Gabe Anoos, Mercy Awosoga, Scarlett Davis, Austin Wilkins ▫ Catholic Central High School, Lethbridge, Alberta, Canada
Ry Baker, Jayabhishek Chaudhary, Morteza Faraji ▫ Chinook High School, Lethbridge, Alberta, Canada
Marie Metz, Nour Salmi ▫ Lethbridge Collegiate Institute, Lethbridge, Alberta, Canada Arman Bidarian, Ziqiao Chen, Amanda Dang, Maliha Kabir, Samreet Mutti, Aubrey Nilsson, Raiyana Shams, Masataro Tatsuno, Samantha Walker, Jerry Wang, Tracy Wen, and Zitong Wu, Steven Yang ▫ Winston Churchill High School, Lethbridge, Alberta, Canada
Reviewed on 6 May 2023; Accepted on 13 July 2023; Published on 16 October 2023
With help from the 2023 BioTreks Production Team.
This study aims to explore the feasibility of using biosensors as a diagnostic tool for autoimmune diseases by detecting anti-nuclear antibodies (ANAs). ANAs are autoantibodies that attack the nucleus of a cell, and their presence is strongly associated with several autoimmune diseases. Current diagnostic methods for ANAs involve laboratory testing, which can be time-consuming and expensive. Biosensors offer a promising alternative, as they can rapidly and accurately detect ANAs in a minimally invasive manner. This study will evaluate the effectiveness of biosensors in detecting ANAs in patients with autoimmune diseases and explore their potential as a diagnostic tool for disease progression while also assessing and analyzing the efficacy of biosensor integration in household settings. Furthermore, this study aims to identify the interplay between the presence of different types of ANAs, such as Nucleolar (N-ANAs), Speckled (S-ANAs), and Homogenous (H-ANAs), and their associated autoimmune diseases. This can potentially lead to the development of more effective diagnostic and prognostic tools for these conditions. The findings from this research will contribute to the growing body of literature on the use of biosensors in healthcare and have implications for the diagnosis and treatment of autoimmune diseases.
Keywords: Antinuclear antibodies, fluorescence, autoimmune, chromoprotein
Authors are listed in alphabetical order. Robert Balog (Catholic Central High School), Laura Keffer-Wilkes, Marcel E. Michailides, and Atta Rahman (University of Lethbridge, Alberta, Canada), and Riley Kostek (Chinook High School) mentored the group. Please direct all correspondence to kefferwilkesl@uleth.ca.
Antinuclear antibodies
ANAs are a particular kind of autoantibodies that target various components of the cell nucleus, including DNA, RNA, and proteins, which usually are not recognized as foreign by the immune system (Kumar et al., 2009). Autoantibodies are antibodies generated when the body’s immune system is unable to differentiate between non-self and self-antibodies and specifically react with self-antibodies (Dutta, 2021). ANAs are specialized to target structures within a cell’s nucleus, such as proteins or nucleic acids via binding. Hence, the presence of ANAs in an individual can be used to indicate an autoimmune disease.
Autoimmune disorders
There are over 80 autoimmune diseases (ADs) that affect more than 4.5% of the world’s population (Bender et al., 2021). These autoimmune diseases cause a wide range of symptoms; the most common being fatigue, weakness, rashes, and fever. Regardless of risk factors, anyone can develop an AD; however, an individual is more likely to develop one if they are: older, overweight, female, have a history of smoking, or have a family history of ADs (7 Risk Factors for Autoimmune Disease, n.d.). The ADs lupus, multiple sclerosis (MS), rheumatoid arthritis (RA), and Sjögren’s syndrome (SS) are of particular concern.
Lupus can cause chronic inflammation and pain in any body part (What Is Lupus?, 2020). Around 30% of lupus patients have anti-dsDNA, a specific ANA that helps distinguish lupus from other autoimmune diseases (Johns Hopkins Lupus Center, 2019). There are four types of lupus: systemic lupus erythematosus (SLE) (the most common). cutaneous lupus (only affects the skin), drug-induced lupus, and neonatal lupus. Lupus is a multifactorial AD whereby a combination of genetic, environmental, hormonal, and immunological factors play a role in its pathogenesis.
MS occurs when the immune system attacks the myelin sheath – the protective covering of nerve fibres – in the central nervous system, which leads to communication problems between the brain and the rest of the body. Over time, MS can cause permanent damage to nerve fibres (Mayo Clinic, 2022). Life expectancy for those with MS is about 5 – 10 years less than the average lifespan of most individuals (Overview: Multiple Sclerosis, n.d.).
RA occurs when the immune system attacks the lining of the joints (synovial membrane), causing swelling and inflammation (Rheumatoid Arthritis – Symptoms and Causes, 2023). RA is characterized by rheumatoid factors (RF) – antibodies that are commonly found in the blood of people with rheumatoid arthritis (Branch, 2023; Figure 1) – and anti-citrullinated peptide antibodies (RA Pathophysiology • Johns Hopkins Arthritis Center, n.d.). People with rheumatoid arthritis usually experience pain, stiffness, swelling, and tenderness in multiple joints (also occurs in both sides of the body, e.g., both hands or knees.) This can make it difficult to do everyday tasks such as making a fist, bending knees, combing hair, or buttoning clothes (Rheumatoid Arthritis | Health Topics | NIAMS, 2022). Without treatment, the risk of death is increased, and in most people with RA, life expectancy is ten years less than those without (How Is Lifespan Affected by RA? | NRAS, 2020).
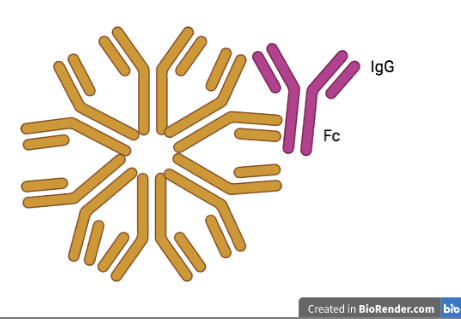
| Figure 1. Illustration of an RF Antibody. The Fc segment of the IgG antigen is attached to the main antibody. |
|---|
SS is a persistent and long-lasting autoimmune disorder identified by its two most prominent symptoms: dry eyes and mouth (Sjogren’s Syndrome – Symptoms and Causes, 2022). The ANAs in patients with SS include anti-Ro/SSA, anti-La/SSB, and RFs (Fayyaz et al., 2016). SS can occur by itself, which is the primary form, or with other rheumatic diseases, such as RA or SLE, which is the secondary form of SS. Most people with SS usually have a normal life expectancy and live without any significant issues, although regular medical check-ups are advised (Sjögren’s Syndrome, n.d.).
Currently, there is no cure for ADs, and treatment focuses on alleviating symptoms and preventing progression. The use of medications is the primary approach in autoimmune disorders management. Treatment of autoimmune disorders typically involves the use of anti-inflammatory drugs, such as nonsteroidal anti-inflammatory drugs (NSAIDs), corticosteroids like prednisone or methylprednisolone, immunosuppressive agents, and biological response modifiers. These medications work by suppressing the immune system and reducing inflammation, thus helping to relieve symptoms and prevent disease progression. However, the use of these medications have to be carefully considered, as they can have significant side effects and may not be effective in all patients. Treatment decisions are made by healthcare professionals based on the individual patient’s clinical presentation, disease severity, and other relevant factors (Lupus – Diagnosis and Treatment, 2022).
Current testing methods
RA can be diagnosed through measuring the level of RF in a patient’s blood. The RF test works by detecting the presence of RF autoantibodies in the blood. A positive RF test result indicates that the patient has RF antibodies in their blood and should receive follow-up testing (Ingegnoli et al., 2013). However, a positive RF test result alone is not enough to diagnose an AD, and further testing and evaluation are usually needed to confirm the diagnosis. Additionally, some people with ADs may not have detectable levels of RF in their blood, so a negative test result does not necessarily rule out the possibility of an autoimmune disease (Rheumatoid Arthritis, n.d.).
Anti-cyclic citrullinated peptide (anti-CCP) tests are blood tests used to detect the presence of antibodies against citrullinated peptides in the blood of people with ADs (MedlinePlus, n.d.). Citrullination is a process in which the amino acid arginine is converted into citrulline, which can result in changes in protein structure and function (Pruijn, 2015). This process is thought to play a role in the development of ADs such as RA. Anti-CCP antibodies are present in up to 80% of people with RA, and their presence is associated with more severe disease and joint damage (Rheumatoid Arthritis, n.d.-b). As such, the anti-CCP test is considered to be a more specific test for RA than the RF test, and It can also be used to monitor disease activity and response to treatment (Braschi, 2016). However, like all diagnostic tests, the anti-CCP test is not perfect and can produce false positive or false negative results in some cases.
ANA tests are a type of blood test used to detect the presence of antibodies that attack the body’s own cells and tissues, specifically targeting the cell nucleus (Mayo Clinic, 2022). ANA biosensors detect ANAs in biological samples such as blood, urine, or saliva. They work when a target antigen binds to ANAs, creating a detectable signal, such as fluorescence. Current ANA detection methods are based on blood samples and are highly inefficient. One study by Robert L Rubin and Konstantin N Konstantinov suggests that an electrochemical method would be most efficient. In which target auto antigens are derived from a membrane made of protein and nucleo-protein mix. As the serum or blood runs through the membrane the auto antigens would bind with the patient’s autoantibodies in the sample. The electric release accompanying the reduction reaction of substrate and enzyme, which is measurable, tells us the amount of ANAs present (Rubin & Konstantinov, 2016). In another study by Konstantinov and Rubin (2017), an alternative biosensor was proposed, which involves the creation of an autoantigen-rich substrate membrane and the forceful transportation of a serum sample through it. In just three minutes, a complex of autoantigen-antibody is formed, which is then detected through the energy of the reduction reaction. The energy of the reduction reaction is measured in energy units, which provides a readout on display indicating the amount of antigen-bound antibodies present (Konstantinov & Rubin, 2017). Both studies share a similar approach: creating an antigen-rich membrane that binds with the present substrates in the serum sample to indicate the presence of ANAs. The detection of ANAs is accomplished by measuring the energy of the reduction reactions, providing a sensitive and reliable means for ANA detection.
The detection of ANAs is commonly achieved by one of two methods. The first method is Fluorescent ANA (FANA) testing, which involves adding a serum to a cell-substrate followed by adding fluorescent antibodies. This causes the antibody bound to ANAs to fluoresce, and the pattern of antibodies in the nucleus can be used to determine the specific disease. FANA is commonly used due to its high accuracy (Anwar et al., 1997). The second method is the Enzyme-Linked Immunosorbent Assay (ELISA), which detects the presence of ANAs by adding antibody-bound specialized enzymes to the serum, causing a color change to indicate the presence of the antibody (Alhajj, 2023). However, there are also some ethical issues with ANA biosensor testing. For example, up to 15% of people who are positive for ANAs do not actually have an AD. This can place unnecessary stress on the individuals who were tested. Additionally, biosensors can be costly and may not be within an individual’s budget.
Although, there are various strengths and advantages of ANA tests compared to anti-CCP and RF tests. Firstly, ANA tests have broader diagnostic potential. ANA tests are highly sensitive and can detect a wide range of ADs, including SLE, SS, and mixed connective tissue disease (Kumar et al., 2009). In contrast, anti-CCP tests are specifically designed to detect the presence of anti-cyclic citrullinated peptide antibodies, which are associated with RA (Rheumatoid Arthritis, n.d.-b). RF tests are used to diagnose RA but are not specific to this disease and can be positive in other autoimmune conditions (Ingegnoli et al., 2013). Furthermore, early detection is another advantage of ANA tests; these tests can often detect ADs in their early stages, before symptoms become apparent (The Royal Australian College of general Practitioners, n.d.). Finally, versatility is another aspect that favors ANA tests. It can be performed using different methods and can be customized to detect specific types of ANA antibodies (Synder, 2019). This allows for greater flexibility in testing and can help improve accuracy and reduce false positives. In contrast, anti-CCP and RF tests are usually performed using standardized methods and are less customizable.
Indirect Immunofluorescence (IIF) or “Secondary Immunofluorescence” is the commonly preferred method for screening ANAs in the body (Allaire et al., 2015). The method utilizes fluorescence microscopes to view the target molecule’s distribution in biological samples and tissues. IIF is often used in tandem with other non-antibody-related staining processes typically for DNA (Allaire et al., 2015). The benefits of this method are a higher sensitivity than other processes such as ELISA and are generally more affordable. Additionally, the samples for IIF are fixated (immobilized), allowing analysis at specific periods (Im et al., 2020). IIF relies on a diluted, primary unlabelled antibody which is specific for the antigen of interest. This antigen could include various targets with the centromere, Mi-2, RNP, and RNA Polymerase III being some of the hundreds of possibilities. After the unlabelled antibody is applied to the samples, a secondary antibody which carries the fluorochrome then recognizes the primary antibody and binds to it, causing fluorescence that can be seen under a microscope (Allaire et al., 2015). Figure 2 illustrates a generalized IIF process.
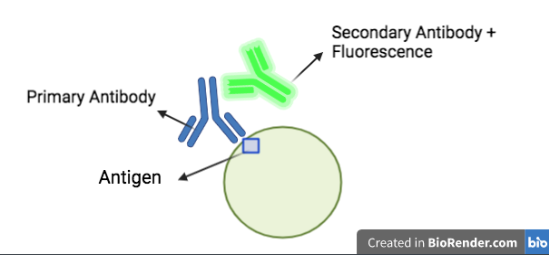
| Figure 2. Diagram of IIF, the secondary antibody attaches to the primary antibody-antigen complex to produce fluorescence. |
|---|
The two main microscopes used in IIF are epifluorescence and confocal microscopes (Allaire et al., 2015). Epifluorescence microscopes are the simpler of the two used in IIF. Fluorescence detection and fluorochrome excitation occur in the same light path of the microscope. However, only reflected light can effectively reach the sample due to most of the excitation light being transmitted through the sample. Hence, confocal microscopes are more advantageous in the IIF procedure. The confocal microscope has greater resolution due to a pinhole on the lens, which allows it to eradicate light that is out of focus. Although, this comes at the cost of longer exposures due to the shortened amount of light total to view the samples (Allaire et al., 2015).
However, there do exist some disadvantages when using IIF. For one, the IIF procedure is more complex and extremely time-consuming (Copple et al., 2014). The application of the unlabelled antibody to samples is required to incubatefrom as long as 1.5 hours to 12 hours (Proteintech, n.d.). The results of IIF are analyzed for specific patterns of immune deposition (Xuan et al., 2017). Hence, interpreting IIF results are prone to subjectivity and reader bias since one reader could see a pattern while another might not. Furthermore, the reagents, primary antibodies, and secondary antibodies used on the samples must be extremely specific so as to avoid false results.
Detection of ANAs in human serum samples have also employed point-of-care (POC) diagnostic methods. The POC methods permit portable on-site diagnosis with high efficiency without the need for centralized clinical laboratories and highly expensive lab instruments. Therefore, these methods can provide a fast diagnosis of ADs, thereby improving patient outcomes and reducing healthcare expenses. The amperometric electrochemical biosensor is a promising POC diagnostic method that relies on converting a biochemical reaction into a measurable electrical signal. The majority of these electrochemical sensors utilize a three-electrode assembly wherein a working electrode is separated from a reference electrode by the electrolyte, and a third counter electrode is connected to the reference to facilitate current flow. The target molecule first binds to the biosensor, an immobilized electroactive species affixed to the working electrode and triggers a reduction-oxidation (redox) reaction. Redox refers to a chemical reaction where electrons are lost or gained between reactants. The loss of electrons results in an increased state of oxidation, and the gaining of electrons results in a decrease of oxidation state, regarded as reduction. As the electrical current generated from this reaction is proportional to the target molecule concentration, the process provides a rapid and robust method of molecular target quantification in the biological sample. For example, an electrochemical amperometric immunoassay flow-through biosensor with the capability to detect ANAs in human serum samples in approximately 20 minutes has been developed by Rubin and Konstantinov (2016).
Systems level
Rapid antigen tests have become widely used for the diagnosis of COVID-19 worldwide. These tests employ immunochromatography techniques, also known as lateral flow assays, to detect viral proteins. They are designed for convenient use outside of hospital laboratories, making them accessible to the general public or non-expert individuals.
Our aim is to develop a lateral flow assay for the detection of ANAs in patients with suspected ADs. Saliva or urine samples will be collected and treated with a lysis buffer to release cellular material. The resulting sample will be applied to an activated membrane (Figure 3). Similar to the rapid COVID-19 test, the appearance of colored lines on the membrane will indicate a positive result, which can be observed by the patient.
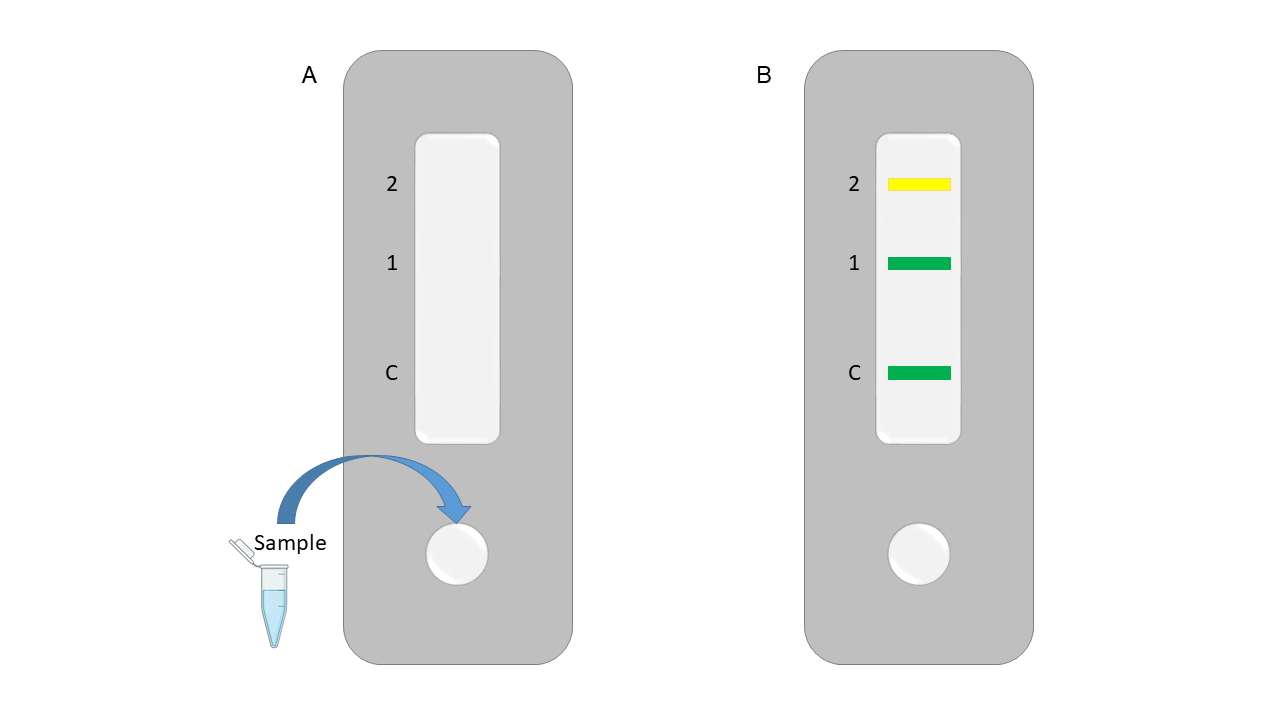
| Figure 3. Rapid diagnostic test schematic for diagnosis of ANAs using a chromoprotein biosensor. |
|---|
To detect the ANAs a recombinant chromoprotein will be constructed that can change colour based on which ANA is present. The engineered chromoprotein will be immobilized on the membrane and upon binding to an ANA change colour. The test will be sensitive enough to differentiate between different ANA species and provide a more specific diagnosis than the current detection methods.
Device level
ANA biosensor design
The ANA biosensor design will be based on work done by Stewart Loh’s lab (John et al., 2022). In their 2022 paper, they created a switchable GFP biosensor that can change colour (green to yellow) upon binding to a ligand. This technology has the potential to be used to detect a wide array of targets and could be expanded to use chromoproteins instead of fluorescent proteins.
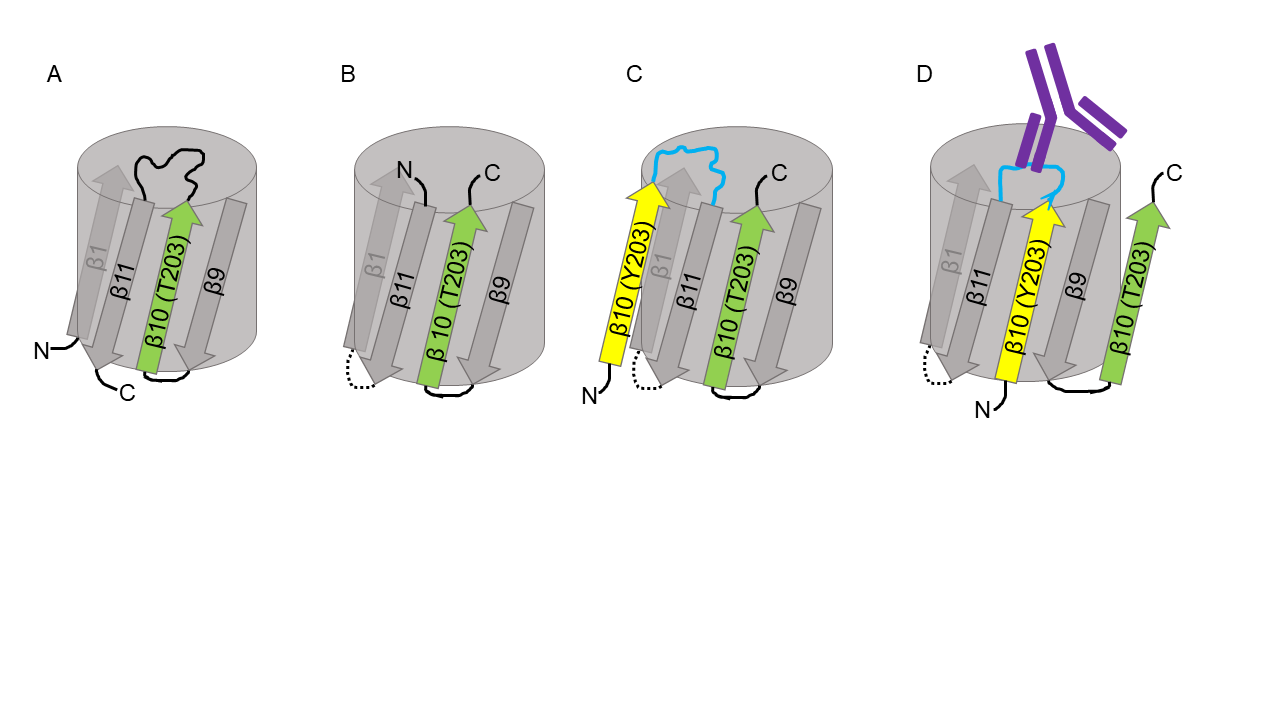
Our design uses the potential modularity of this engineered protein where any unstructured peptide region could replace the cpFKBP sequence (Figure 4 in blue) and upon binding to its target to cause the overall conformational change of the protein and a change in fluorescence. For the ANA biosensor, the epitope sequence would be replaced with antigens that will bind to ANAs (see next section).
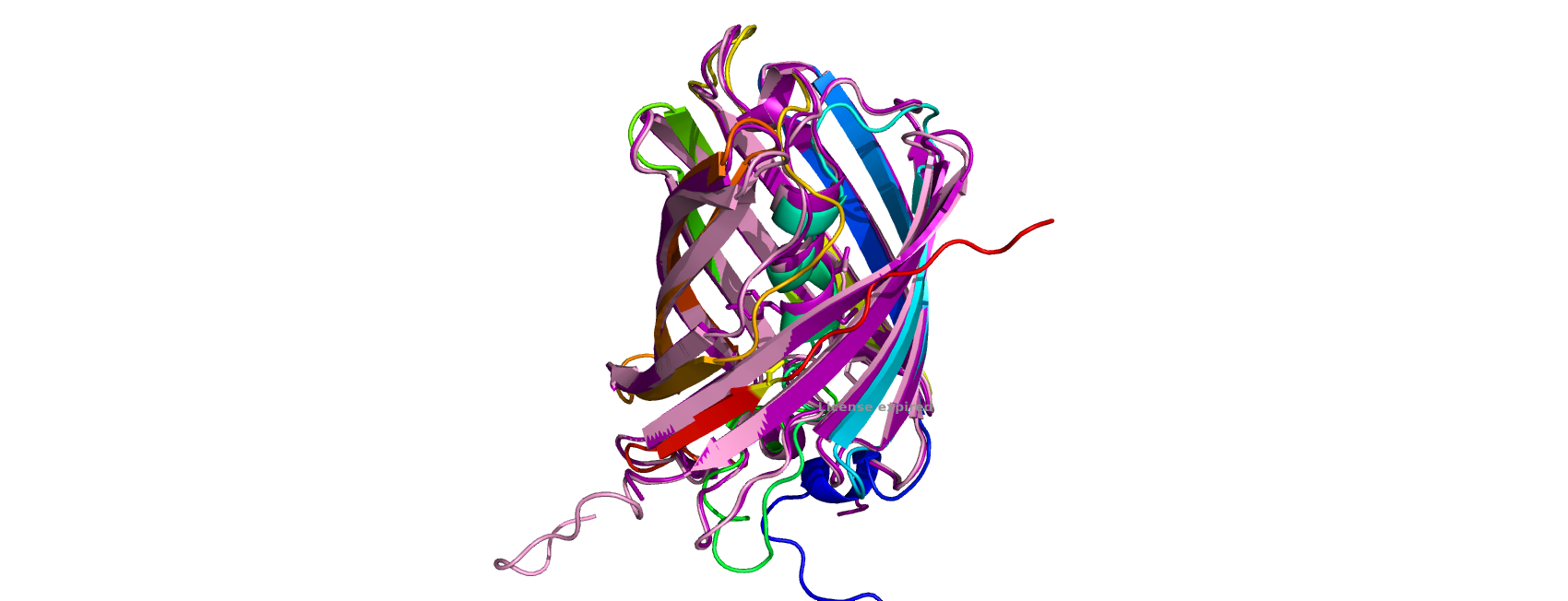
While a fluorescent protein was used in John et al. (2022), our design will attempt to utilize a chromoprotein instead. The advantage of this method is that the chromoprotein colour change will be more visible without the use of any specialized equipment. Several chromoproteins were developed and characterized by the 2013 Uppsala iGEM team. From these, we chose to use tsPurple (BBa_K1033905) as our starting chromoprotein model system. This protein produces a rich purple colour when expressed in Escherichia coli and was found to be quite stable and easy to express (Liljeruhm et al., 2018). The chromophore sequence of tsPurple is CMYG. Another chromoprotein, asPink, has the same chromophore sequence. We created 3D models using Swiss-model of tsPurple and asPink since no structural data were available. Both proteins have the common beta-barrel shape of most known fluorescent and chromoproteins with the chromophore running through the center (Figure 5). The models were aligned with the GFP biosensor from John et al. (2022).
Next, an amino acid sequence alignment was completed in order to identify the residues that may contribute to the colour difference between tsPurple and asPink (Figure 6). Through systematic site-directed mutagenesis and analysis of the predicted protein structure, the identity of the essential amino acids will be elucidated. The tsPurple will then be further engineered to be able to switch colours from purple to pink, similar to the biosensor published by John et al. (2022).
ANA Epitopes
In the biosensor design, an epitope is required for specific binding to the ANA target. Upon binding, the epitope region would fold, causing a conformational rearrangement of the biosensor and changing the colour output (Figure 7). The first epitope to be tested will be from Scl-70 or DNA topoisomerase I (Gussin et al., 2001). ANAs against Scl-70 have been found to be elevated in patient’s suffering from SLE. Interactions between an immunodominant peptide of topoisomerase I (residues 349-368) and anti-topoisomerase I antibodies was shown through molecular dynamics modeling (Kongkaew et al., 2019). Further investigation into the specific Scl-70 amino acid sequence that lupus associated ANAs interact with has not been completed but will be necessary for this project.
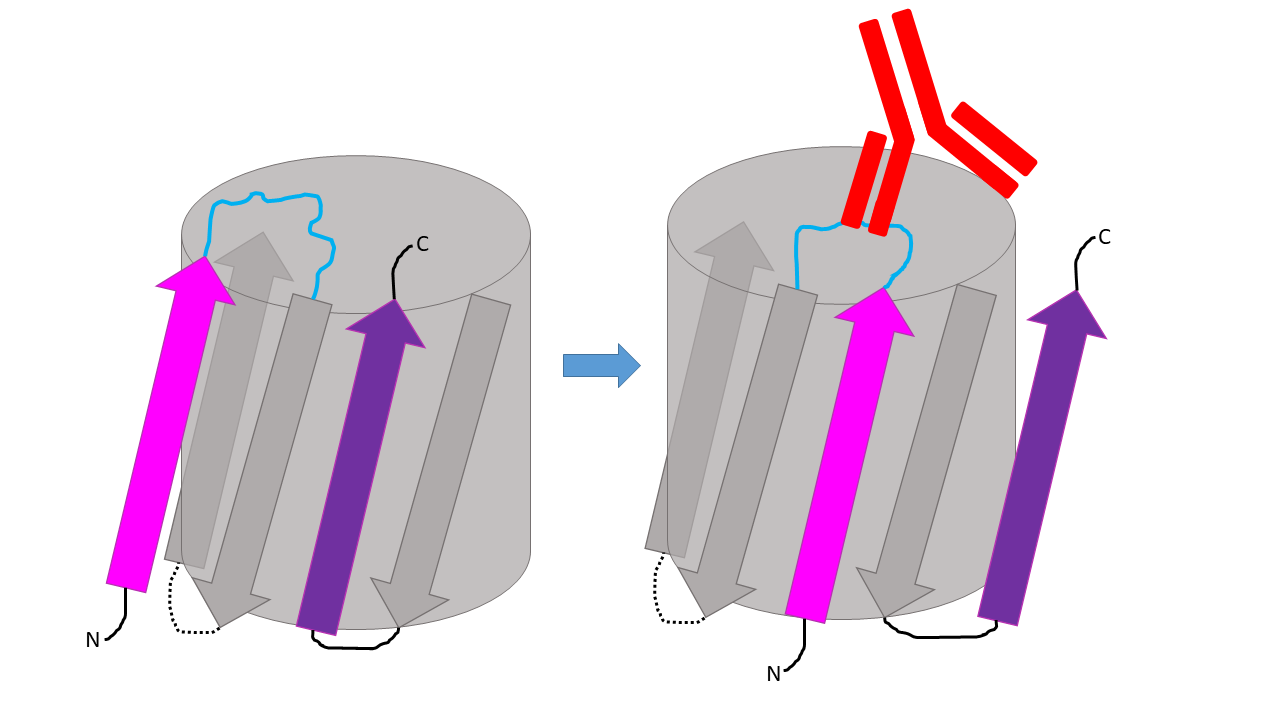
| Figure 7. Proposed structural changes of chromoprotein biosensor in response to ANA binding of epitope. |
|---|
PNP Stabilization
The stability of the designed chromoprotein biosensor is critically important. Long-term shelf life is necessary for the product to be effectively utilized as an at-home test. In 2011, a study from the Department of Chemical and Biological Engineering at Korea University, they researched the stability of protein nanoparticles (PNPs) and the efficacy of lyophilization for the long-term stability of PNPs encapsulating GFP. The team lyophilized three kinds of gFPNP samples and measured their fluorescence intensities in diverse storage temperatures (-20 °C – 37 °C). They found that regular gFPNPs maintained most of their fluorescence intensities for about 2 weeks before they denatured rapidly, while lyophilized ones maintained their intensities for up to 4 weeks before denaturing. Afterwards, they tested lyophilized gFPNPs with added stabilizers, especially tween 80 and sucrose. They found that when the stabilizers were present, the fluorescence and structure of the lyophilized gFPNPS was maintained successfully derived for up to 10 weeks. In our design, we can also utilize the lyophilization process combined with the tween 80 and sucrose stabilizers to enhance the stability of our GFPs.
Parts level
Our project will utilize chromoproteins as they are easily visible under ambient light when expressed, through absorption of visible light rather than fluorescence like GFP.
tsPurple (BBa_K1033905), or TinselPurple, is a synthetic purple chromoprotein designed for expression in E. coli. The protein has a molecular mass of 26.27 kDa and sequence length of 708bp.
asPink (BBa_K1033933), named for anemonia sulcata, the anemone from which it was derived, is a pink chromoprotein. It has a molecular mass of roughly 25.9 kDa, sequence length of 702bp, and an absorption maximum of 572nm.
Both tsPurple and asPink emit strong colour within 24 hours of incubation. Chromoproteins are homologous with GFP, and share its beta barrel structure and central chromophore.
Plasmid design
The plasmid is currently available through Addgene and utilizes the following iGEM parts: constitutive promoter (BBa_J23110), ribosome binding site, and a double terminator (BBa_B0014).
For our biosensor testing, the tsPurple gene will be cloned into a pET28a plasmid with an IPTG inducible promoter and tagged with a hexa-histidine label for purification.
Safety
Team members will complete a Workplace Hazardous Materials Information System WHMIS safety course before they can enter and work in the lab. All BSL-1 precautions will be followed when completing any lab work. Since this study focuses on a very early biosensor prototype, no patient samples will be used. Instead in vitro protein biosensors and purchased antibodies will be used for the model system, thus avoiding the use of any potential hazardous biological material.
ANA testing has become common in clinical practice as it allows earlier diagnosis and more effective treatment of autoimmune conditions. However, the 2023 Lethbridge High School iGEM Team will face numerous challenges when it comes to creating a mobile testing kit because as with any new technology, there are ethical considerations that must be taken into account.
One of the most notable ethical concerns surrounding ANA testing is informed consent. Patients must be fully informed of the risks and benefits of testing before undergoing the procedure and subsequent treatment. Additionally, patient confidentiality must be maintained, as the results of ANA testing can have long lasting implications for a patient’s health and well-being (Courtright-Lim & Drago, 2020). Providers, such as our iGEM team promoting our testing methods, must ensure that patients fully understand the potential consequences of testing, and that their consent is obtained in a meaningful, private, and informed manner.
The use of ANA testing may have significant societal implications. As with any new technology, there is a risk that ANA testing may exacerbate existing health disparities around the world. This is because it is difficult to control who has financial access, so as such, it is essential that equitable access to testing is maintained, regardless of a patient’s race, ethnicity, socioeconomic status, or geographic location. Additionally, the interpretation of ANA testing results may be influenced by cultural factors, which the iGEM team must take careful considerations to ensure that patients receive appropriate care.
Discussions
This multicolour chromoprotein biosensor based rapid test holds the ability to detect ANAs quickly and accurately. By utilizing multiple colourful proteins that are designed to bind specifically to different ANA subtypes, this test will allow a comprehensive analysis of patient serum. These specified results will allow the patient to receive specialized care towards their projected AD. This diagnostic tool offers a new, non-invasive, and easy to administer approach for the early detection of autoimmune diseases. In this regard, the hardware of our project will be designed to allow patients to test from the comfort of their own home and receive results in a matter of hours. With our solution we hope to achieve an eco- and user-friendly alternative to high maintenance hospital tests, which have been a significant contribution in producing medical waste. (Cheng et al., 2009)
An area that we must be cautious of is the probability of false positives. Due to the sensitivity of our biosensor, a false positive is possible. When low concentrations of the target ligands are present in the tested sample, it can mean nothing. Having a medical history containing some different infectious diseases as well as cancer can lead to production of ANA’s that is not correlated to the presence of an AD. It is also most common to have a false positive test if you are a woman over 65 years of age. Given this flaw in ANA test design, our group will work to minimize the risk of false positives while maintaining high levels of sensitivity. A standard curve will be developed with known amounts of ANAs to act as a control mechanism for testing. If a solution cannot be found through the design of a biosensor, we will make sure to be aware of a given patient’s medical history, so we may fully understand the probability of receiving a false positive test and work to minimize panic (Mayo Clinic, 2022b).
Next steps
To build the biosensor, we will first utilize site-directed mutagenesis (SDM). This in vitro procedure is a technique used to introduce specific changes or mutations at desired locations in a DNA molecule. SDM involves the use of synthetic primers that contain a chosen mutation. These primers are designed to anneal to a template DNA molecule in a way that successfully introduces the desired changes. (Edelheit et al., 2009) Most often a pair of oligonucleotides are used as primers to bind with the template on opposite DNA strands, using deoxynucleic triphosphates as building blocks to synthesize new DNA strands in the presence of a thermostable DNA polymerase enzyme. These newly synthesized DNA strands serve as templates for subsequent PCR rounds. This procedure initially requires the creation of DNA primers designed to contain a desired mutation, which also has the ability to hybridize to the template DNA at a specified location. From here, these primers are used in a PCR to amplify the target DNA sequence (Reikofski & Tao, 1992) When a sufficient amount of mutant DNA is generated, it can be isolated from the original template by gel electrophoresis or by direct transformation into a suitable host organism, such as (E. coli).
By employing this process in our project, we hope to identify the essential amino acids that contribute to the colour of the protein for our biosensor. Once these have been identified, we can introduce a new beta-strand and epitope region into the protein. Upon binding to a specific analyte, a conformational change will occur. This change will be coupled with a change in the protein’s optical properties. In this case, our protein will switch from a purple colour to a pink colour upon the detection of analyte. Protein overexpression in E. coli is centered on the production of recombinant proteins. E. coli is used because of its incredibly fast growth which allows it to have a doubling time of around 20 minutes. This naturally makes it a fantastic protein expression tool. The gene of interest will be cloned into an appropriate plasmid that is compatible with bacterial expression. Conveniently, both tsPurple and asPink are available through Addgene.org. Both genes have been cloned into pSB1C3 plasmid and are BioBrick compatible. Unfortunately, the construct does not contain a purification tag. Therefore, primers will be used to add an N-terminal his-tag to the protein for downstream affinity purification.
While John et al. (2022) could utilize fluorescence spectroscopy to measure changes in their biosensor, the proposed chromoprotein biosensor must use direct visualization. Therefore, a standard colour spectrum will be created where users can compare their test to determine the level of detection, similar to a pH test strip.
Affinity assays will also be completed to measure the sensitivity of the biosensor. Additionally, stability testing of the biosensor will have to be completed to determine the shelf life of our product.
Author contributions
Video was produced by A.D., J.W., S.W., A.N., S.M., M.A., M.S., Z.C., S.D., N.S., M.K., and T.W. Background section was written by M.A., G.A., A.D., S.Y., M.F., M.T., M.C., A.N., and M.M. Systems, device, and parts sections were written by J.C., R.S., M.T., M.K., and Z.W. Safety section was written by A.B, A.D., J.W., and A.W. Discussion and next steps sections were written by S.D., A.B., Z.C., and T.W. Article was edited by L.K.W., M.E.K., and A.R.
Acknowledgements
The authors of this manuscript would like to thank our mentors for their instruction and editing assistance during the writing process. We’d also like to recognize BioRender as we used their software to create many of the figures included in this manuscript. Finally, we’d like to recognize the many individuals in our lives who have been diagnosed and are affected every day by autoimmune diseases; for they have served as the inspiration behind our project.
References
Alhajj, M. (2023, January 30). Enzyme linked immunosorbent assay. StatPearls Publishing.
Allaire, A., Picard-Jean, F., & Bisaillon, M. (2015). Immunofluorescence to monitor the cellular uptake of human lactoferrin and its associated antiviral activity against the hepatitis C virus. Journal of Visualized Experiments, 104. https://doi.org/10.3791/53053
Affinity chromatography – An overview | ScienceDirect topics. (n.d.). Retrieved April 29, 2023, from https://www.sciencedirect.com/topics/neuroscience/affinity-chromatography#:~:text=Affinity%20chromatography%20is%20a%20method,interaction%20between%20the%20two%20molecules
American College of rheumatology (ACR) (n.d.). Rheumatoid Arthritis. https://rheumatology.org/patients/rheumatoid-arthritis
American College of Rheumatology (ACR). (2023). Sjögren’s Syndrome. https://rheumatology.org/patients/sjogrens-syndrome
Anwer, F., Abou-Azama, A. M., Mandourah, A. Y., Borja, M., Soriano, R., & Mercado, S. G. (1997). Fluorescent antinuclear antibody and anti-double-stranded DNA antibody testing: A four years experience. Journal of Pakistan Medical Association (JPMA), 47(9), 226–228.
Bender, M. B., Christiansen, J. C., & Quick, M. Q. (2021, September 1). The terrible toll of 76 autoimmune diseases. Scientific American. https://www.scientificamerican.com/article/the-terrible-toll-of-76-autoimmune-diseases/
Bhalla, N., Jolly, P., Formisano, N., & Estrela, P. (2016). Introduction to biosensors. Essays in Biochemistry, 60(1), 1–8. https://doi.org/10.1042/ebc20150001
Bio-Rad. (n.d.) Introduction to size exclusion chromatography. https://www.bio-rad.com/en-ca/applications-technologies/introduction-size-exclusion-chromatography?ID=MWHAXJKG4#:~:text
Bio. (n.d.). https://www.bio-rad.com/en-ca/applications-technologies/introduction-affinity-chromatography?ID=MWHAVG4VY
National Institute of Arthritis and Musculoskeletal and Skin Diseases Branch, N. S. C. a. O. (2021, April 7). Sjögren’s syndrome.. https://www.niams.nih.gov/health-topics/sjogrens-syndrome/diagnosis-treatment-and-steps-to-take
Branch, N. S. C. a. O. (2023, March 3). Rheumatoid Arthritis. National Institute of Arthritis and Musculoskeletal and Skin Diseases. https://www.niams.nih.gov/health-topics/rheumatoid-arthritis
Branch, N. S. C. a. O. (2023b, March 3). Rheumatoid Arthritis. National Institute of Arthritis and Musculoskeletal and Skin Diseases. https://www.niams.nih.gov/health-topics/rheumatoid-arthritis/diagnosis-treatment-and-steps-to-take
Braschi, E. (2016, March 1). Anti-CCP: A truly helpful rheumatoid arthritis test? PubMed Central (PMC). https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4984588/
CCTS. (n.d.). Procedures/risks: Blood draws, IV lines, vitals_template. Retrieved April 17, 2023, from https://ccts.osu.edu/sites/default/files/inline-files/Procedures_Risks-Blood_draws-IVs-vitals-etc.pdf
Centers for Disease Control and Prevention (CDC). (2022, June 28). Lupus in Women. https://www.cdc.gov/lupus/basics/women.htm
Cheng, Y. W., Sung, F. C., Yang, Y., Lo, Y. H., Chung, Y. T., & Li, K. C. (2009). Medical waste production at hospitals and associated factors. Waste Management, 29(1), 440–444. https://doi.org/10.1016/j.wasman.2008.01.014
Copple, S. S., Jaskowski, T. D., Giles, R., & Hill, H. R. (2014). Interpretation of ANA indirect immunofluorescence test outside the darkroom using NOVA view compared to manual microscopy. Journal of Immunology Research, 2014, 1–7. https://doi.org/10.1155/2014/149316
Courtright-Lim, A., & Drago, M. (2020). Ethics of genetic testing. Medicine. https://doi.org/10.1016/j.mpmed.2020.07.014
Dutta, S. S. (2021, March 9). What are Autoantibodies? News-Medical.net. https://www.news-medical.net/life-sciences/What-are-Autoantibodies.aspx
Edelheit, O., Hanukoglu, A., & Hanukoglu, I. (2009). Simple and efficient site-directed mutagenesis using two single-primer reactions in parallel to generate mutants for protein structure-function studies. BMC Biotechnology, 9(1), 61. https://doi.org/10.1186/1472-6750-9-61
Fayyaz, A., Kurien, B. T., & Scofield, R. H. (2016). Autoantibodies in Sjögren’s Syndrome. Rheumatic Diseases Clinics of North America, 42(3), 419–434. https://doi.org/10.1016/j.rdc.2016.03.002
Gai. (2023). 7 risk factors for autoimmune disease. Global Autoimmune Institute. https://www.autoimmuneinstitute.org/articles/about-autoimmune/7-risk-factors-for-autoimmune-disease/
Government of Alberta. (2021). Blood draw for donation or treatment: Care Instructions. Myhealth.alberta.ca https://myhealth.alberta.ca/Health/aftercareinformation/pages/conditions.aspx?hwid=abs2863
Gussin, H. A., Ignat, G. P., Varga, J., & Teodorescu, M. (2001). Anti–topoisomerase I (Anti–Scl‐70) antibodies in patients with systemic lupus erythematosus. Arthritis & Rheumatism: Official Journal of the American College of Rheumatology, 44(2), 376-383. https://doi.org/10.1002/1529-0131(200102)44:2%3C376::aid-anr56%3E3.0.co;2-2
Im, K., Mareninov, S., Diaz, M. F. P., & Yong, W. H. (2019). An introduction to performing immunofluorescence staining. Methods in Molecular Biology, 299–311. https://doi.org/10.1007/978-1-4939-8935-5_26
Ingegnoli, F., Castelli, R., & Gualtierotti, R. (2013). Rheumatoid factors: Clinical applications. Disease Markers, 35, 727–734. https://doi.org/10.1155/2013/726598
John, A. M., S, H., Ha, J. H., & Loh, S. N. (2022). Engineering a fluorescent protein color switch using entropy-driven β-strand exchange. ACS Sensors, 7(1), 263-271. https://pubs.acs.org/doi/10.1021/acssensors.1c02239
Johns Hopkins Arthritis Center. (2019, March 27). RA pathophysiology https://www.hopkinsarthritis.org/arthritis-info/rheumatoid-arthritis/ra-pathophysiology-2/
Johns Hopkins Lupus Center. (2019, March 27). Lupus blood tests https://www.hopkinslupus.org/lupus-tests/lupus-blood-tests/
Konstantinov, K., & Rubin, R. H. (2017). The universe of ANA testing: A case for point-of-care ANA testing. Auto-Immunity Highlights, 8, 4. https://doi.org/10.1007/s13317-017-0093-6
Kongkaew, S., Rungrotmongkol, T., Punwong, C., Noguchi, H., Takeuchi, F., Kungwan, N., Wolschann, P., & Hannongbua, S. (2019). Interactions of HLA-DR and topoisomerase I epitope modulated genetic risk for systemic sclerosis. Scientific Reports, 9(1), 745. https://doi.org/10.1038/s41598-018-37038-z
Kumar, Y., Bhatia, A., & Minz, R. W. (2009). Antinuclear antibodies and their detection methods in diagnosis of connective tissue diseases: A journey revisited. Diagnostic Pathology, 4(1), 1. https://doi.org/10.1186/1746-1596-4-1
Liljeruhm, J., Funk, S., Tietscher, S., Edlund, A., Jamal, S., Wistrand-Yuen, P., Dyrhage, K., Gynna, A., Ivermark, K., Lovgren, J., Tronblom, V., Virtanen, A., Lundin, E., Wistrand-Yuen, E., & Forster, A. (2018). Engineering a palette of eukaryotic chromoproteins for bacterial synthetic biology. Journal of Biological Engineering, 10(12), 8. https://doi.org/10.1186/s13036-018-0100
Lupus Foundation of America (n.d). What is lupus?. https://www.lupus.org/resources/what-is-lupus
Lupus Foundation of America. (n.d.). Prognosis and life expectancy. https://www.lupus.org/resources/prognosis-and-life-expectancy
Martinez, B., Harker. M. D., Robinson, A., Peplow, V. P. (2020) False positive ANA testing in the setting of hypovitaminosis D. Journal of Clinical Studies & Medical Case Reports, 7(3), 1–5. http://dx.doi.org/10.24966/CSMC-8801/100098
Mayo Clinic. (2022, August 17). ANA test https://www.mayoclinic.org/tests-procedures/ana-test/about/pac-20385204
Mayo Clinic. (2022, December 24). Multiple sclerosis – Symptoms and causes. https://www.mayoclinic.org/diseases-conditions/multiple-sclerosis/symptoms-causes/syc-20350269
Mayo Clinic. (2022, October 21). Lupus – Diagnosis and treatment https://www.mayoclinic.org/diseases-conditions/lupus/diagnosis-treatment/drc-20365790
Mayo Clinic. (2022, October 21). Lupus – Symptoms and causes. https://www.mayoclinic.org/diseases-conditions/lupus/symptoms-causes/syc-20365789
Mayo Clinic. (2023, January 25). Rheumatoid arthritis – Symptoms and causes. https://www.mayoclinic.org/diseases-conditions/rheumatoid-arthritis/symptoms-causes/syc-20353648
Mayo Clinic. (2022, August 2). Sjogren’s syndrome – Symptoms and causes – https://www.mayoclinic.org/diseases-conditions/sjogrens-syndrome/symptoms-causes/syc-20353216
MedlinePlus. (n.d.) CCP Antibody Test. https://medlineplus.gov/lab-tests/ccp-antibody-test/
Meryhew, N. L., Messner, R. P., & Tan, E. M. (1983). Urinary excretion of antinuclear antibodies. The Journal of Rheumatology, 10(6), 913–919.
National Rheumatoid Arthritis Society (NRAS). (2021, April 15). How is lifespan affected by RA? https://nras.org.uk/resource/how-is-lifespan-affected-by-ra/
Proteintech (n.d.). Seizing the day: How to save time on your immunoassay experiments. https://www.ptglab.com/news/blog/seizing-the-day-how-to-save-time-on-your-immunoassay-experiments
Pruijn, G. J. M. (2015). Citrullination and carbamylation in the pathophysiology of rheumatoid arthritis. Frontiers in Immunology, 6. https://doi.org/10.3389/fimmu.2015.00192
Reikofski, J., & Tao, B. Y. (1992). Polymerase chain reaction (PCR) techniques for site-directed mutagenesis. Biotechnology Advances, 10(4), 535–547. https://doi.org/10.1016/0734-9750(92)91451-J
Rosano, G. L., & Ceccarelli, E. A. (2014). Recombinant protein expression in Escherichia coli: Advances and challenges. Frontiers in Microbiology, 5, 172. https://doi.org/10.3389/fmicb.2014.00172
Rubin, R. H., & Konstantinov, K. (2016). Biosensor for total antinuclear antibody determination at the point-of-care. Biosensors and Bioelectronics, 83(15), 306-311. https://doi.org/10.1016/j.bios.2016.04.048
Sullivan, M., McLean-Tooke, A., & Loh, KS. R. (2013). Antinuclear antibody test. Australian Family Physician, 42(10).
Synder, R. M. (2019) A basic guide to ANA testing. American Association for Clinical Chemistry (AACC). https://www.aacc.org/cln/articles/2019/april/a-basic-guide-to-antinuclear-antibody-ana-testing
Tetyana, P., Shumbula, P., & Njengele-Tetyana, Z. (2021). Biosensors: Design, development and applications. IntechOpen eBooks. https://doi.org/10.5772/intechopen.97576
Thavendiranathan, P., Bagai, A., Ebidia, A., Detsky, A. S., & Choudhry, N. K. (2005). Do blood tests cause anemia in hospitalized patients? Journal of General Internal Medicine, 20(6), 520–524. https://doi.org/10.1111/j.1525-1497.2005.0094.x
Thermo Fisher Scientific – U.S. (n.d.). Overview of affinity purification. Retrieved April 29, 2023, from https://www.thermofisher.com/ca/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-affinity-purification.html.
Website, N. (2022, June 28). Multiple sclerosis. NHS.UK. https://www.nhs.uk/conditions/multiple-sclerosis/ Xuan, R., Yang, A., Murell, F. D., Oakley, A. (2017). Indirect immunofluorescence. DermNet. https://dermnetnz.org/topics/indirect-immunofluorescence