Brennan Bhattacharjee, Aarit Chauhan▫ BioBuilderClub, Andover High School, Andover, Massachusetts, United States
Alisa Kolstova ▫ Oak Ridge High School, El Dorado Hills, California, United States
Adrian Luck, Alexander Luck ▫ BioBuilderClub, Bozeman High School, Bozeman, Montana, United States
Reviewed on 6 May 2023; Accepted on 13 July 2023; Published on 16 October 2023
With help from the 2023 BioTreks Production Team.
The Limulus amebocyte lysate (LAL) assay is one of the most important bio-safety tests used in the pharmaceutical industry to detect endotoxins, which are toxic chemicals excreted by bacteria, and to ensure product safety and purity. Horseshoe crabs’ blue blood is the source of LAL. Horseshoe crabs are routinely captured and bled. Although after the bleeding process many horseshoe crabs are released back into their natural habitat, a significant percentage die. Several research groups successfully developed a synthetic alternative to LAL with the recombinant Factor C (rFC) as the active ingredient. Unfortunately, the synthetic alternative is not being utilized in the industry and the bleeding of horseshoe crabs continues. We plan to improve the already developed synthetic assay by co-expressing rFC along with another intracellular serine protease zymogens, recombinant Factor B (rFB), to increase accuracy and specificity. We plan to deliver the new alternative to the biomedical industry and to the general public in the form of a spray for detection of endotoxins. Our efforts will serve to highlight that horseshoe crabs no longer need to suffer as valid synthetic alternatives are readily available. We also hope that our research will educate the public and pressure policy makers to convince biomedical companies and regulatory agencies to stop exploiting horseshoe crabs and incorporate synthetic alternatives into routine practice.
Keywords: Amebocyte lysates, Limulus test, Limulus amebocyte lysate, recombinant Factor C, bacterial endotoxins, lipopolysaccharides, horseshoe crab (Tachypleus tridentatus), innate immunity, clotting factors, recombinant technologies
Authors are listed in alphabetical order. Barbara Tevelev from BioBuilderClub, Bozeman High School, Bozeman, Montana, United States mentored the group. Please direct all correspondence to .
Horseshoe crabs (Tachypleus tridentatus) are arthropods of the family Limulidae and the only living members of the order Xiphosura (Walls et al, 2002). Horseshoe crabs have been around for over 200 million years, making them even older than the dinosaurs (Kin and Blazejowski, 2014; Walls et al, 2002). Their body is divided into three sections, the prosoma or head, abdomen or opisthosoma, and the tail which is called the telson (Figure 1). The head of the horseshoe crab resembles a large letter U and contains most of their body, including their heart, brain, eyes, nervous system and glands. The opisthosoma is the triangular body of the crab and holds muscles, spines and gills. The telson or tail of the crab is not dangerous or poisonous. It is used to help the crabs flip over when they are turned upside down (National Wildlife Federation et al, 2022).
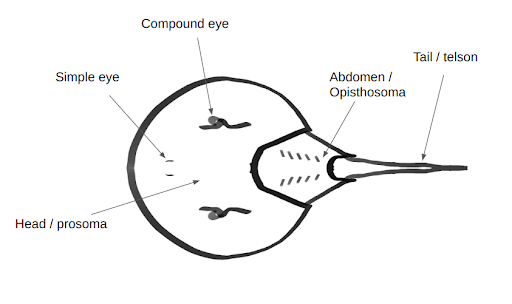
| Figure 1. Horseshoe crab anatomy. Dorsal view showing head/prosoma, simple eyes, compound eyes, abdomen/opisthosoma, and tail/telson. |
|---|
Horseshoe crabs have an open circulatory system with a long and tubular heart that runs down the middle of their head and abdomen (Medzhitov and Janeway, 2000; Walls et al., 2002). Horseshoe crabs have no veins and their blood flows freely through their body cavity being in direct contact with different tissues. Unlike most other animals, horseshoe crabs blood does not contain hemoglobin which is a protein that transports oxygen in many animals. Instead, it contains hemocyanin, a chemical that gives horseshoe crabs’ blood a blue color and distributes oxygen (Medzhitov & Janeway, 2000; Walls et al., 2002). Their open circulatory system puts horseshoe crabs at risk as pathogens like bacteria and viruses come in direct contact with blood and can spread quickly. To adapt, horseshoe crabs’ blood evolved to contain amebocytes which are special immune cells that produce LAL (Medzhitov and Janeway, 2000; Walls et al., 2002; Krisfalusi-Gannon, 2018). The LAL system allows the horseshoe crabs to quickly and effectively combat infections and protect from bacterial endotoxins (John et al., 2010; Krisfalusi-Gannon, 2018). LAL is a coagulation system for detecting and removing pathogens and harmful toxins (Figure 2). The system is induced and controlled by three main endotoxin-sensitive, intracellular serine protease zymogens: Factor C, Factor B, and Factor R. Factor C is activated by the endotoxin (John et al., 2010; Krisfalusi-Gannon, 2018). The activated Factor C then activates Factor B which in turn activates the proclotting enzyme. The proclotting enzyme triggers clotting enzymes that coagulate around endotoxins and bacteria forming gelatines. The proclotting enzyme can also be initiated by Factor G when in the presence of β-D-glucan. The Factor G stimulates the proclotting enzyme and continues the reaction.
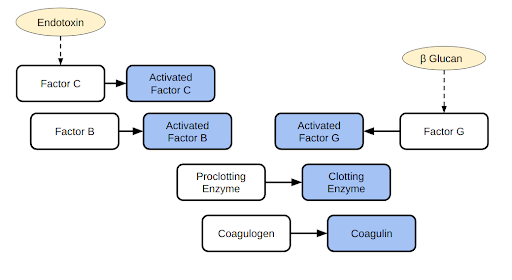
| Figure 2. Schematic representation of the LAL reaction. Shown are the endotoxin pathway leading to coagulogen conversion and an alternative beta glucan activated pathway. |
|---|
It is the LAL’s endotoxin binding and clotting ability that makes horseshoe crabs’ blood so valuable to the pharmaceutical and medical industries (John et al., 2010; Krisfalusi-Gannon, 2018; Walls et al., 2002). Many of the pharmaceutical products are made using bacterial systems and bacterial endotoxins which may be potential contaminants. Also, medical devices that are used during surgeries, for administration of medicines, and during a variety of medical procedures may be potentially contaminated by bacteria and bacterial endotoxins. Endotoxins are fever inducing substances that are harmful to humans and so all materials produced in living cells must be tested for such contaminants (Bolden et al., 2017; Bolden, 2019). Bacteria and bacterial endotoxins when left in drug products and on medical devices will hurt humans, cause hemorrhaging, inflammation, and other ill effects. Respectively, purity tests are important. The LAL from horseshoe crabs’ blood is a critical component for testing purity of pharmaceutical products such as vaccines, proteins, antibodies and other biotherapeutics as well as medical devices such as needles, surgical instruments, and medical aids (Bolden et al., 2017; Bolden, 2019; Krisfalusi-Gannon, 2018).
Horseshoe crabs are harvested in large numbers and bled to obtain LAL (Smith et al., 2020). They are lined up on tables and straws are pierced through their arthrodial membrane; the tubes leak their blood into bottles (Figure 3). Such vampirism along with other negative environmental factors causes horseshoe crab numbers to dwindle. Over 500,000 specimens are captured from the wild for bleeding each year and many are killed during the process (Smith et al., 2020). It is estimated that the mortality rate of this procedure is 15%, which kills up to around 75,000 crabs every year (Atlantic States Marine Fisheries Commission, 2019). Another alarming observation is the staggering drop in the number of female horseshoe crabs returning to the beaches where specimens were harvested (Leschen et al., 2010). While most researchers focused on calculating survival rates of bleeding, some studies demonstrated that post-bleeding causes reduced behavioral activity and physiological changes that potentially altered immune function and survival and mating activity (Anderson et al., 2013; Kurz & James-Pirri, 2002; Owings et al., 2019).

| Figure 3. An Example of Horseshoe Crab Exploitation (Jaquci, et al. 2020) |
The decrease in horseshoe crabs’ population also impacts the aquatic ecosystem, endangering other species (National Wildlife Federation et al., 2022). Horseshoe crabs can be found in the North American Atlantic coastline, the Indian Ocean and along the coast of Asia in the Pacific Ocean. Every year in the late spring and early summer, horseshoe crabs gather from the deep in order to breed. Their eggs are a vital source of nutrients for various species of birds, reptiles, and fish. They play a critical role in the ecosystem and ensure that many different species get the nutrients they need (National Wildlife Federation et al., 2022). With the death of many horseshoe crabs this fragile ecosystem is suffering.
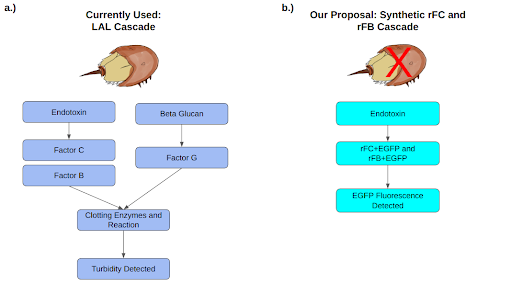
However, there still may be hope for these critters. Scientists have designed and developed a synthetic alternative to LAL: rFC (Maloney et al., 2018; Bolden and Smith, 2017; Bolden, 2019). Similar to LAL, rFC detects endotoxins released by bacteria. Although the synthetic version existed on the market for over 15 years, and it was officially approved for usage by the regulatory agency in 2020, its usage in the pharmaceutical industry is lacking (Bolden, 2019; Dubczak et al., 2021; Gorman, 2020; Maloney et al., 2018; Piehler, 2020). Single source supplier concerns and lack of inclusion in pharmaceutical companies may be the culprit (Gorman, 2020). Because companies and suppliers have been transacting for a long time, it is difficult for them to break the chain, restructure workflows, make organizational changes, and potentially lose profit by changing procedures that are already in place.
Despite some initial setbacks, great benefits await the companies that decide to transition away from LAL. Certain studies have shown that rFC and other coagulation factors in LAL, rFB, are as good if not better than LAL (Kobayashi et al, 2015; Mizumura, 2017; Piehler, 2020). In fact, the horseshoe crab’s LAL system contains Factor G. Factor G is disadvantageous because it does not activate coagulation in presence of endotoxins. Instead, it activates in the presence of β-D-glucan, which initiates the clotting cascade regardless of endotoxic presence and thus will produce a false positive result (Iwanaga, 2007). It would be extremely beneficial to companies to use synthetic alternatives produced without Factor G, such as rFC assays and our proposed spray, as these products have a potential to enhance the overall performance of the procedure by increasing specificity and accuracy of the assay. The costs of production may also be lower for synthetic alternatives, as they do not require the collection and handling of horseshoe crabs and can be produced on a bigger scale. Some pharmaceutical companies, such as Pfizer, are already leading efforts to switch to synthetic alternatives and dedicating many resources to convince other businesses to join the paradigm shift. This shift must take place globally. Pharmaceutical companies across the world and respective regulatory agencies must align in supporting the change to the synthetic alternatives of LAL assay. Decrease in cost and increase in assay performance as well as environmental responsibility should be sufficient drivers for the private sector to make this possible.
In order to protect horseshoe crabs and the fragile aquatic ecosystem that they are part of, we want to further optimize and expand the usage and application of rFC and rFB. We hope to create a spray which contains not only rFC but also rFB in a stable solution at room temperature and pressure. The two coagulation factors will increase sensitivity of the purity assay as well as broaden its application. Our solution can be used to test for presence or absence of endotoxins and any residual bacterial components. The solution can be used to test clearance of medical devices, lot testing of drug substances and products and potentially lot testing of foods. The reagents in the spray will work in conjunction with UV light. The rFC and rFB will bind to endotoxins and upon the binding reaction, a fluorophore will be activated and detected in the UV range by exposing the sprayed surface to UV light. In contrast to the original LAL assay, which was based on visual inspection of a coagulation reaction in a glass tube and relatively newer detection via absorbants using a spectrophotometer or an absorbance microplate reader. Our method is simpler and requires less instrumentation.
Systems level
To alleviate reliance on horseshoe crabs’ blood, we plan to create a synthetic substitute for LAL in the form of rFC and rFB. The currently available synthetic alternative relies only on rFC. By including rFB in addition torFC, we hope to increase the assay’s specificity and accuracy by eliminating false negatives (as may be the case with synthetic rFC) and false positives (as may be the case with the harvested LAL from horseshoe crabs) (Figure 4). Both Factor C and Factor B are serine-protease zymogens essential for the horseshoe crab coagulation cascade. Factor B is the primary substrate for activation of Factor C (Kobayashi et al, 2015; Mizumura, 2017). And so, addition of rFB may help to improve rFC sensitivity.
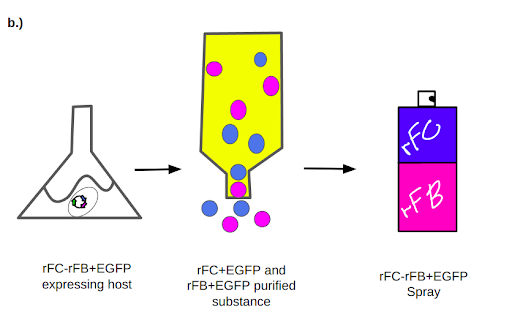
We plan to create a spray of rFC and rFB that can be readily available for use in the pharmaceutical, medical and food industry to quickly test for endotoxins (Figure 5). The same spray can be also used in home settings to ensure cleanliness and test contamination levels on surfaces and foods.
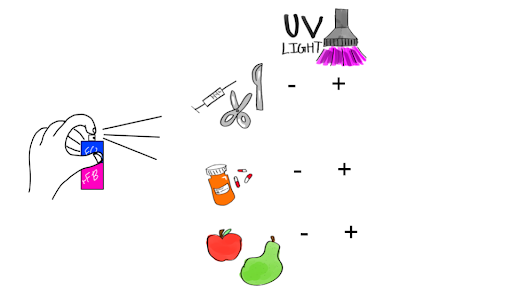
| Figure 5. Proposed rFC-rFB+EGFP spray solution. The spray may be used to test for endotoxins across different items: medical devices, medicines, foods, and home items. |
|---|
Device level
Using recombinant DNA technology, we propose to design a mammalian chinese hamster ovary (CHO) plasmid expression system to produce rFC and rFB (Figure 6). A yeast system could be used for initial proof of principle, while the main production should be completed via the CHO system because of its high output and consistent productivity. We plan to run tests to understand the safety and effectiveness of our product. If the tests prove successful, we hope that our spray of rFC and rFB provides an efficient, high-yielding, affordable and safe solution for the pharmaceutical industry and the public while saving the horseshoe crab population and helping to stabilize the ecosystem.
We plan to provide the rFC and rFB in a stable spray solution that can bind to harmful endotoxins and bacteria. Subsequently, a fluorophore attached to the rFC and rFB will trigger a fluorescent output which will be detectable by UV light. Upon binding of rFC and rFB to endotoxins, the EGFP will be activated and detected when exposed to ultraviolet light (Figure 6).
Below, we designed an expression vector for transfection into CHO cells. By using a mammalian host system, we are preventing potential contamination that may occur when using a bacterial host system such as Escherichia coli. Using E. coli may or may not result in false positives, and so eliminating any known risk is optimal. Furthermore, mammalian cells are known to be more productive and thus resulting in increased titer of the proteins we seek to produce. The expression vector contains genes for rFC and rFB fused to EGFP and a neomycin resistance gene for selection. The CMV promoter and SV40 late polyadenylation signal are transcriptional elements that are known to increase gene expression and may result in increased protein production as well as stability (Radhakrishnan, 2008).
We selected electroporation as a preferred method of choice for transfection. Electroporation uses an electrical pulse to create temporary pores in the cell membrane through which the rFC and rFB containing vector will pass and integrate within the host genome. After transfection, we will allow the cells to recover for 48 hrs in media containing cellastim, which is a recombinant human serum albumin that enhances recovery and performance of cell growth. On the 3rd day, we plan to add a selection marker, neomycin, to eliminate untransfected cells. The neomycin is toxic to the cells that do not contain the neomycin resistance gene. Only cells with successful integration of the entire expression vector will be able to survive the selection procedure as they will obtain a neomycin resistance gene. The transfected pools of cells will be cultured and passaged into media+neomycin until viable cell density drops below 50%, after which point the selection marker will be removed from the media. This is done by spinning down the cells, removing the media with neomycin, and resuspending the cells in fresh media (without neomycin). Cells will be allowed to recover until viable cell density reaches above 90%.
Post-selection recovery, the surviving pools of cells will be cultured in nutrient rich media to enhance growth and productivity. The cells will be assessed from day 3 and day 4 passaging cultures to determine the titer levels and quality of rFC and rFB proteins via high-performance liquid chromatography (HPLC). Once we obtain positive results and proper product quality data from passaging cultures, we will focus on developing a fed-batch production process as well as harvest and purification process (out of scope for the initial experiments) (Kobayashi et al., 2015; Liu et al., 2010).
Parts level
Our approach involves designing a single expression plasmid capable of stably integrating within the CHO mammalian host. The expression plasmid is designed for co-expression of both rFC and rFB fused to EGFP (Figure 7). Upon successful integration within the host genome, the expression plasmid carries a neomycin resistance gene which will confer resistance to the host during selection. Blastcidin resistance gene was also added in case there may be a need for additional more stringent selection. Cells which integrate the entire expression plasmid will survive selection and will be screened for production of rFC+EGFP and rFB+EGFP. The EGFP is a commonly used green fluorescent protein, which will be utilized to confirm if the plasmid is integrated within the host genome and allow detection with UV light in the final spray product. For all of the genes of interest, each open reading frame (ORF) is preceded with a CMV promoter and a Kozak sequence. CMV is a strong mammalian promoter and Kozak facilitates translation initiation of ATG start codon downstream of the Kozak sequence. Downstream of each ORF, we included SV40 late poly A tail which allows transcription termination and polyadenylation of mRNA transcribed by Pol II RNA polymerase.
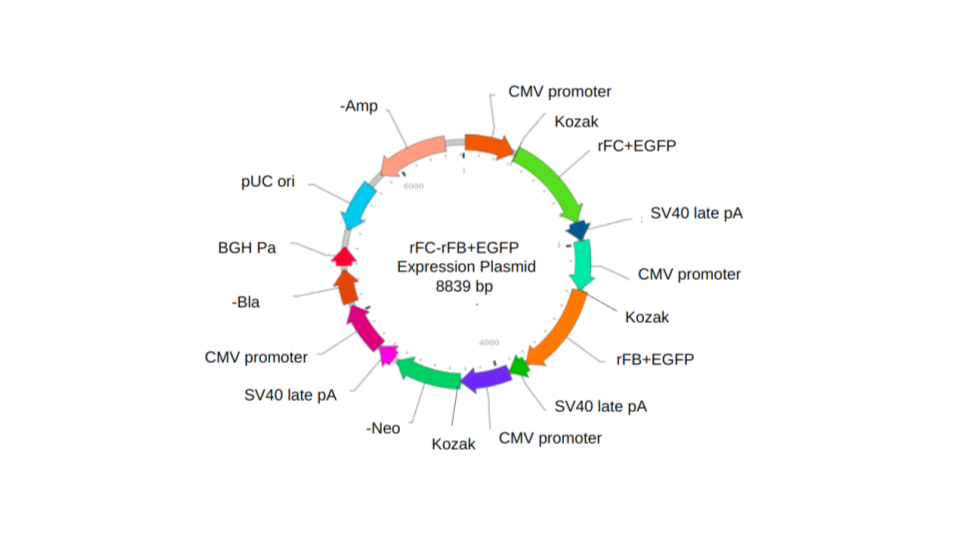
| Figure 7. Diagram of the rFC-rFB+EGFP expression vector for the CHO mammalian host system. |
|---|
Other elements included are the pUC ori and -Amp, which serve to amplify and generate plasmid substrate for transfection. The pUC ori facilitates plasmid replication in E. coli and regulates high-copy plasmid number (500- 700). The -Amp confers ampicillin resistance and allows E. coli to be resistant to ampiciIIin during plasmid selection and amplification step.
All the vector elements are described in detail in Table 1.
e202313t01
| Name | Size (bp) | Type | Description | Application Notes |
|---|---|---|---|---|
| CMV | 589 | Promoter | Human cytomegalovirus immediate early enhancer/promoter | Strong promoter; may have variable strength in some cell types. |
| Kozak | 6 | Miscellaneou | Kozak translation initiation sequence | Facilitates translation initiation of ATG start codon downstream of the Kozak sequence. |
| SV40 late pA | 222 | PolyA_signal | Simian virus 40 late polyadenylation signal | Allows transcription termination and polyadenylation of mRNA transcribed by Pol II RNA polymerase. |
| Neo | 795 | ORF | Neomycin resistance gene | Allows cells to be resistant to geneticin (G418). |
| Bla | 396 | ORF | Blasticidin resistance gene | Allows cells to be resistant to blasticidin. |
| BGH pA | 225 | PolyA_signal | Bovine growth hormone polyadenylation signal | Allows transcription termination and polyadenylation of mRNA transcribed by Pol II RNA polymerase. |
| pUC ori | 589 | Rep_origin | pUC origin of replication | Facilitates plasmid replication in E. coli; regulates high-copy plasmid number (500- 700). |
| AmpiciIIin | 861 | ORF | AmpiciIIin resistance gene | Allows E. coli to be resistant to ampiciIIin. |
| rFC fused to EGFP | ORF | Recombinant Factor C; intracellular serine protease zymogens | Gene of Interest | |
| rFB fused to EGFP | ORF | Recombinant Factor B; intracellular serine protease zymogens | Gene of Interest | |
| EGFP | 720 | ORF | Enhanced green fluorescent protein; codon optimized based on a variant of wild type GFP from the jellyfish Aequorea victoria | Commonly used green fluorescent protein; ranked high in brightness, photostability and pH stability among all fluorescent proteins. |
Safety
We plan to adhere to good laboratory practices and to wear personal protective equipment such as gloves, goggles and lab coats.
The CHO cells, CHO expression systems and all of the components associated with CHO cell culture and recombinant protein production are safe to use. All of these reagents are common in the mammalian research laboratories and are considered non-hazardous. CHO cells and their respective reagents have been used in laboratory settings for over 100 years.
Discussions
Our design strives to transition the global pharmaceutical industry and supporting regulatory agencies away from using the limulus amebocyte lysate (LAL) assay, which is produced through the exploitation and bleeding of horseshoe crabs, to an animal-friendly synthetically derived spray. This is the central objective of our plan, but other distinct advantages and disadvantages should be taken into account when discussing the potential success of our bacterial spray.
An important part of our approach is designing a single expression plasmid that would integrate both rFC and rFB within the CHO mammalian host simultaneously. This increases efficiency and reduces the potential
costs of having to complete separate processes for rFC and rFB transfer, which may require a more intense selection process and extra optimization. Additionally, selecting a cell line with two successfully integrated vectors through clone screening will require genetic characterization and more analytics. By combining the expressions of both proteins into a single vector, we increase the chances of both genes integrating within the host genome, while
decreasing the complexity of the selection process. We also reduce the number of resistance genes needed for selection, as only one set would be needed per pair of rFC and rFB expression genes, lowering theoretical costs of production.
Another significant benefit of our plan is the improved accuracy and specificity of the synthetic substitute. Our spray will include rFB, which may help detect endotoxins that the current rFC assay might miss, eliminating false negatives. The spray may also exceed the capability of the LAL assay, for its system contains FG which can be falsely triggered to form clotting cascades by the β-D-glucan, leading to false positives (Iwanaga, 2007). By combining rFC with rFB, while eliminating FG, we optimize the accuracy of the assay.
However, multiple disadvantages and setbacks are present in our design as well. The production and use of synthetic rFC has already been demonstrated by many research groups, but the utilization of rFB is not common and may require qualification and validation proof (Piehler, 2020). Multiple years of concurrent sample testing by LAL and rFC+rFB assays might be needed to prove the synthetic alternative comparable.
The biggest challenge lies in popularizing synthetic rFC and rFB for use in the biomedical and pharmaceutical industry. Bleeding horseshoe crabs may be more cost-effective as the LAL assays are already established as a staple of biosafety testing and are commonly accepted by regulatory agencies. Even though many research groups have improved the synthetic rFC assays and lowered production costs, the main obstacle is not the cost, but the acceptance of synthetic alternatives by the world’s regulatory and governing organizations. For now, only the FDA and the European Pharmacopoeia recognize the use of rFC as a valid alternative to LAL (Council of Europe, 2020). Unfortunately, the US Pharmacopoeia (USP) is hesitant in declaring the rFC testing methods equivalent to LAL assays, thus discouraging US drugmakers from changing their practices (Jimenez, 2021). Furthermore, South American, Chinese, and Middle Eastern regulatory institutions only approve LAL results. Thus, a company with synthetic test results and product clearance would be at a disadvantage as multitudes of countries, including the US, may not validate such testing. Therefore, to maintain testing consistency and retain access to the global market, pharmaceutical companies are forced to use the LAL assay, continuing the exploitation of the horseshoe crab. To make a difference, pharmaceutical companies and the public must advocate for change across the world.
Next steps
To continue pursuing our goal of eliminating global horseshoe crab exploitation in return for our synthetically derived spray, we plan to refocus our efforts outside the lab and connect with a range of professionals who may offer feedback and support.
We hope to interview scientists and subject matter experts from pharmaceutical organizations like Pfizer, Eli Lilly, Hyglos GmbH, and various universities, who may point out nuances and setbacks we should expect when trying to integrate our system into accepted practices (Maloney et al., 2018). Explaining our methods and current plan, we would look forward to getting feedback on how to appeal to big organizations. We will consult policy makers and the current chapters in pharmacopeias to learn what tests are needed to qualify our spray as an alternative method. We hope to contact the leading manufacturers of LAL, such as Lonza, Charles River Labs, and Associates of Cape Cod, who may have influence over USP policies, to better understand the appeal of LAL and what effective means could be used to spread a similar popularity of our synthetic alternative (Jimenez, 2021). We hope to talk to the leading rFC manufacturers as well, like Novartis AG and Hyglos GmbH, to learn more about improving consistency and reducing handling time for our spray (Onkar, 2023). We would try to contact government officials, like the US Senate Committee on Environment and Public Works to ask to modify regulatory policies to make rFC + rFB assays more accepted and widespread.
We also hope to write to conservation companies and their representatives like Ryan Phelan, the co-founder and executive director of Revive & Restore (a California-based conservation non-profit), and Deborah Cramer, a representative of the Massachusetts Horseshoe Crab Recovery Initiative, who have spoken out for the incorporation of rFC testing into standard practices, and ask them to consider advertising our spray as well (Jimenez, 2021; Welcome to Massachusetts Horseshoe Crab Recovery Initiative, 2023). We plan to introduce our idea to a range of environmental activists, who may not have been aware of horseshoe crab exploitation, and would support our humanitarian concern for the animals. Thus, we could gain the support needed to challenge the accepted practices and bring change for the better.
Author contributions
A.J.L and A.E.L initiated the topic, developed the idea, designed the vector strategy and the expression system as well as designed all the proposed experiments. A.J.L and A.E.L were the primary authors, drafting and finalizing the manuscripts and executing all the figures. A.K. designed and created the summary powerpoint and video as well as wrote the Discussion and Next Steps section. B.B and A.C were instrumental to the review process and initial project’s discussions. All authors contributed to literature search and review.
Acknowledgements
We would like to acknowledge and thank our mentor, Barbara Televev as well as our teacher, Lindsey L’Ecuyer. We appreciate their guidance, support and help. We also want to thank the AHS BioBuilderClub for providing us with the opportunity to pursue our interests in synthetic biology and bioengineering.
References
Anderson, R. L., Watson III, W. H., & Chabot, C. C. (2013). Sublethal behavioral and physiological effects of the biomedical bleeding process on the American horseshoe crab, Limulus polyphemus. The Biological Bulletin, 225(3), 137-151.
Atlantic States Marine Fisheries Commission. [ASMFC]. (2019). Horseshoe crab stock assessment report.
Bolden, J., & Smith, K. (2017). Application of Recombinant Factor C reagent for the detection of bacterial endotoxins in pharmaceutical products. PDA Journal of Pharmaceutical Science and Technology, 71(5), 405–412. https://doi.org/10.5731/pdajpst.2017.007849
Bolden, J. (2019). Recombinant Factor C. In: Williams, K. (Ed.), Endotoxin detection and control in pharma, limulus, and mammalian systems (pp. 497-521). Springer. https://doi.org/10.1007/978-3-030-17148-3_13
Council of Europe. (2020, July 1). Recombinant factor C: New ph. eur. chapter available as of 1 July 2020. European directorate for the quality of medicines & healthcare – EDQM. https://www.edqm.eu/en/-/recombinant-factor-c-new-ph.-eur.-chapter-available-as-of-1-july-2020
Dubczak, J., Reid, N., & Tsuchiya, M. (2021). Evaluation of limulus amebocyte lysate and recombinant endotoxin alternative assays for an assessment of endotoxin detection specificity. European Journal of Pharmaceutical Sciences, 159, 105716. https://doi.org/10.1016/j.ejps.2021.105716
Gorman, R. (2020). Atlantic horseshoe crabs and endotoxin testing: perspectives on alternatives, sustainable methods, and the 3Rs (replacement, reduction, and refinement). Frontiers in Marine Science, 7, 582132. https://doi.org/10.3389/fmars.2020.582132
Isakova, V., & Armstrong, P. B. (2003). Imprisonment in a death-row cell: The fates of microbes entrapped in the Limulus blood clot. The Biological Bulletin, 205(2), 203-204. https://doi.org/10.2307/1543253
Iwanaga, S., Kawabata, S. I., & Muta, T. (1998). New types of clotting factors and defense molecules found in horseshoe crab hemolymph: Their structures and functions. The Journal of Biochemistry, 123(1), 1-15. https://doi.org/10.1093/oxfordjournals.jbchem.a021894
Jimenez, D. (2021, October 22). Pharma’s reliance on horseshoe crabs is threatening the species. Pharmaceutical Technology. https://www.pharmaceutical-technology.com/features/pharma-horseshoe-crabs-threatening-species/
John, B. A., Jalal, K. C. A., Kamaruzzam, Y. B., & Zaleha, K. (2010). Mechanism in the clot formation of horseshoe crab blood during bacterial endotoxin invasion. Journal of Applied Sciences, 10(17), 1930–1936. https://doi.org/10.3923/jas.2010.1930.1936
Kin, A., & Błażejowski, B. (2014). The horseshoe crab of the genus Limulus: Living fossil or stabilomorph?. PLoS One, 9(10), e108036. https://doi.org/10.1371/journal.pone.0108036
Kobayashi, Y., Takahashi, T., Shibata, T., Ikeda, S., Koshiba, T., Mizumura, H., Oda, T., & Kawabata, S. I. (2015). Factor B is the second lipopolysaccharide-binding protease zymogen in the horseshoe crab coagulation cascade. Journal of Biological Chemistry, 290(31), 19379-19386. https://doi.org/10.1074/jbc.M115.653196
Krisfalusi-Gannon, J., Ali, W., Dellinger, K., Robertson, L., Brady, T. E., Goddard, M. K., Tinker-Kulberg, R., Kepley, C. L., & Dellinger, A. L. (2018). The role of horseshoe crabs in the biomedical industry and recent trends impacting species sustainability. Frontiers in Marine Science, 5. https://doi.org/10.3389/fmars.2018.00185
Kurz, W., & James-Pirri, M. J. (2002). The impact of biomedical bleeding on horseshoe crab, Limulus polyphemus, movement patterns on Cape Cod, Massachusetts. Marine and Freshwater Behaviour and Physiology, 35(4), 261-268. https://doi.org/10.1080/1023624021000019315
Leschen, A. S., & Correia, S. J. (2010). Mortality in female horseshoe crabs (Limulus polyphemus) from biomedical bleeding and handling: Implications for fisheries management. Marine and Freshwater Behaviour and Physiology, 43(2), 135-147. https://doi.org/10.1080/10236241003786873
Liu, H. F., Ma, J., Winter, C., & Bayer, R. (2010). Recovery and purification process development for monoclonal antibody production. mAbs, 2(5), 480-499. https://doi.org/10.4161/mabs.2.5.12645
Loveland, R. E., Botton, M. L., & Shuster Jr, C. N. (1996). Life history of the American horseshoe crab (Limulus polyphemus L.) in Delaware Bay and its importance as a commercial resource. In Proceedings of the horseshoe crab forum: Status of the resource. University of Delaware Sea Grant College Program, Lewes, Delaware (pp. 15-22).
Maloney, T., Phelan, R., & Simmons, N. (2018). Saving the horseshoe crab: A synthetic alternative to horseshoe crab blood for endotoxin detection. PLoS Biology, 16(10), e2006607. https://doi.org/10.1371/journal.pbio.2006607
Medzhitov, R., & Janeway J., C. (2000). Innate immune recognition: Mechanisms and pathways. Immunological Reviews, 173, 89-97. https://doi.org/10.1034/j.1600-065x.2000.917309.x
Mikkelsen, T. (1988). The secret in the blue blood. Science Press.
Mizumura, H., Ogura, N., Aketagawa, J., Aizawa, M., Kobayashi, Y., Kawabata, S. I., & Oda, T. (2017). Genetic engineering approach to develop next-generation reagents for endotoxin quantification. Innate Immunity, 23(2), 136-146. https://doi.org/10.1177/1753425916681074
Onkar, S. (2023, April). Recombinant Factor C Assay Market. Allied Market Research. https://www.alliedmarketresearch.com/recombinant-factor-c-assay-market-A13438
Owings, M., Chabot, C., & Watson III, W. (2019). Effects of the biomedical bleeding process on the behavior of the American horseshoe crab, Limulus polyphemus, in its natural habitat. The Biological Bulletin, 236(3), 207-223. https://doi.org/10.1086/702917
Piehler, M., Roeder, R., Blessing, S., & Reich, J. (2020). Comparison of LAL and rFC Assays—Participation in a Proficiency Test Program between 2014 and 2019. Microorganisms, 8(3), 418. https://doi.org/10.3390/microorganisms8030418
Radhakrishnan, P., Basma, H., Klinkebiel, D., Christman, J., & Cheng, P. W. (2008). Cell type-specific activation of the cytomegalovirus promoter by dimethylsulfoxide and 5-Aza-2′-deoxycytidine. The International Journal of Biochemistry & Cell Biology, 40(9), 1944-1955. https://doi.org/10.1016/j.biocel.2008.02.014
Smith, D. R., Newhard, J. J., McGowan, C. P., & Butler, C. A. (2020). The long-term effect of bleeding for Limulus Amebocyte Lysate on annual survival and recapture of tagged horseshoe crabs. Frontiers in Marine Science, 7. https://doi.org/10.3389/fmars.2020.607668
Walls, E. A., Berkson, J., & Smith, S. A. (2002). The horseshoe crab, Limulus polyphemus: 200 million years of existence, 100 years of study. Reviews in Fisheries Science, 10(1), 39-73. https://doi.org/10.1080/20026491051677