seem to play an important role in the production of Bifidobacterium, we still suggest that 37°C would be preferable. Both Bifidobacterium and E. coli are documented to have their best growth under 37 °C living conditions. If we identify E. coli as a factor in our result from the oxygen level experiment, the same analysis should apply to the temperature control experiment. To create a more obvious comparison in the future, we should pour the culture medium that contains antibiotics, leave the streaked plates in the 4 °C fridge and the 37 °C incubator, and compare the growth after two days.
Lastly, regarding the practicality of antibiotics, we believe that the problem is related to the concentration of kanamycin. In our lab, the particles of solid kanamycin are larger than those of chloramphenicol. Hence, when we tried to mix the solution, the antibiotic probably did not dissolve completely, meaning that the concentration in our calculation may be incorrect. A higher concentration of antibiotics will lead to bacterial death. A solution to the situation is to add less kanamycin into our media and observe the growth. We can continuously test and adjust the concentration of antibiotics based on the growth of Bifidobacterium.
Next steps
We still need to evaluate the feasibility of genetically engineering Bifidobacterium. S. epidermis. requires less restrictive living conditions than Bifidobacterium. Without antibiotics, it still grows nicely with multiple individual colonies. However, its ability to perform DNA transformation still needs to be tested. Electroporation is an effective method to make membranes of E. coli more permeable (Liu et al., 2018). Whether it is more effective to render the membrane of Bifidobacteria or to the membrane of S. epidermis adequately permeable is yet to be assessed (Figure 7).
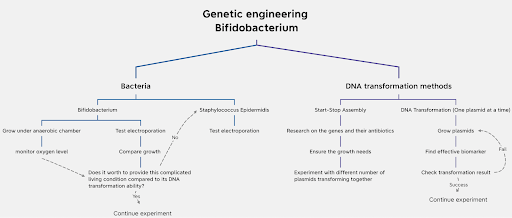
| Figure 7. Workflow plan of the project |
The 100 trillion gut microbiota settling in the human body form a complicated, codependent microbial system (Thursby & Juge, 2017). Although sufficient studies demonstrated that gut nutrients do manage to come into effect in the eyes, we are not sure about the specific transportation route from the intestine to the eye.
Yet, we can infer from research that quercetin would travel through the vagus nerve and the bloodstream to reach the brain. Whether there would be enough concentration of quercetin that ends up in the eyes to decelerate the progression of presbyopia is yet to be determined. We hope to trace Bifidobacterium’s activities using a gene tag once it enters the body – once gut microbiota enters the intestine, they reproduce and colonize for the remainder of their lives. pMGC-mCherry, a type of fluorescent protein (FP) that combines a chloramphenicol resistance marker and expression of the FP mCherry under the control of Pgap, is chosen for its ability to determine gastrointestinal transit time of our bacteria (Grimm et al., 2014).
The relationship between the concentration of Bifidobacterium and its effect on the gut system should be investigated in order to determine the appropriate concentration of freeze-dried bacteria. We would design experiments to observe bacterial reproduction and the level of quercetin production in the gut. Another pathway will be potentially necessary if the absorption doesn’t reach the ideal level of concentration. One solution would be utilizing carrier-mediated membrane transporters to actively transfer quercetin into circulation. Transporters like PEPT1 could significantly raise intestine absorption and oral bioavailability of nutrients (Katsura & Inui, 2003). Here we created a workflow plan to explain the possible next steps of the project (Figure 7).
Author contributions
B.L generated the original idea. B.L., D.L., C.M., and P.W. contributed to the preliminary research on the feasibility of the project, wrote and proofread the abstract and introduction of the design article collaboratively. D.L. continued with the idea and edited the abstract and introduction. D.sL. completed the experiments and wrote the rest parts of the report. B.L and D.L created images in this paper, and D.L. produced the video.
Acknowledgements
The Flaveyenoid synthetic biology team would like to thank our teacher, Dr. Beth Pethel, for her encouragement and guidance throughout this project. She has advised us in many aspects from experimental skills to design improvements. Without her, we wouldn’t be able to push our projects to go further. We would like to acknowledge Western Reserve Academy for providing the resources and opportunity, especially the Science department for experimental materials and space to practice our experiment; as well as biorender.com for the assistance in creating figures, and Animaker.com for the assistance in producing our video.
References
Brookshire, B., & Saey, T. H. (2020, July 16). Explainer: How our eyes make sense of light. Science News Explores. https://www.sciencenewsforstudents.org/article/explainer-how-our-eyes-make-sense-of-light
Black, T. (2020, July 14). Oxidative stress and the eye part 1: What is oxidative stress? ACVO Public. https://www.acvo.org/tips-treatments-tricks/oxidative-stress-and-the-eye-part-1-what-is-oxidative-stress
David, A. V. A., Arulmoli, R., & Parasuraman, S. (2016). Overviews of biological importance of quercetin: A bioactive flavonoid. Pharmacognosy Reviews, 10(20), 84-89. https://doi.org/10.4103%2F0973-7847.194044
Eye Doctors of Washington. (n.d.). What is the difference between Presbyopia and hyperopia?. https://www.edow.com/farsighted/what-is-the-difference-between-presbyopia-and-hyperopia/
Fisher Scientific. (2003, March 18). Material safety data sheet. https://fscimage.fishersci.com/msds/89308.htm
Fricke, T. R., Tahhan, N., Resnikoff, S., Papas, E., Burnett, A., Ho, S. M., Naduvilath, T., & Naidoo, K. S. (2018). Global prevalence of presbyopia and vision impairment from uncorrected presbyopia: Systematic review, meta-analysis, and modelling. Ophthalmology, 125(10), 1492-1499. https://doi.org/10.1016/j.ophtha.2018.04.013
Grimm, V., Gleinser, M., Neu, C., Zhurina, D., & Riedel, C. U. (2014). Expression of fluorescent proteins in Bifidobacteria for analysis of host-microbe interactions. Applied and Environmental Microbiology, 80(9), 2842–2850. https://doi.org/10.1128/AEM.04261-13
Hutmacher F. (2019). Why is there so much more research on vision than on any other sensory modality?. Frontiers in Psychology, 10, 2246. https://doi.org/10.3389/fpsyg.2019.02246
Katsura, T., & Inui, K. I. (2003). Intestinal absorption of drugs mediated by drug transporters: Mechanisms and regulation. Drug Metabolism and Pharmacokinetics, 18(1), 1-15. https://doi.org/10.2133/dmpk.18.1
Kawasaki, S., Mimura, T., Satoh, T., Takeda, K., & Niimura, Y. (2006). Response of the microaerophilic Bifidobacterium species, B. boum and B. thermophilum, to oxygen. Applied and Environmental Microbiology, 72(10), 6854–6858. https://doi.org/10.1128/AEM.01216-06
Larson, J. (2020, October 20). Human eye fps: How much can we see and process visually? Healthline. https://www.healthline.com/health/human-eye-fps#how-vision-works
Libretexts. (n.d.). 5.12b: Biosynthesis and Energy. https://bio.libretexts.org/Bookshelves/Microbiology/Book%3A_Microbiology_(Boundless)/5%3A_Microbial_Metabolism/5.12%3A_Biosynthesis/5.12B%3A_Biosynthesis_and_Energy
Liu, J., Chang, W., Pan, L., Liu, X., Su, L., Zhang, W., Li, Q., & Zheng, Y. (2018). An improved method of preparing high efficiency transformation Escherichia coli with both plasmids and larger DNA fragments. Indian Journal of Microbiology, 58, 448-456. https://doi.org/10.1007/s12088-018-0743-z
Majumdar, S., & Srirangam, R. (2010). Potential of the bioflavonoids in the prevention/treatment of ocular disorders. The Journal of Pharmacy and Pharmacology, 62(8), 951–965. https://doi.org/10.1211/jpp.62.08.000
Mayo Clinic. (2021, November 20). Presbyopia. https://www.mayoclinic.org/diseases-conditions/presbyopia/symptoms-causes/syc-20363328
Mukamal, R. (2022). Corneal inlays: A surgical alternative to reading glasses. American Academy of Ophthalmology. https://www.aao.org/eye-health/treatments/corneal-inlays-alternative-to-reading-glasse
Otto, M. (2009). Staphylococcus epidermidis—The’accidental’pathogen. Nature Reviews Microbiology, 7(8), 555-567. https://doi.org/10.1038/nrmicro2182
Pescosolido, N., Barbato, A., Giannotti, R., Komaiha, C., & Lenarduzzi, F. (2016). Age-related changes in the kinetics of human lenses: Prevention of the cataract. International Journal of Ophthalmology, 9(10), 1506-1517. https://doi.org/10.18240%2Fijo.2016.10.23
Phaniendra, A., Jestadi, D. B., & Periyasamy, L. (2015). Free radicals: properties, sources, targets, and their implication in various diseases. Indian Journal of Clinical Biochemistry, 30, 11-26. https://doi.org/10.1007/s12291-014-0446-0
Rottinghaus, A. G., Ferreiro, A., Fishbein, S. R., Dantas, G., & Moon, T. S. (2022). Genetically stable CRISPR-Based kill switches for engineered microbes. Nature Communications, 13(1), 672. https://doi.org/10.1038/s41467-022-28163-5
Ruiz, L., Álvarez-Martín, P., Mayo, B., de los Reyes-Gavilán, C. G., Gueimonde, M., & Margolles, A. (2012). Controlled gene expression in Bifidobacteria by use of a bile-responsive element. Applied and Environmental Microbiology, 78(2), 581-585., https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3255758/
Stahlhut, S. G., Siedler, S., Malla, S., Harrison, S. J., Maury, J., Neves, A. R., & Forster, J. (2015). Assembly of a novel biosynthetic pathway for production of the plant flavonoid fisetin in Escherichia coli. Metabolic Engineering, 31, 84-93, https://www.sciencedirect.com/science/article/pii/S1096717615000828
Thermo Fisher Scientific. (2021, December 28). Safety Data Sheet. https://www.fishersci.com/store/msds?partNumber=AC611290050&productDescription=KANAMYCIN+SULFATE+5GR&vendorId=VN00033901&countryCode=US&language=en
Tungmunnithum, D., Thongboonyou, A., Pholboon, A., & Yangsabai, A. (2018). Flavonoids and other phenolic compounds from medicinal plants for pharmaceutical and medical aspects: An overview. Medicines, 5(3), 93. https://doi.org/10.3390/medicines5030093
Wolffsohn, J. S., & Davies, L.N. (2019). Presbyopia: Effectiveness of correction strategies, Progress in Retinal and Eye Research, 68, 124-143, https://doi.org/10.1016/j.preteyeres.2018.09.004
Wood, W. N., Smith, K. D., Ream, J. A., & Lewis, L. K. (2016). Enhancing yields of low and single copy number plasmid dnas from Escherichia coli cells. Journal of Microbiological Methods. https://www.sciencedirect.com/science/article/abs/pii/S0167701216303591?via%3Dihub
Wride, M. A. (2011). Lens fibre cell differentiation and organelle loss: many paths lead to clarity. Philosophical Transactions of the Royal Society B: Biological Sciences, 366(1568), 1219-1233. https://doi.org/10.1098/rstb.2010.0324