Anusheh Hashmi, Shambhavi Jayakumar, Angela Mac, and Emily Packer ▫ BioBuilderClub, Andover High School, Andover, Massachusetts, United States
Reviewed on 6 May 2023; Accepted on 13 July 2023; Published on 16 October 2023
With help from the 2023 BioTreks Production Team.
Sickle cell disease (SCD) is a genetic blood disorder characterized by the presence of sickle-shaped red blood cells, which can lead to complications such as pain, organ damage, and stroke. Sickle cell retinopathy (SCR), a complication that can result in vision loss or possibly blindness, is one of the less well-known effects of SCD. In SCR, sickled red blood cells obstruct the retina’s blood vessels, reducing oxygen delivery and causing tissue damage. For SCD and SCR, many of the current treatments unfortunately only focus on managing the symptoms, rather than addressing the underlying pathophysiology of the condition. However, our innovative strategy increases blood flow to the retina by placing a biological stent in. In medicine, a stent is a device which is inserted into a blood vessel or other internal duct to expand it to prevent or alleviate a blockage. In the context of the retina, a biological stent is a treatment method that improves blood flow through the retinal blood vessels, thereby reducing the risk of complications associated with ischemia, or insufficient blood flow. Our gene circuit, which encodes for the synthesis of NO, will do the same. We chose NO because it acts as a potent vasodilator of blood vessels by relaxing the smooth muscles in the walls of blood vessels. The circuit would be injected into the retina, then diffused into surrounding blood vessels. It will then produce NO to dilate the blood vessels when it detects the presence of sickled red blood cells and the occurrence of hypoxia around the eye. The circuit will contain a sickle cell sensor, an oxygen sensor, and an NO synthase coding sequence. Together, they will work as a circuit in order to increase blood flow to the eye and reverse the effects of SCR. Overall, this gene circuit has the potential to be a powerful and targeted therapy for the prevention and treatment of SCR in sickle cell patients. It leverages the body’s own response mechanisms to address the underlying cause of the disease, rather than simply managing its symptoms, which can drastically improve the quality of life for SCR patients.
Keywords: Sickle cell disease, sickle cell retinopathy, genetic circuit, retina, nitric oxide
Authors are listed in alphabetical order. Lindsey L’Ecuyer from BioBuilderClub, Andover High School, Andover, Massachusetts, United States mentored the group. Please direct all correspondence to .
SCD is a part of a group of blood disorders that affects hemoglobin, which is the molecule in red blood cells that delivers oxygen to cells throughout the body (Sickle Cell Disease, 2020). SCD is an autosomal recessive disorder and is caused by a mutation of the HBB gene – which is the gene that provides instructions for making the β-globin subunit of hemoglobin. This particular HBB mutation, which a person must inherit two copies of, produces an abnormal version of β-globin called hemoglobin S (HbS). In order for a person to have sickle cell disease and not just be a carrier, they must have at least one of the β-globin subunits in hemoglobin replaced with HbS (Sickle Cell Disease, 2020). This mutation causes red blood cells to sickle or become crescent-shaped.
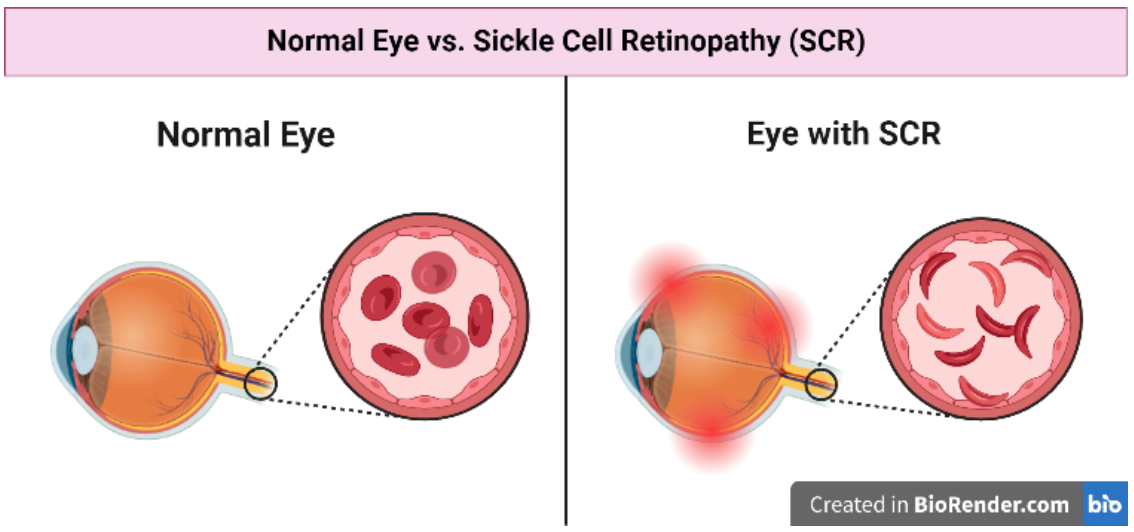
Due to the abnormal shape, a patient’s red blood vessels will often get blocked, which can cause a low number of blood cells, repeated infections, and periodic episodes of pain (CDC, 2022). SCR is a complication of SCD that is caused by vascular occlusion, or the blockage of blood vessels in the retina and choroid (Figure 1). The retina includes the light-sensitive layers of nerve tissue at the back of the eye. Its function is to capture incoming photons and transmit them along neuronal pathways (Yu et al., 2017). The choroid is the thin layer of tissue that is part of the middle layer of the wall of the eye, between the white outer layer of the eye (sclera) and the retina. The choroid is filled with blood vessels that bring oxygen and nutrients to the eye (Yu et al., 2017). Therefore, vascular occlusion, or the blockage of blood vessels can result in the prevention of oxygen to the eye, and can ultimately lead to blindness (Bakri et al., n.d.).
The current only cure to SCD, and its complications, is a stem cell or bone marrow transplant (NHS, 2022). In these procedures, stem cells from a healthy donor are taken through a tube from a vein, the blood then goes through a machine that takes out the stem cells in a process called apheresis. The stem cells are given to the SCD patient, and the blood with no stem cells is returned to the donor (Mayo Clinic, 2022). However, a stem cell transplant is an intensive treatment that has many risks, the main being graft versus host disease (GVHD). This is a life-threatening complication where the transplanted cells start to attack other cells in the body. This is because the immune cells in the transplant view the recipient’s body as foreign and attack it (Cancer Research UK, 2022).
Normal T cells do not attack someone’s own body cells because they recognize human leukocyte antigens (HLA), which are inherited from parents, and are unique to each person except in identical twins. After the transplant, the stem cells start making new body cells, which will have the donor’s HLA pattern. However, the T cells will view these HLA patterns as unfamiliar and will attack the recipient’s body. Therefore, the more different the SCD patient’s HLA is to the donor’s HLA, the more likely the chance is that the patient gets GVHD (Cancer Research UK, 2022).
SCR is a complication that affects the blood vessels in the retina of individuals with SCD, affecting up to 40% of patients by the age of 40 (Sambhara & Hsu, 2022). There are two types of SCR: non-proliferative and proliferative. Non-proliferative SCR is characterized by the presence of small, dot-like intraretinal hemorrhages, which are patches on the retina from loss of blood or damaged blood vessels. In addition, patients of non-proliferative SCR will have cotton wool spots, or abnormal white patches on the retina, and venous beading. Proliferative SCR, on the other hand, is characterized by the growth of new blood vessels in the retina that are fragile and prone to bleeding (Sambhara & Hsu, 2022). Both types of SCR have different stages, ranging from mild to severe. In non-proliferative SCR, the disease can progress from stage 1, which is characterized by the presence of isolated cotton wool spots and intraretinal hemorrhages, to stage 4, which is marked by retinal ischemia, which occurs when a sufficient amount of blood is no longer able to get to the retina and cell death becomes a risk. In proliferative SCR, the disease can progress from stage 1, which is characterized by the presence of peripheral neovascularization, or the presence of new and abnormal blood vessels, to stage 3, which is marked by the presence of vitreous hemorrhage, or blood in the vitreous, and eventually retinal detachment. Our proposed treatment for SCR targets the early stages of non-proliferative SCR, specifically stages 1 and 2. In these stages, there are cotton wool spots, intraretinal hemorrhages, and venous beading. These signs indicate retinal ischemia and can lead to further complications if left untreated.
Currently, there are no specific treatments for non-proliferative SCR, one can only manage symptoms and prevent the disease from progressing to the severe stage through regular eye exams and monitoring for signs of progression (Scott, 2022). There are only treatments for proliferative SCR, such as anti-VEGF injections, which inhibit the growth of new blood vessels in the retina, preventing further damage or laser therapy, which can shrink abnormal blood vessels and seal off those that are leaking (Scott, 2022). However, though these treatments are effective in treating proliferative SCR, they can have adverse effects such as infection, inflammation, and scarring. In addition, the systemic approach that is used to manage non-proliferative SCR primarily focuses on preventing the development of the complication rather than treating it.
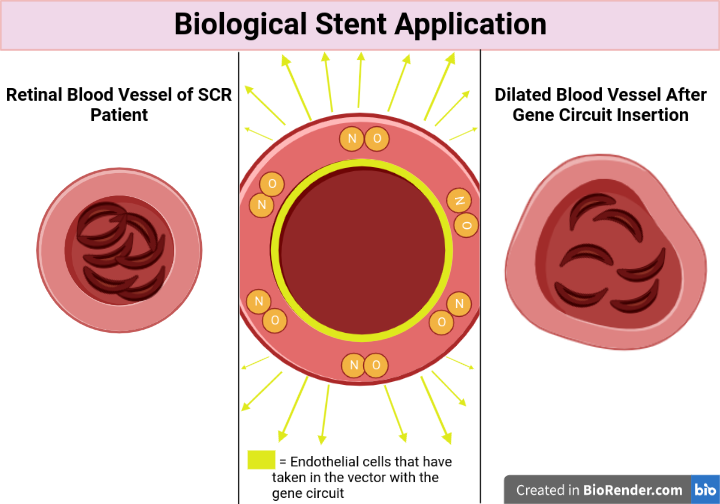
Our design is a genetic circuit that acts as a biological stent and can detect the presence of sickled red blood cells and hypoxia around the retina, then subsequently produce nitric oxide (NO) to dilate the vessels and allow sickled cells to pass through to deliver oxygen to the eye. This targeted treatment has several advantages over stem cell transplants. Because our gene circuit targets only the blood vessels around the retina, it should have very limited chance for off-target effects (Kryzszton et al., 2021), compared to the many risks that come with the current available treatments for SCD, which are not as accessible, as donors have many requirements. Our therapy, on the other hand, involves an injection that may only need to be done one time. In the case of our design, an injection may only need to be done more than once because we cannot guarantee that gene circuit will remain effective forever, especially depending on the patient. However, overall, we still believe that our targeted gene circuit has the potential to provide a safer and more effective treatment option for SCD patients with complications such as SCR and we will make sure that the treatment is effective enough to limit the number of injections the patient would need.
Systems level
Our proposed solution is a gene circuit that senses the presence of sickle cells in the bloodstream and induces the production of NO, which promotes vasodilation, or the dilation of blood vessels, and reduces sickle cell adhesion (Figure 2). A gene circuit is a set of genetic components such as promoters, coding sequences, and terminators that interact to produce a specific output in response to a particular input signal (Brophy & Voigt, 2014). This solution will allow sickled cells to flow to the eye and deliver all of the necessary nutrients and oxygen needed to sustain the retina and choroid. The circuit contains a sickle cell sensor promoter and HBB sensor promoter. These promoters drive the expression of the nitric oxide synthase (NOS) gene, which converts arginine into NO (Wu et al., 2021). The NOS gene is regulated by an AND gate, which means that an output of NOS would require two inputs. In our case, our AND gate requires both sickle cell and low oxygen level signals to activate NO production.
This gene circuit functions as a feedback loop (Brophy & Voigt, 2014) that regulates blood flow and reduces sickle cell adhesion. The sensing components in our gene circuit are proteins that can detect specific molecules or conditions in the environment. In the case of our gene circuit, the HBB sensor is designed to detect low oxygen levels, or hypoxia which occurs in the retinas of SCR patients. Our HBB sensor contains the HIF-1α protein, which has a naturally occurring component called the oxygen-dependent degradation (ODD) domain (Percy et al., 2003), which allows for its regulation in response to changes in oxygen levels. The ODD domain allows for the degradation of HIF-1α protein under normal oxygen conditions, preventing the protein from accumulating and activating hypoxia-response genes, however, under hypoxic conditions, the ODD domain is inhibited, allowing for the stabilization of HIF-1α (Percy et al., 2003). Similarly, the sickle cell sensor in our gene circuit is designed to detect the presence of sickle red blood cells in the retina. The sensor is a modified version of the HBB protein, which is mutated in SCD. The mutated HBB protein (HbS) has a different conformation than normal HBB, which is recognized by the sensor. The output component produces NO to dilate blood vessels and prevent sickled cells from getting blocked in blood vessels. In addition, the gene circuit operates in a self-sustaining manner, continuously sensing and responding to changes in the environment (Figure 2).
Once the gene circuit is injected, it begins to detect sickle cells and low oxygen levels using the sickle cell and HBB sensor promoters, respectively. When both signals are present, the NOS gene is activated, leading to the production of NO. The NO then diffuses out and into the surrounding tissues (Chen et al., 2008). NO acts as a potent vasodilator, or dilator of blood vessels. It relaxes the smooth muscles in the walls of blood vessels, resulting in their dilation (Chen et al., 2008). This is accomplished through the activation of the enzyme guanylyl cyclase, which increases the production of cyclic guanosine monophosphate (cGMP) within the smooth muscle cells of the vessel wall. cGMP then activates the protein kinase G (PKG), a protein heavily involved in muscle contraction (Marín-García, 2014). Its activation leads to the relaxation of smooth muscle cells, resulting in vasodilation (Ataabadi et al., 2020). In the case of the blood vessels and capillaries surrounding the retina, NO can help improve blood flow and oxygen delivery to the retina, which is crucial for its proper function. The retina has a high metabolic demand and requires a constant supply of oxygen and nutrients, which are delivered by the capillaries (Liu & Prokosch, 2021). If the capillaries are constricted, damaged, or blocked, it can lead to ischemia (lack of oxygen) and damage to the retina. By dilating the blood vessels around the retina, NO can help improve blood flow and oxygen delivery, which can help prevent ischemia and reverse the effects of SCR. The increased blood flow helps prevent the formation of other vaso-occlusive crises, which is the main cause of SCR (Sambhara & Hsu, 2022).
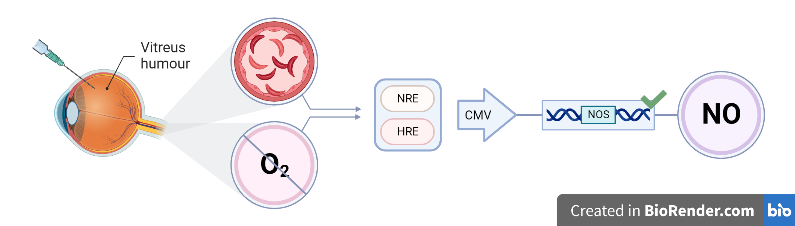
The delivery of our gene circuit is through an intravitreal injection (Figure 3), which is a less invasive and more accessible alternative with the current standard of care, stem cell transplants. Unlike stem cell transplants, our gene circuit does not require a donor match and does not have the risk of GVHD. Additionally, our gene circuit therapy can be given in a single injection that could last for months or years (Chopra et al., 2021), making it a more convenient treatment option for patients with SCR.
We can ensure that our treatment is as targeted as possible due to the method of delivery. The advantage of using an intravitreal injection is that we can ensure that the gene circuit and the NO have a very limited chance of affecting other cells in the body and causing problems (Chopra et al., 2021). Since we are injecting the therapy directly into the vitreous humor of the eye, we can limit the diffusion of the gene circuit to other parts of the body (Wang et al., 2019). In addition, the promoter that we are using on our gene circuit, the cytomegalovirus (CMV) promoter, has been shown to have limited expression in non-target tissues when delivered locally (Xia et al., 2006), as we plan to do with our gene therapy. In addition, adding our plasmid to a viral vector, as opposed to just injecting the plasmid by itself helps keep the treatment targeted. Viral vectors have the ability to specifically target certain cell types, due to the regulatory elements added in them (Xia et al., 2006). In the case of our proposed design, our vector is able to specifically target the endothelial cells around the retina because of the regulatory elements (NRE and HRE) and our gene enhancer (HIF-1α). Additionally, viral vectors are capable of efficiently delivering the gene circuit into cells, increasing the likelihood that it will be expressed in the desired cells in the retina (Wang et al., 2019). All these factors can further reduce the risk of the gene circuit of the gene circuit affecting other parts of the body. However, it will still be important to carefully design and test the viral vector to ensure that it is safe and effective for use in humans.
Device level
The root of SCR is the sickled cells blocking the many arteries that lead to the retina and choroid (Sambhara & Hsu, 2022). These include the posterior ciliary arteries (PCAs) and central retinal artery (CRA), and branches of the ophthalmic artery (OA) (Yu et al., 2017). We are targeting the therapy to the CRA and its branches, as the CRA is the most involved in the development of SCR (Menaa et al., 2017). This is a better approach than trying to fix the damaged retina for several reasons. First, it is difficult to access and repair damaged retinal tissue directly, especially with the delicate and complex structure of the eye (Holmes, 2018). Second, targeting the blood vessels allows for a more systemic approach to treating SCR, addressing the root cause of the disease rather than the symptoms.
In our gene circuit, a set of components will react to produce NO and dilate blood vessels in the presence of both the sickle cell sensor gene and the NO synthase gene. The circuit will subsequently utilize the genetic code and regulatory mechanisms of endothelial cells to control gene expression (Brophy & Voigt, 2014). The circuit consists of three genes: the HBB (oxygen) sensor gene, the sickle cell sensor gene, and the NOS gene. The sickle cell sensor gene is designed to be expressed in the retina of the eye and to detect the presence of sickled cells. When activated, it would trigger the oxygen sensor promoter, which would detect the low oxygen levels that often occur in the retinal blood vessels of SCR patients (Sambhara & Hsu, 2022). When these two conditions are detected, the expression of the NOS gene is activated, which produces NO in response to the presence of sickled red blood cells and hypoxia (Figure 3). NO is known to increase blood flow and reduce vaso-occlusion, (Chen et al., 2008) which is what majorly causes the damage in the retina due to SCR (Ventocilla & Lee, 2021). By increasing blood flow and reducing vaso-occlusion, the NO produced by the circuit could help reverse and prevent the damage to the retina caused by SCD.
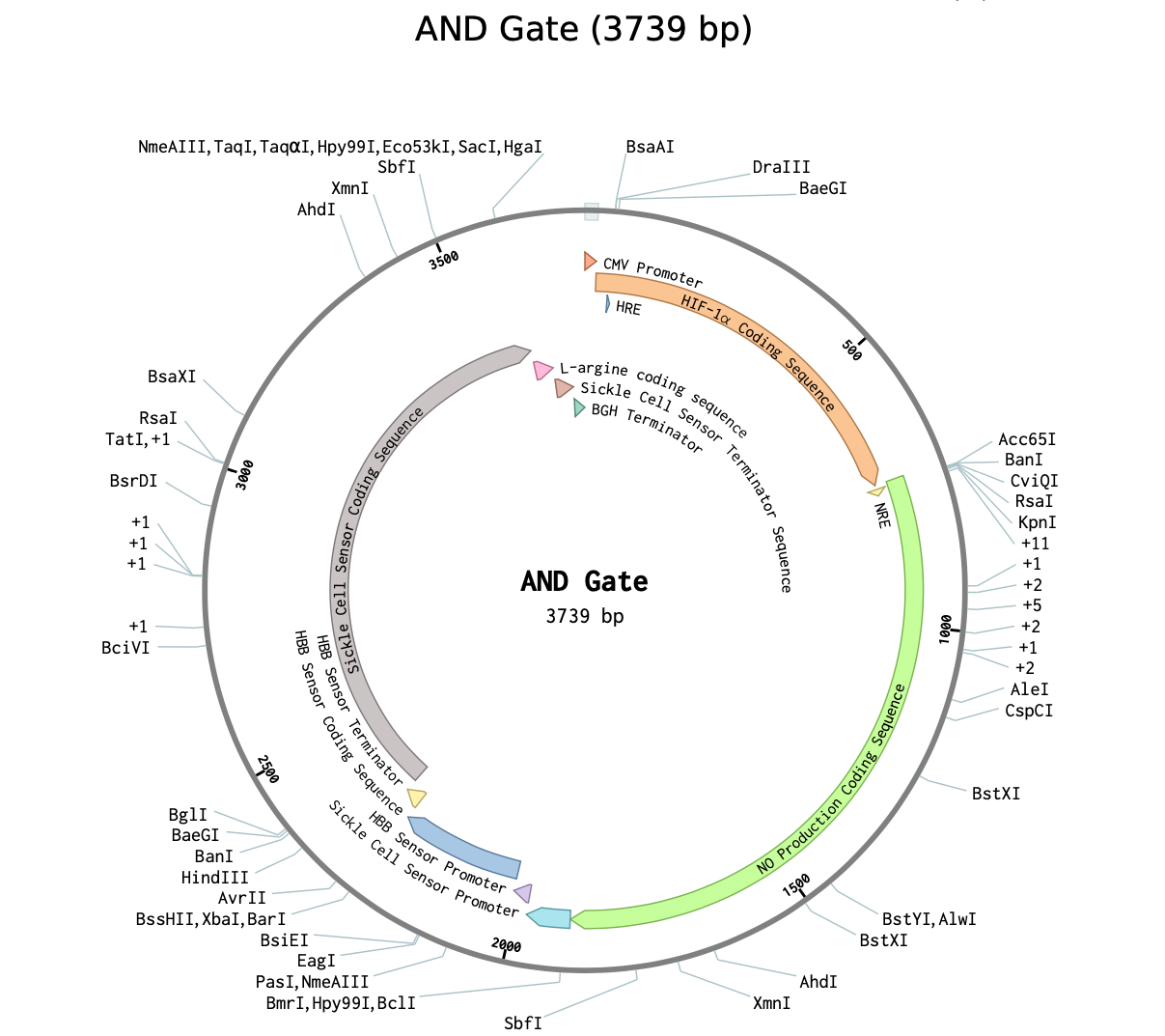
Our circuit functions as an AND gate (Figure 4) requiring the presence of both the sickle cell sensor and the lack of oxygen in order to produce NO and repair the damage caused to the retina (Wang et al., 2011). The AND gate is a fundamental logic gate used in many different kinds of circuits (digital, biological, etc.). The term “AND” means that the output of the gate will be “true” only when both inputs to the gate are “true”. If one or both inputs are “false”, the output of the gate will be “false”. So, the term “AND” refers to the fact that the gate requires two inputs to produce an output (Singh, 2014).
Inserting the AND gate into the blood vessels around the retina would involve a medical procedure called an intravitreal injection.
This procedure is minimally invasive in which a small needle is used to inject a viral vector containing the AND gate directly into the vitreous, which is a gel-like substance that fills the center of the eye. The injection would be performed under local anesthesia and would only take a few minutes (Wang et al., 2019). To ensure a longer and more stable production of NO, the plasmid will be inserted into a viral vector and then injected into the vitreous. Only injecting the DNA plasmid would cause only a very short and limited production of the NO gas, as it has a very short half-life, ranging from only 9 to 900 minutes (Kelm, 1999).
The viral vector we are using is an Adeno-Associated Virus (AAV) vector. The AAV vector would have been engineered to express our gene circuit under the control of the CMV promoter and the HIF-1α enhancer, which are capable of driving gene expression in retinal cells (Hughes et al., 2010). The AAV vector will be injected into the vitreous, it will diffuse across the retina and come in contact with the cells of the retinal blood vessels. The AAV vector will then bind to the surface of the cells in the retinal blood vessels, where it will be internalized and transported to the nucleus of the cell. Once in the nucleus, the vector will release the plasmid of our circuit and integrate into the host cell’s DNA which will then be transcribed and translated into the gene circuit (AskBio, 2020).
The gene circuit will then be secreted by the retinal blood vessels and will act only locally due to the promoters and enhancers that will ensure accuracy (Chopra et al., 2021).
Parts level
The CMV promoter is a strong promoter that drives gene expression in a wide variety of cell types. In this gene circuit, the CMV promoter drives the expression of every component of the gene circuit (Xia et al., 2006).
Our hypoxia sensor is a modified version of the HIF-1α protein, which is known to play a role in regulating oxygen levels in cells. The HIF-1α coding sequence encodes the HIF-1α protein, which is a transcription factor that regulates the gene involved in oxygen homeostasis (Weidemann & Johnson, 2008). When oxygen levels are low, HIF-1α is stabilized and activates genes that promote adaptation to hypoxic conditions. In this gene circuit, the HIF-1α protein activates the expression of the NOS gene in response to hypoxia (Weidemann & Johnson, 2008).
The sickle cell sensor promoter is a promoter that is activated specifically in the presence of sickle cells (Wang et al., 2011). This promoter also drives the expression of the NRE and HRE regulatory elements, which activate the HBB sensor promoter. The sickle cell sensor coding sequence would detect the presence of sickled cells by their abnormal β-globin HbS (Sickle Cell Disease, 2020). To detect the HbS specifically, the sensor is designed with a high affinity for HbS and a low affinity for other forms of hemoglobin. It contains a domain that can bind to the mutant HBB protein, triggering downstream signaling pathways. This specifically allows for accurate and precise detection of sickled red blood cells.
The HBB sensor promoter and coding sequence are used to detect low oxygen levels in red blood cells. This sensor contains a regulatory element known as a HRE that can detect low oxygen levels in the surrounding environment (Bruick, n.d.). Under normal oxygen conditions, a specific transcription factor known as von Hippel-Lidau binds to the HRE and targets it for degradation. However, when oxygen levels are low, HIF-1α is stabilized and binds to the HRE, activating the HBB sensor promoter to initiate the transcription of downstream genes (Weidemann & Johnson, 2008). This then facilitates the production of more β-globin to increase the amount of hemoglobin available for oxygen transport. This allows the HBB coding sequence to sense hypoxia, or low oxygen levels, which is a hallmark of SCD (Sambhara & Hsu, 2022).
The regulatory elements NRE and HRE are found in DNA sequences and will be utilized for our gene circuit. NREs are regulatory elements that negatively regulate gene expression. They act as binding sites for transcriptional repressors, which prevent RNA polymerase from binding to the DNA and transcribing the gene (Oh et al., 1997). NREs are important for controlling the level of gene expression, as they provide a way to turn off or decrease the expression of a gene. HREs are regulator elements that are activated by hypoxia. HREs act as binding sites for transcriptional activators, which help to turn on or off the expression of a gene. When oxygen levels are low, HREs help to promote the expression of genes that help improve oxygen levels (Bruick, n.d.). In the context of our gene circuit, the HRE is used to activate the expression of the NOS gene in response to hypoxia. When oxygen levels are low, the HRE binds to a transcriptional activator, which then promotes the expression of the NOS gene. This leads to the production of NO, which helps to dilate blood vessels and in turn increase blood flow to the retina. The NRE is used to provide negative regulation to the gene circuit, ensuring that the expression of the NOS gene is only activated when both sickle cell sensor and HBB sensor detect the presence of sickle cells and low oxygen levels.
The NOS coding sequence encodes the enzyme NOS, which produces NO in response to activation by the NRE and HRE regulatory elements. L-arginine is a substrate that is required for the production of NO by NOS. By adding L-arginine to the gene circuit, we could ensure that there is enough substrate available for NOS to produce NO in response to the sickle cell sensor signal (Wu et al., 2021).
The bovine growth hormone (BGH) polyadenylation signal is a commonly used terminator that is used to ensure proper termination of transcription. It also plays an essential role in stabilizing mRNA and preventing its degradation (Riberio et al., 2004). In the context of our gene circuit, the BGH polyadenylation signal would be added downstream of the NOS gene to ensure that the transcription is terminated correctly (Riberio et al., 2004). Without a proper terminator, the transcription process may continue past the end of the gene, leading to the production of unneeded transcripts which could have detrimental effects on the function of the gene circuit (Taylor, 2016). Therefore, the addition of the BGH signal will be the last step that ensures proper expression of the components of our gene circuit.
The AND gate is formed by the combination of our two inputs: the sickle cell sensor promoter (to sense sickle cells) and the HBB sensor promoter (to sense hypoxia). After sensing the two main hallmarks of SCR, the NOS gene is activated. The NOS gene is activated by the NRE and HRE regulatory elements. When both the sickle cell promoter and the HBB sensor promoter are activated, the NOS gene is expressed, and the NO is produced, which dilates the blood vessels and increases blood flow to the retina (Figure 3).
Safety
Our gene circuit for SCR treatment has the potential to be a safe and effective therapy for patients, but it is important to ensure that it is constructed, tested, and employed in a way that minimizes all harm. One key aspect of ensuring safety would be to first conduct preclinical studies with cell assays and animal models, then move on to human clinical trials (Drude et al., 2021). In addition, the gene circuit should be designed and constructed using appropriate safety measures to minimize the potential for unintended consequences. This includes the use of biocontainment measures to prevent the spread of the gene circuit outside of the intended target cells, as well as the use of safety mechanisms to ensure that the gene circuit does not activate in unintended situations or produce excessive levels of NO (Adamovicz, 2016).
To create the plasmid containing the gene, various molecular biology techniques will be employed in the lab. The DNA sequence for the various components of the circuit, such as the HBB sensor and the NO synthase gene, will be assembled using PCR amplification and restriction enzyme digestion. The final sequence will be confirmed through DNA sequencing (Chino et al., 2010). Once the plasmid is created and optimized, it will need to be able to target specific types of cells for the gene therapy. Our approach involves using a viral vector to deliver the plasmid DNA into cells (Bhisey, 2020). For our design, we will use anAV vector to deliver our plasmid as they are commonly used in other gene therapies (Wang et al., 2019). In addition, these types of viral vectors have low immunogenicity, high transduction efficiency, and have a low risk of insertional mutagenesis (Wang et al., 2019). The creation of the viral vector to deliver the plasmid will involve several steps. First, the plasmid containing the gene circuit will be amplified and purified. Then, the viral vector will be generated by packaging the plasmid DNA into the viral capsid using transfection methods. The vector will be tested for purity and functionality. The vector’s ability to specifically target desired cells will be confirmed through in vitro assays (Aronson et al., 2020).
Before the vector is used for gene therapy, it will be rigorously tested for safety and efficacy. The first preclinical study to be performed will be in a laboratory setting, before proceeding to animal studies and clinical trials (Drude et al., 2021). Cell-based assays, a standard method for testing the effectiveness and safety of gene therapies will be used. These will be performed by using cultured cells from SCD patients (Cell-Based Assays, n.d.) and measuring the response of the gene circuit to the two input conditions: low oxygen levels and sickled cells. Additionally, the assays will test the specificity of the circuit in order to ensure that it does not activate unless the two inputs are sensed (Krzyston et al., 2021). The next stage of preclinical testing would involve testing the effects of the gene circuit on blood vessels or tissue samples taken from animal models or deceased individuals (Preclinical Research | AVAC, n.d.). This would allow for the evaluation of the effectiveness and potential side effects of the circuit on a more complex biological system before moving onto clinical trials.
Once preclinical testing is complete, the treatment can move on to human trials. They will be divided into the typical three phases of human clinical trials that are recommended by the FDA (FDA, 2020). Phase I will involve the testing and treatment of a small group of SCR patients who are otherwise healthy. The focus of this phase will be on safety and feasibility (NIH, n.d.). Phase II would involve testing the treatment on a larger group of patients with SCR to evaluate safety and efficacy. The focus of this phase will be on dosing (NIH, n.d.). Phase III will involve testing the treatment in an even larger group of patients with the target condition to further evaluate safety and efficacy, as well as compare it to existing treatments. The focus of this on this phase will be on long-term safety (NIH, n.d.). The inclusion criteria for our clinical trial would include patients with SCD who are experiencing SCR symptoms and who are at least 18 years old. They also must have a confirmed diagnosis of SCD with a baseline hemoglobin level in the threshold needed to be diagnosed, which is typically in the range of 6–11 g/dL (Rishe, 2022). In addition, these patients must have not undergone a bone marrow transplant. Finally, each patient must not have a history of heart disease, severe liver or kidney disease, or any other significant disease. The trial would use a randomized, double-blind, placebo-controlled design to minimize bias and ensure accurate assessment of the gene circuit’s effects (NIH, n.d.).
The success of treatment for non-proliferative SCR stages 1 and 2 can be defined by the reduction in the frequency and severity of symptoms associated with SCR. Specifically, a significant reduction (~50%) in the number of intraretinal hemorrhages, cotton wool spots, and venous beading, all of which would be observed using retinal imaging. In terms of the treatment of blindness, we would consider an improvement of visual acuity being that of at least one line on the Snellen chart (Precision Vision, n.d.). There would also need to be a reduction in pain and an overall improvement in the quality of life of the SCR patient.
Discussions
Curative sickle cell treatment places a heavy burden on the body, with stem cell transplants and full body blood transfusions being only semi-effective. By targeting our treatment to the blood vessels around the damaged retina, we ensure a higher chance of reversing the effects of SCR.
There are potential side effects and challenges that come with our proposed gene circuit. This is especially true because gene circuits are relatively new forms of technology (Kryzszton et al., 2021). Challenges in implementing this treatment include too much NO being produced when the sickle cell sensor gene is activated. In addition, the effects of possible continuous NO production in the retina area are not well understood (Luiking et al., 2012), and could lead to potential impairment and damage in the eye (Chen et al., 2008). In the process of vasodilation, there is an inevitable risk of the blood vessel being dilated too much, leading to a deficient amount of blood pressure which could result in damaged eyesight and hypotension (Cleveland Clinic, 2022). When the sensor gene is activated and produces NO in response to the presence of sickled red blood cells and hypoxia, there is a chance then that NO is produced in the absence of sickle cells as well. This could result in increased vasodilation and inhibitions of platelet activation.
Another challenge is the potential for an immune response to the viral vector used to deliver the plasmid, which could limit the effectiveness of the treatment or cause adverse effects (Wang et al., 2019). Finally, it is important to note that there will still be associated costs for the development and production of the plasmid, as well as for the intravitreal injections (Wang et al., 2019).
Benefits of using a targeted treatment include a lower chance of off target effects. By focusing on only the retina region, the chance of the treatment binding to targets other than those intended are significantly decreased. Compared to a stem cell transplant or a full body blood transfusion which are prone to off target effects such as GVHD (Cancer Research UK, 2022).
Instead of just repairing the damaged retina, the gene circuit fixes the root cause of SCR by dilating the blood vessels around the retina (Chen et al., 2008). The sickled cells that had blocked blood flow, and therefore depriving the eye of oxygen, can now pass through and deliver the needed oxygen. It is vital that the root issue is solved instead of fixing the damaged retina that may become damaged again due to hypoxia. Using an intravitreal injection, as opposed to other methods, is beneficial. It is more accessible as it is one of the most common procedures in ophthalmology. It is less invasive than a stem cell or bone marrow transplant. There is also no need for a donor match which eliminates the complications that come with it.
It is vital that the levels of NO production in the eye are carefully monitored during all stages of testing. By keeping track of the NO levels, the dose of plasmid can be adjusted accordingly. It is also necessary to monitor patients and any symptoms that they may show or experience. Doing so could lead us to a more refined design.
One way to improve this design is to use an alternative delivery method. For instance, a non-viral delivery method may be safer and more effective (Ramamoorth & Narvekar, 2015). Instead of using a viral vector, electroporation, lipid mediated delivery, or nanocarriers could be used. In addition, we could also employ combination therapy, where we combine our gene circuit with other therapeutic approaches to further improve the treatment of SCR (LaPoint, 2022). For example, we could use it in combination with other agents that increase NO production.
Next steps
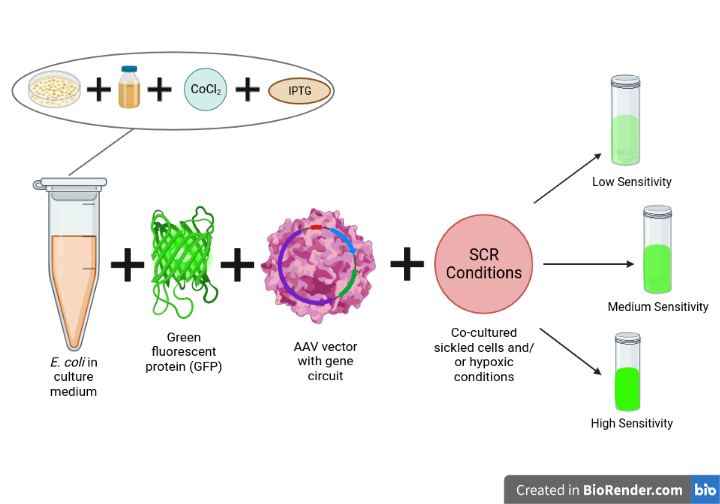
To optimize the performance of our gene circuit, we will start by optimizing the sensitivity of the components of our AND gate. To do this, we will perform experiments to determine the sensitivity of each component (Figure 5). These experiments will be designed to test the sickle cell sensor, HBB sensor, the degree of NO production, and the transcription factors.
To start, we would grow Escherichia coli cells containing our gene circuit in a sterile culture medium. We will use a sterile inoculating loop to transfer some of the E. coli cells from the stock culture into the culture medium. The cells will be grown overnight at 37°C and the culture will be shaken at 200 rpm to ensure proper aeration (Cooper, n.d.). Once the E. coli cells have grown, we will induce the expression of the gene circuit using an inducer molecule such as IPTG. We will then add a fluorescent reporter protein, such as green fluorescent protein (GFP), to the culture medium (Lentini et al., 2013). This will allow us to monitor the gene circuit’s activity by measuring the fluorescence intensity using a fluorescence microscope or plate reader (Lentini et al., 2013). Next, we will expose the E. coli cells to sickle cell conditions by co-culturing the E. coli cells containing the gene circuit with sickle cells. This will allow us to measure the sensitivity of the sickle cell sensor. We will then expose the cells to hypoxic conditions. We can use cobalt (II) chloride (CoCl2), which imitates hypoxia in vitro by stabilizing HIF-1α, and this will promote the expression of our sensors and transcription factors (Wagatsuma et al., 2020). The cells can be treated with CoCl2 for a set period of time before measuring the fluorescence intensity.
To measure the sensitivity of the transcription factors, we will introduce the expression of the reporter gene, GFP, and we will expose the cells to varying concentrations of inducer molecules and measure the fluorescence intensity using a spectrophotometer (Lentini et al., 2013). The intensity of the fluorescence will indicate the level of the expression of the fluorescent protein, which for this experiment, we will make under the control of the gene circuit. By measuring the intensity using a fluorescence microscope or plate reader, we can infer the level of gene expression, and therefore the sensitivity of the gene circuit to the different stimuli. For instance, if we observe a higher fluorescence intensity in response to sickle cells, but a lower intensity in response to hypoxic conditions, we can infer that our sickle cell sensor is more sensitive than our HBB sensor. By comparing fluorescence intensity under different conditions, we can determine the relative sensitivity of each component of the gene circuit and optimize the circuit accordingly (Figure 5).
Author contributions
All authors were instrumental in doing research on the desired topic and brainstorming a potential design. In addition, all authors contributed equally to writing the abstract and the background. E.P created the video, and wrote parts level, device level, and safety. S.J. wrote systems level and next steps. A.H and A.M wrote the discussions section, and A.M made the images and captions. In addition, all authors contributed to the editing process.
Acknowledgements
We would like to thank our BioBuilder club mentor, Mrs. Lindsey L’Ecuyer for her invaluable guidance and mentorship throughout the course of our project. Her expertise in the field of synthetic biology and her unwavering dedication to our success have been instrumental to the completion of this research. Furthermore, her passion for science and her enthusiasm for teaching have been an inspiration for all of us and have motivated us to pursue further research in the field of synthetic biology. We are truly thankful for her support and guidance, and we could not have completed this project without her.
This project was accomplished through participation in the BioBuilderClub, an after-school program organized by BioBuilder Educational Foundation. BioBuilderClub engages high school teams around the world to combine engineering approaches and scientific know-how to design/build/test their own project ideas using synthetic biology.
References
Adamovicz, J. (2016). Select agent program impact on the IBC. In C. R. Baskin & A. P. Zelicoff (Eds.), Ensuring national biosecurity (pp. 169-184). Academic Press. https://doi.org/10.1016/B978-0-12-801885-9.00010-X
Aronson, S. J., Bakker, R. S., Moenis, S., van Dijk, R., Bortolussi, G., Collaud, F., Shi, X., Duijst, S., Bloemendaal, L. T., Ronzitti, G., Muro, A. F., Mingozzi, F., Beures, U., & Bosma, P. J. (2020). A quantitative in vitro potency assay for adeno-associated virus vectors encoding for the UGT1A1 transgene. Molecular Therapy-Methods & Clinical Development, 18, 250-258. https://doi.org/10.1016/j.omtm.2020.06.002
AskBio. (2020). Adeno-Associate Virus (AAV) for Gene Therapy. https://www.askbio.com/aav-gene-therapy/
Ataabadi, A. E., Golshiri, K., Jüttner, A., Krenning, G., Danser, A. J., & Roks, A. J. (2020). Nitric oxide-cGMP signaling in hypertension: Current and future options for pharmacotherapy. Hypertension, 76(4), 1055-1068. https://doi.org/10.1161/HYPERTENSIONAHA.120.15856
Bakri, S. J., Berrocal, A., Capone, A., Choudhry, N., Ciulla, T., Dugel, P. U., Emerson, G. G., Freund, B. K., Goldberg, R. A., & Goldman, D. R. (n.d.). Sickle cell retinopathy – Patients. The American Society of Retina Specialists. https://www.asrs.org/patients/retinal-diseases/41/sickle-cell-retinopathy
Benchling. (2012). Benchling. Retrieved April 25, 2023, from https://benchling.com/epacker2005/f/lib_V80PLHAV-no-production-for-sickle-cell/seq_ghbQCe54-and-gate/edit
Bhisey, R. (2020, August 28). Viral vector & plasmid DNA manufacturing market: Plasmid DNA segment accounted for major market share. BioSpace. https://www.biospace.com/article/viral-vector-and-plasmid-dna-manufacturing-market-plasmid-dna-segment-accounted-for-major-market-share/
Brophy, J. A., & Voigt, C. A. (2014). Principles of genetic circuit design. Nature Methods, 11(5), 508-520. https://doi.org/10.1038/nmeth.2926
Bruick, R. K. (n.d.). Oxygen sensing in the hypoxic response pathway: Regulation of the hypoxia-inducible transcription factor. Genes & Development. https://genesdev.cshlp.org/content/17/21/2614#:~:text=Hypoxia%2Dinducible%20factor%20is%20regulated,inducible%20transcription%20factor%20(HIF).
Cancer Research UK. (2022, March 8). What is graft versus host disease (GvHD)?. Cancer Research UK. https://www.cancerresearchuk.org/about-cancer/coping/physically/gvhd/about
CDC. (2022, August 18). What is sickle cell disease?. Centers for Disease Control and Prevention. https://www.cdc.gov/ncbddd/sicklecell/facts.html
Sigma-Aldrich. (n.d.). Cell-based assays. Merck. https://www.sigmaaldrich.com/US/en/applications/cell-culture-and-cell-culture-analysis/cell-analysis/cell-based-assays
Chen, K., Pittman, R. N., & Popel, A. S. (2008). Nitric oxide in the vasculature: Where does it come from and where does it go? A quantitative perspective. Antioxidants & Redox Signaling, 10(7), 1185-1198. https://doi.org/10.1089/ars.2007.1959
Chino, A., Watanabe, K., & Moriya, H. (2010). Plasmid construction using recombination activity in the fission yeast Schizosaccharomyces pombe. PloS one, 5(3), e9652. https://doi.org/10.1371/journal.pone.0009652
Chopra, R., Preston, G. C., Keenan, T. D., Mulholland, P., Patel, P. J., Balaskas, K., Hamilton, R. D., & Keane, P. A. (2022). Intravitreal injections: Past trends and future projections within a UK tertiary hospital. Eye, 36(7), 1373-1378. https://doi.org/10.1038/s41433-021-01646-3
Cleveland Clinic. (2022, June 23). Vasodilation: What causes blood vessels to widen. https://my.clevelandclinic.org/health/diseases/23352-vasodilation
Cooper, G. M. (n.d.). The cell: A molecular approach. NIH.
Drude, N. I., Gamboa, L. M., Danziger, M., Dirnagl, U., & Toelch, U. (2021). Improving preclinical studies through replications. eLife, 10, e62101. https://doi.org/10.7554/eLife.62101
FDA. (2020, June 30). Conducting clinical trials. https://www.fda.gov/drugs/development-approval-process-drugs/conducting-clinical-trials
Holmes, D. (2018). Reconstructing the retina. Nature, 561(7721), S2-S3. https://doi.org/10.1038/d41586-018-06111-y
Hughes, J. M., Groot, A. J., van der Groep, P., Sersansie, R., Vooijs, M., Van Diest, P. J., Van Noorden, C. J. F., Schlingemann, R. O., & Klaassen, I. (2010). Active HIF-1 in the normal human retina. Journal of Histochemistry & Cytochemistry, 58(3), 247-254. https://doi.org/10.1369/jhc.2009.953786
Kelm, M. (1999). Nitric oxide metabolism and breakdown. Biochimica et Biophysica Acta (BBA)-Bioenergetics, 1411(2-3), 273-289. https://doi.org/10.1016/S0005-2728(99)00020-1
Krzysztoń, R., Wan, Y., Petreczky, J., & Balázsi, G. (2021). Gene-circuit therapy on the horizon: Synthetic biology tools for engineered therapeutics. Acta Biochimica Polonica, 68(3), 377. https://doi.org/10.18388%2Fabp.2020_5744
LaPoint, L. (2022, January 14). What is combination drug therapy?. Tufts Now. https://now.tufts.edu/2022/01/14/what-combination-drug-therapy
Lentini, R., Forlin, M., Martini, L., Del Bianco, C., Spencer, A. C., Torino, D., & Mansy, S. S. (2013). Fluorescent proteins and in vitro genetic organization for cell-free synthetic biology. ACS Synthetic Biology, 2(9), 482-489. https://doi.org/10.1021/sb400003y
Liu, H., & Prokosch, V. (2021). Energy metabolism in the inner retina in health and glaucoma. International Journal of Molecular Sciences, 22(7), 3689. https://doi.org/10.3390/ijms22073689
Luiking, Y. C., Engelen, M. P., & Deutz, N. E. (2012). Regulation of nitric oxide production in health and disease. Current Opinion in Clinical Nutrition and Metabolic Care, 13(1), 97. https://doi.org/10.1097%2FMCO.0b013e328332f99d
Marín-García, J. (2014). Post-genomics cardiovascular signaling pathways. In J. Marín-García (Ed.), Post-genomic cardiology (pp. 57-112). Academic Press. https://doi.org/10.1016/B978-0-12-404599-6.00003-2
Mayo Clinic. (2022, May 25). Blood and bone marrow stem cell donation. https://www.mayoclinic.org/tests-procedures/bone-marrow-donation/about/pac-20393078
Menaa, F., Khan, B. A., Uzair, B., & Menaa, A. (2017). Sickle cell retinopathy: Improving care with a multidisciplinary approach. Journal of Multidisciplinary Healthcare, 335-346. https://doi.org/10.2147/JMDH.S90630
NHS. (2022). Sickle cell disease – Treatment. https://www.nhs.uk/conditions/sickle-cell-disease/treatment/
NIH. (n.d.). The basics. https://www.nih.gov/health-information/nih-clinical-research-trials-you/basics
NIH. (2022, March 24). Stents – What are stents?. https://www.nhlbi.nih.gov/health/stents
Oh, C. K., Neurath, M., Cho, J. J., Semere, T., & Metcalfe, D. D. (1997). Two different negative regulatory elements control the transcription of T-cell activation gene 3 in activated mast cells. Biochemical Journal, 323(2), 511-519. https://doi.org/10.1042/bj3230511
Percy, M. J., Mooney, S. M., McMullin, M. F., Flores, A., Lappin, T. R., & Lee, F. S. (2003). A common polymorphism in the oxygen-dependent degradation (ODD) domain of hypoxia inducible factor-1α (HIF-1α) does not impair Pro-564 hydroxylation. Molecular Cancer, 2, 1-7. https://doi.org/10.1186/1476-4598-2-31
Precision Vision. (n.d.). Snellen eye test charts interpretation. https://www.precision-vision.com/snellen-eye-test-charts-interpretation/
AVAC. (n.d.). Preclinical research. Retrieved April 27, 2023, from https://www.avac.org/preclinical-research
Ramamoorth, M., & Narvekar, A. (2015). Non viral vectors in gene therapy-an overview. Journal of Clinical and Diagnostic Research: JCDR, 9(1), GE01-GEO6. https://doi.org/10.7860%2FJCDR%2F2015%2F10443.5394
Ribeiro, S. C., Monteiro, G. A., & Prazeres, D. M. (2004). The role of polyadenylation signal secondary structures on the resistance of plasmid vectors to nucleases. The Journal of Gene Medicine: A Cross‐disciplinary Journal for Research on the Science of Gene Transfer and its Clinical Applications, 6(5), 565-573. https://doi.org/10.1002/jgm.536
Rishe, J. (2022, July 30). What to know about sickle cell anemia and hemoglobin. Medical News Today. https://www.medicalnewstoday.com/articles/sickle-cell-anemia-hemoglobin
Sambhara, D., & Hsu, J. (2022, December 18). Sickle cell retinopathy. EyeWiki. Retrieved April 13, 2023, from https://eyewiki.aao.org/Sickle_Cell_Retinopathy
Scott, A. W. (2022, December 10). Sickle cell retinopathy: An update on management. Retina Specialist. https://www.retina-specialist.com/article/sickle-cell-retinopathy-an-update-on-management
MedlinePlus. (2020, July 1). Sickle cell disease. https://medlineplus.gov/genetics/condition/sickle-cell-disease/
Singh, V. (2014). Recent advances and opportunities in synthetic logic gates engineering in living cells. Systems and Synthetic Biology, 8(4), 271-282. https://doi.org/10.1007/s11693-014-9154-6
Taylor, J. (2016, March 31). Plasmids 101: Terminators and PolyA signals. Addgene Blog. https://blog.addgene.org/plasmids-101-terminators-and-polya-signals
Ventocilla, M., & Lee, A. G. (2021, July 22). Ophthalmologic manifestations of sickle cell disease (SCD). Medscape https://emedicine.medscape.com/article/1918423-overview
Wagatsuma, A., Arakawa, M., Matsumoto, H., Matsuda, R., Hoshino, T., & Mabuchi, K. (2020). Cobalt chloride, a chemical hypoxia-mimicking agent, suppresses myoblast differentiation by downregulating myogenin expression. Molecular and Cellular Biochemistry, 470, 199-214. https://doi.org/10.1007/s11010-020-03762-2
Wang, B., Kitney, R. I., Joly, N., & Buck, M. (2011). Engineering modular and orthogonal genetic logic gates for robust digital-like synthetic biology. Nature Communications, 2(1), 508. https://doi.org/10.1038/ncomms1516
Wang, D., Tai, P. W., & Gao, G. (2019). Adeno-associated virus vector as a platform for gene therapy delivery. Nature Reviews Drug Discovery, 18(5), 358-378. https://doi.org/10.1038/s41573-019-0012-9
Weidemann, A., & Johnson, R. S. (2008). Biology of HIF-1α. Cell Death & Differentiation, 15(4), 621-627. https://doi.org/10.1038/cdd.2008.12
Wu, G., Meininger, C. J., McNeal, C. J., Bazer, F. W., & Rhoads, J. M. (2021). Role of L-arginine in nitric oxide synthesis and health in humans. In G. Wu (Ed.), Amino acids in nutrition and health: Amino acids in gene expression, metabolic regulation, and exercising performance (pp. 167-187). https://doi.org/10.1007/978-3-030-74180-8_10
Xia, W., Bringmann, P., McClary, J., Jones, P. P., Manzana, W., Zhu, Y., Wang, S., Liu, Y., Harvey, S., Madlansacay, M. R., McLean, K., Rosser, M. P., MacRobbie, J., Olsen, C. L., & Cobb, R. R. (2006). High levels of protein expression using different mammalian CMV promoters in several cell lines. Protein Expression and Purification, 45(1), 115-124. https://doi.org/10.1016/j.pep.2005.07.008
Paula, K. Y., McAllister, I. L., Morgan, W. H., Cringle, S. J., & Yu, D. Y. (2017). Inter-relationship of arterial supply to human retina, choroid, and optic nerve head using micro perfusion and labeling. Investigative Ophthalmology & Visual Science, 58(9), 3565-3574. https://doi.org/10.1167/iovs.17-22191