Akarapu Saisiri, Burke Kensley, Chelnokov Sasha, Christopher Jenny, Lee Sean, Mehta Vedant, Murugula Rachna, Ramireddy Shashank, Verma Aarushi, and Yeleti Sankalp. ▫ Lambert High School, Suwanee, Georgia, United States; BioBuilderClub, Lambert High School iGEM Seminar Team, Suwanee, Georgia, United States
Reviewed on 6 May 2023; Accepted on 13 July 2023; Published on 16 October 2023
With help from the 2023 BioTreks Production Team.
Diamondback moths currently cost annual damages of four to five billion dollars to cruciferous plants worldwide. The main problem with current pest management methods is that moths develop resistance through natural mutations and limited exposure to insecticides. This article aims to propose a solution for increasing plant production by creating transgenic broccoli plants that express both crystalline (cry1Ac) and nutritive (vip3Aa) insecticidal proteins. The cry1Ac and vip3Aa insecticidal proteins kill pests upon consumption of the crops, effectively controlling diamondback moths and preventing any natural mutations through the secondary toxin. The modifications to the plants would be done via creating plasmids with both toxins obtained from the bacteria Bacillus thuringiensis. The recombinant DNA plasmid containing the genes-encoding cry1Ac and vip3Aa toxins would be inserted in the bacteria Agrobacterium tumefaciens. The Agrobacterium’s large cargo space and permanent integration of replicating DNA made it the optimal choice for our transgenic plants.
Keywords: Cruciferous plants, diamondback moths, biopesticide, Brassicaceae plants
Authors are listed in alphabetical order. Matarrese Caroline, Robichon-Iyer Carine, and Sharer Catherine from Lambert High School, Suwanee, Georgia, United States; BioBuilderClub, Lambert High School iGEM Seminar Team, Suwanee, Georgia, United States mentored the group. Please direct all correspondence to .
Cruciferous plants, vegetables of the Brassicaceae family, include a multitude of nutrient-rich commercial crops such as broccoli, mustard seed, arugula, and napa cabbage. Apart from the various vitamins and minerals contained in these plants such as vitamins A, C, and K, folic acid, which assists new cell growth, and chromium, an essential mineral that regulates blood sugar levels, cruciferous vegetables also contain phytonutrients called glucosinolates (Ellis, 2020). Glucosinolates, sulfurous chemicals that give cruciferous plants a bitter taste, act as antioxidants and combat inflammation associated with cancer by forming sulforaphane, an organic compound that serves to inhibit the expression of carcinogenic nitrosamines and polycyclic aromatic hydrocarbons, protecting cells from DNA damage (Cleveland Clinic, 2023; National Cancer Institute, 2012). Sulforaphane, easily found in many cruciferous vegetables, correlates to regulatory functions against lung and colorectal cancer (NCI, 2012).
Diamondback moths (Plutella xylostella) pose a global issue as they feed off cruciferous plants. The parasitic influence of diamondback moths has grown over the past few years with the rise in commercial production of Brassicaceae family plants (Furlong et al., 2013). Studies estimate that consumption of broccoli and similar Brassicaceae family plants has nearly tripled, and it is projected to rise even higher, thus cementing their importance as global commercial crops (De Innocentis, 2020). Unfortunately, the moth species cost the global agriculture industry billions of dollars in damages annually, resulting in losses in cruciferous plant production and putting pressure on farmers worldwide. As a result, more intensive forms of agricultural practices are adopted, which can lead to higher probabilities of soil depletion and less yields in the future. Thus, there is a strong need to develop technology for managing diamondback moths, such as biological control to prevent further loss of crops worldwide. Along with helping farmers around the world, addressing this issue could alleviate the burden of food insecurity by providing access to billions of dollars’ worth of food.
Current efforts taken to combat diamondback moths have been inadequate. Present solutions include the use of insecticides and integrated-pest management (IPM) with the use of a singular toxic protein (Furlong et al., 2013). However, both of these methods have significant flaws. First, insecticides can result in more harm than benefit. Chemical pesticides such as those used against diamondback moths have resulted in severe environmental harm and a plethora of health effects in humans, ranging from skin rashes to birth defects and various cancers (Aktar et al., 2009). Furthermore, insecticides have been found to damage nearby populations of different species, namely aquatic species such as fish, due to the run-off of chemicals into water sources (British Columbia Ministry of Agriculture, 2017).
Secondly, integrating toxic proteins into cruciferous plants has been explored as part of an IPM system. However, research has found that moths easily develop resistance to individual toxins and insecticides due to their quick life cycle and overuse of pesticide chemicals. As a result, the efforts made by genetic engineers to mitigate the problem are rendered ineffective (Hurst et al., 2019).
Our solution for creating a biopesticide puts forth a method for eliminating pests by integrating multiple toxic proteins into Brassicaceae plants to ensure those resistante to only one toxin is still neutralized. The bacteria B. thuringiensis creates crystalline (cry) proteins known as delta endotoxins; poisons present in organisms that are released when consumed. Cry endotoxins are viable for insect control due to their harmlessness to people and biodegradability (Bravo et al., 2007). Once consumed, these proteins form pores that release crystals that circulate the moth’s cell membranes and use binding proteins such as GPI-anchored alkaline phosphatases and glycoconjugates, resulting in cell lysis (Bravo et al., 2007).
B. thuringiensis also contains secretory proteins known as vegetative insecticidal proteins (Vip), widely used to control lepidopteran pests. Vip proteins are classified as inactive protoxins, as these require chemical altering before activating, which can be done by midgut proteases in the digestive gut of pests, killing moths with natural poisons after consumption of the crops (Melton & Tweten 2006; Nuñez-Ramírez et al., 2020). The two proteins that were found to be best suited for IPM against diamondback moths were the cry1Ac and vip3Aa proteins, as they are the most toxic and do not compete for binding sites (Lemes et al., 2017). Therefore, our design uses a plasmid that can incorporate multiple proteins and integrate into cruciferous plants.
Our team’s design for inserting the cry1Ac and vip3Aa protoxins from B. thuringiensis was based upon a plasmid system from A. tumefaciens. The Agrobacterium vector used is based on a Ti plasmid for transgenic plants, which has a large cargo space and permanent integration of T-DNA regions into host plant genomes, thus making it a good candidate for optimized replication inside of Agrobacterium and delivery of toxins. Additionally, we will incorporate an enhanced green fluorescence protein (eGFP) in order to visualize if our protoxin plasmids were incorporated into the plants.
The plasmid used is an Agrobacterium binary vector system based upon natural Ti (tumor-inducing) plasmids with two distinct parts (see Figure 1). First, the T-DNA (transfer DNA) vector (binary vector) region encloses the DNA sequence prepared for integration with the host plant’s DNA via gene cloning. The second portion of the system, known as the vir helper plasmid, codes the components for integration of the T-DNA region, creating the final product of the system. The two plasmids are assembled in A. tumefaciens and then make up the binary vector system that is used to insert the sequences into a Brassicaceae family plant (VectorBuilder, n.d.).
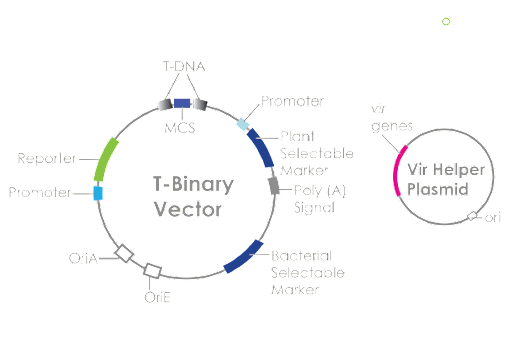
| Figure 1: Agrobacterium binary vector system |
|---|
As previously stated, our design relies on two separate toxins. These are the cry1Ac, cry/crystalline protein, and vip3Aa, vegetative insecticidal protein. As explained in the section about current problems with pest management, one of the biggest problems is moths having natural mutations to certain toxins. Due to their short lifespan and their speed at reproducing, these mutations can occur quickly. Moths that are impervious to a toxin are the only ones that can survive, they quickly take over the gene pool, leading to a population that is completely resistant to the insecticide and rendering the insecticides useless. Our project would aim to eradicate the moths before they develop complete genetic resistance. If a singular toxin is used, for example, cry1Ac, the moths would eventually gain resistance and reproduce, causing the insecticide to be ineffective. On the other hand, the use of two endotoxins, cry1Ac and vip3Aa, would eliminate any moth with a natural resistance to the first by the second. This would make any moths with a singular resistance unable to survive and therefore no longer be capable of taking over the gene pool.
Systems level
Our designed Ti-plasmid, which allows for the insertion of the two unique endotoxins, would be tested for success and compatibility by using Escherichia coli. To accomplish this, we would insert Ti-plasmid into a competent E. coli strain via a heat-shock transformation. After heating and cooling, the E. coli should repair their cell walls and be able to express the endotoxins. If the plasmid is successfully integrated, the expression of a fluorescent eGFP will allow the visualization of the T-DNA’s incorporation.
After proving the success of our endotoxins’ ability for integration, we will first transgenically modify an arugula (Eruca vesicaria) plant. Our team would complete the transgenic modification through floral dipping, a process that combines the transformed A. tumefaciens into the chosen cruciferous vegetable after cutting its flowers and dipping the plant into the culture, and letting incubation occur. After a successful incubation period, the plant will continue to undergo natural growth with the recombinant DNA incorporated inside.
In order to test our endotoxins, our team would experiment on diamondback moths to show how efficient the plants are against the pests. By comparing data on the populations of the transgenic arugula plants and the populations of the diamondback moths in a dry lab, the success rate of the biopesticide will be visualized. In order to prove successful replications of the recombinant DNA into generations of plants, collected data should show an increase in arugula plants which survive until germination, and a decrease in diamondbacks.
Device level
The binary vector Ti-plasmid system that will be inserted into the Brassicaceae family plants contains two toxin-coding genes – the delta endotoxin cry1Ac and secretory protoxin vip3Aa – as well as an eGFP gene that allows for visualization of the plasmid when it is expressed (see Figure 2). Our team chose the Ti-system for its ability to house both toxins and its previously reported high efficiency of permanent integration into plants, making it a great candidate for the transgenic cruciferous vegetables. Both toxins are derived from the bacteria B. thuringiensis and will be inserted into A. tumefaciens via heat shock due to its ease in entering plant genomes. The final Agrobacterium product will then be expressed in a cruciferous plant and used to kill diamondback moths with the properties of protoxins.
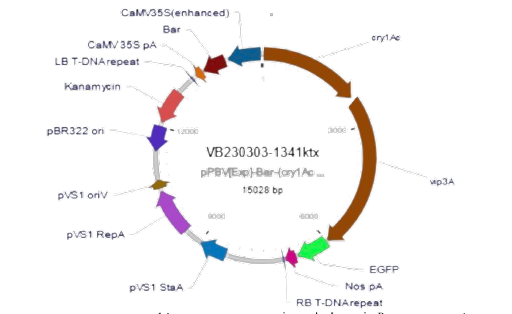
| Figure 2: Designed Biopesticide plasmid in T-DNA region of binary vector system. Note. Brown: Toxin genes, Green: eGFP gene |
|---|
Parts level
The cry1Ac gene, vip3Aa gene, and eGFP fluorescent gene will be assembled into the plant expression binary vector system taken from Ti-plasmids that use two different plasmids to incorporate all genes (see Figure 1). The Ti-plasmid, with its large area available for genes of interest (T-DNA) as well as ease of integration into plants via the second plasmid in the system (vir helper), is the optimal choice for creating transgenic plants with recombinant DNA. The integration works by inducing tumors in the plant and forcing itself into permanent integration of the plant after floral dipping (VectorBuilder, n.d.). The tumor-inducing nature of A. tumefaciens and its ability to insert its DNA through crown gall disease contributes to its high-quality gene expression. Crown gall disease is a condition caused by the Agrobacterium, also known as Rhizobium radiobacter, where the bacterium infects plants through their roots, the same pathways that floral dipping will target, and the bacteria’s DNA combines with the DNA of the Brassicaceae family to transform the plant cells (The Royal Horticultural Society, n.d.). Additionally, A. tumefaciens is favorable to use due to its ability to survive in the soil for long periods of time, which may occur if the floral dipping fails, allowing the Agrobacterium to be able to re-enter the plant if it failed.
The two endotoxins cry1Ac and vip3Aa are both protoxins that activate once ingested by the diamondback moths (Melton & Tweten, 2006). Once ingested, the two endotoxins work inside the organism through the first step of solubilization. Solubilization increases the solubility of a substance through a high number of surfactants, substances which lower the surface tension of a targeted liquid, thus allowing for more ingested cruciferous plants to become dissolved into the toxin plants (Intertek, n.d.; Lau, 2001; see Figure 3).
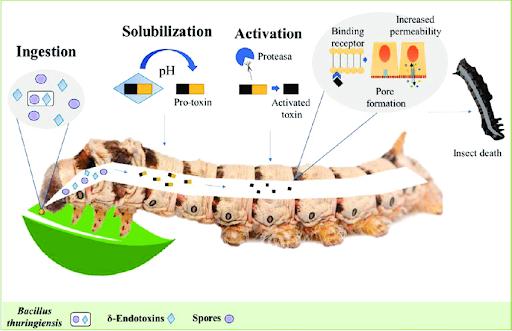
| Figure 3. Activation Pathway of Protoxins. Note. Light blue & purple: cruciferous plant cells. |
|---|
The pH of the moths’ digestive systems solubilizes protoxins by changing the structure exposing the proteins for activation, the second step of the process, in the gut. Activation occurs when proteases, enzymes that amplify molecular signals through transduction and phosphorylation cascades, bind with the exposed proteins and activate them, allowing for the toxins to have the capacity for binding to the insect’s cells (López-Otin & Bond, 2008). The final step of the process occurs when the now active endotoxins bind to the walls of the moths’ gastrointestinal tract and cause increased permeability. This increase in the vascular permeability of the moth cells allows for important molecules such as water to be lost and harmful pathogens to enter more easily, thus progressing disease and infection in insects and causing death (Claesson-Welsh, 2015).
In order to track success or failure of our binary vector system, we additionally implemented an eGFP promoter. eGFP works by binding to specific proteins or cells and expressing genes for color that will appear under a fluorescent microscope. eGFP is chosen by many biotechnologists due to the protein’s ability to allow for insight into the smallest parts of cells, such as the plasmids that our team is using for our recombinant DNA (Goodsell, 2003). Furthermore, eGFP holds additional benefits when compared to regular GFP, as eGFP is better manufactured in order to tag very specific cells for viewing and can create images 35 times brighter than regular GFP (Samanthi, 2018).
Safety
The insecticidal proteins we chose, cry1Ac and vip3Aa, work by inhibiting the digestion of pests and are derived from the bacteria B. thuringiensis. Thus, safety concerns pertaining to human consumption may arise from the usage of biochemical insecticides. Luckily, the safety of chemicals in B. thuringiensis can be proven, as in 2010, there were over 58 million hectares of vegetation grown with toxins from the bacteria B. thuringiensis, and they have been proven to be entirely safe for human consumption with no adverse side effects unless the consumer had a condition that could be easily worsened by the bacteria (James, 2011). Furthermore, safety concerns arise from a failed integration and unintended side effects which may bring harm to humans. We are able to prevent this issue by using our eGFP promoter, which will prove whether or not the plasmid has been integrated, showing if the integration of our Ti-system successfully created the desired transgenic Brassicaceae plants.
Another concern that may arise is the resistance of pests to the chosen pesticidal proteins. As mentioned earlier, our team was able to combat this issue via our unique use of two different proteins that would eliminate any moths in the gene pool with resistance to one protein or the other. Additionally, even moths that potentially develop mutations to resist both will not consume as many cruciferous vegetables as their behavior has been altered to perceive the crops as toxic and nutrition that should be avoided.
Discussions
Overall, our project serves to kill the pests in order to boost the agricultural production of cruciferous vegetables. It also acts as a major steppingstone that would resolve the biggest problem most insecticides face today. Natural mutations are something that happens in pests, regardless of chemicals and the diets of the organisms, and by highlighting one way that these mutations can be stopped, projects such as ours can become more globally applicable and eventually lead to more pest-specific targeting. Furthermore, eliminating diamondback moths will allow for more capital to be produced, furthering funding that may be used for research against other harmful insects such as locusts or aphids.
While our project has a plethora of benefits, one negative impact would be that current fields of crops would have to be cleared, as our project could only be put into practice if all the plants in an area possess the binary vector system. Thus, farmers currently growing Brassicaceae plants such as broccoli would either need to sell off their crops or discard them. This process will initially cost farmers large sums of money, deterring some individuals from implementing the practice.
Another obstacle in our project is the widespread uncertainty of data that may be collected. Various variables may contribute to different outcomes in an open agricultural field, leading to data that supports the benefits and rationale of our research or contribute to other results impacted by confounding variables. For example, a decrease in cruciferous plants that are consumed by diamondback moths may occur not because of the two endotoxins but rather the outspread of a disease which targets diamondback moths, skewing the data that we collected.
Next steps
Once our Ti-system’s success is proven in plants with fast reproduction, such as arugula, the application of our multi-protein biopesticide will be used for more widespread commercial Brassicaceae crops like broccoli or napa cabbage. We would also test the project in the real-world industry, where many factors are constantly changing, by planting our transgenic plants in a farm setting and monitoring the activity of the moths with the aforementioned graphs. Finally, we would reach out to local professors and researchers in the agricultural genetic modification industry to further examine the project’s feasibility at a national or even global scale.
Author contributions
S.L. is the main author for this project. He researched the necessary connections between genetic components to create a concise understanding of the topic. S.Y. is the creator of the project’s initial proposal, setting forth all of the ideas written. V.M. is responsible for the design of our plasmid using modeling programs to allow for a visualization of the system.
Acknowledgements
We would like to thank Catherine Sharer and Caroline Matarresse for their continuous support and weekly feedback, as well as Carine Robichon-Iyer for providing relevant scientific articles and recommending different research.
Our project was made in conjunction with the BioBuilder synthetic biology research program. The program spans 47 states and 55 countries, spreading research techniques and other essential life skills over a 7–8-month period.
References
Aktar, M. W., Sengupta, D., & Chowdhury, A. (2009). Impact of pesticides use in agriculture: Their benefits and hazards. Interdisciplinary Toxicology, 2(1), 1. https://doi.org/10.2478/v10102-009-0001-7
Latté, K. P., Appel, K. E., & Lampen, A. (2011). Health benefits and possible risks of broccoli–An overview. Food and Chemical Toxicology, 49(12), 3287-3309. https://doi.org/10.1016/j.fct.2011.08.019
Liu, Y., & Tabashnik, B. E. (1997). Inheritance of resistance to the Bacillus thuringiensis toxin Cry1C in the diamondback moth. Applied and Environmental Microbiology, 63(6), 2218-2223. https://doi.org/10.1128/aem.63.6.2218-2223.1997
Bischoff, K. L. (2021). Glucosinolates. In R. C. Gupta, R. Lall, & A. Srivastava (Eds.), Nutraceuticals: Efficacy, safety and toxicity (pp. 903-909). Academic Press. https://doi.org/10.1016/B978-0-12-821038-3.00053-7
Bravo, A., Gill, S. S., & Soberón, M. (2007). Mode of action of Bacillus thuringiensis Cry and Cyt toxins and their potential for insect control. Toxicon, 49(4), 423-435. https://doi.org/10.1016/j.toxicon.2006.11.022
Bravo, A., Likitvivatanavong, S., Gill, S. S., & Soberón, M. (2011). Bacillus thuringiensis: A story of a successful bioinsecticide. Insect Biochemistry and Molecular Biology, 41(7), 423-431. https://doi.org/10.1016/j.ibmb.2011.02.006
British Columbia Ministry of Agriculture. (2017). Environmental protection and pesticides. https://www2.gov.bc.ca/assets/gov/farming-natural-resources-and-industry/agriculture-and-seafood/animal-and-crops/plant-health/environmental-protection-and-pesticides.pdf
Cell Guidance Systems. (2021, December 20). Green fluorescent protein: GFP legend and legacy. https://www.cellgs.com/blog/green-fluorescent-protein-gfp-legend-and-legacy.html
Claesson-Welsh, L. (2015). Vascular permeability—The essentials. Upsala Journal of Medical Sciences, 120(3), 135-143. https://doi.org/10.3109/03009734.2015.1064501
Cleveland Clinic. (2023, June 7). What are cruciferous vegetables – And why are they so good for you?. https://health.clevelandclinic.org/crunchy-and-cruciferous-youll-love-this-special-family-of-veggies/
De Innocentis, I. (2020, November 13). The history of broccoli: From the Etruscan period to now. La Cucina Italiana. https://www.lacucinaitaliana.com/trends/healthy-food/history-broccoli-etruscans-today?refresh_ce=
Ellis, E. (2020, August 13). The beginners guide to cruciferous vegetables. Eat Right: Academy of Nutrition and Dietetics. https://www.eatright.org/food/food-groups/vegetables/the-beginners-guide-to-cruciferous-vegetables
Furlong, M. J., Wright, D. J., & Dosdall, L. M. (2013). Diamondback moth ecology and management: Problems, progress, and prospects. Annual Review of Entomology, 58, 517-541. https://doi.org/10.1146/annurev-ento-120811-153605
Puig Garcia, E., Gonzalez, F., Hamilton, G., & Grundy, P. (2015). Assessment of crop insect damage using unmanned aerial systems: A machine learning approach. In T. Weber, M. J. McPhee, & R.S. Anderssen (Eds.), Proceedings of MODSIM2015, 21st International Congress on Modelling and Simulation (pp. 1420-1426). Modelling and Simulation Society of Australia and New Zealand. https://doi.org/10.36334/MODSIM.2015.F12.puig
Gouffon, C., Van Vliet, A., Van Rie, J., Jansens, S., & Jurat-Fuentes, J. L. (2011). Binding sites for Bacillus thuringiensis Cry2Ae toxin on heliothine brush border membrane vesicles are not shared with Cry1A, Cry1F, or Vip3A toxin. Applied and Environmental Microbiology, 77(10), 3182–3188. https://doi.org/10.1128/AEM.02791-10
Intertek. (n.d.). Surface active agents and surfactants analysis. https://www.intertek.com/chemicals/surface-active-agent-surfactant/
James, C. (2011). Brief 43: Global status of commercialized biotech/GM crops: 2011. The International Service for the Acquisition of Agri-biotech Applications (ISAAA).
Lau, E. (2001). Preformulation studies. In S. Ahuja, & S. Scypinski (Eds.), Separation science and technology: Handbook of modern pharmaceutical Aanalysis (pp.173 – 233). Elsevier. https://doi.org/10.1016/S0149-6395(01)80007-6
Lemes, A. R. N., Figueiredo, C. S., Sebastião, I., da Silva, L. M., da Costa Alves, R., de Siqueira, H. Á. A., Lemos, M. V. F., Fernandes, O. A., & Desidério, J. A. (2017). Cry1Ac and Vip3Aa proteins from Bacillus thuringiensis targeting Cry toxin resistance in Diatraea flavipennella and Elasmopalpus lignosellus from sugarcane. PeerJ, 5, e2866. https://doi.org/10.7717/peerj.2866
Lin, J., Yu, X. Q., Wang, Q., Tao, X., Li, J., Zhang, S., X. Xia, & You, M. (2020). Immune responses to Bacillus thuringiensis in the midgut of the diamondback moth, Plutella xylostella. Developmental & Comparative Immunology, 107, 103661. https://doi.org/10.1016/j.dci.2020.103661
López-Otín, C., & Bond, J. S. (2008). Proteases: Multifunctional enzymes in life and disease. Journal of Biological Chemistry, 283(45), 30433-30437. https://doi.org/10.1074/jbc.R800035200
Melton, J., & Tweten, R. K. (2006). Clostridium septicum pore-forming α-toxin. In J. E. Alouf & M. R. Popoff (Eds.), The comprehensive sourcebook of bacterial protein (pp. 623-630). Academic Press. https://doi.org/10.1016/B978-012088445-2/50039-1
Scallan, E. (2017). Food source information: A food production wiki for public health professional. Proceedings of INFORMS.
National Cancer Institute. (2012, June 7). Cruciferous vegetables and cancer prevention. https://www.cancer.gov/about-cancer/causes-prevention/risk/diet/cruciferous-vegetables-fact-sheet
Navya, R., Balasubramani, V., Raveendran, M., Murugan, M., & Lakshmanan, A. (2022). Characterization of indigenous Bacillus thuringiensis isolate RM11 toxic to the diamondback moth, Plutella xylostella (L.) (Lepidoptera: Plutellidae). Egyptian Journal of Biological Pest Control, 32(1), 1-10. https://doi.org/10.1186/s41938-022-00553-3
George, Z., & Crickmore, N. (2012). Bacillus thuringiensis applications in agriculture. In E. Sansinenea (Ed.), Bacillus thuringiensis biotechnology (pp.19-39). Springer. https://doi.org/10.1007/978-94-007-3021-2_2
Núñez-Ramírez, R., Huesa, J., Bel, Y., Ferré, J., Casino, P., & Arias-Palomo, E. (2020). Molecular architecture and activation of the insecticidal protein Vip3Aa from Bacillus thuringiensis. Nature Communications, 11(1), 3974. https://doi.org/10.1038/s41467-020-17758-5
Goodsell, D. (2003, June). Molecule of the month: Green fluorescent protein (GFP). RCSB PDB. https://pdb101.rcsb.org/motm/42
Hurst, M. R. H., Jones, S. A., Beattie, A., van Koten, C., Shelton, A. M., Collins, H. L., & Brownbridge, M. (2019). Assessment of Yersinia entomophaga as a control agent of the diamondback moth Plutella xylostella. Journal of Invertebrate Pathology, 162, 19-25. https://doi.org/10.1016/j.jip.2019.02.002
The Royal Horticultural Society. (n.d.). Crown gall. https://www.rhs.org.uk/disease/crown-gall
Samanthi. (2018, October 15). Difference between GFP and EGFP. Difference Between. https://www.differencebetween.com/difference-between-gfp-and-egfp/
Srinivasan, R. (2012). Integrating biopesticides in pest management strategies for tropical vegetable production. Journal of Biopesticides, 5, 36-45.
Sun, C. N., Wu, T. K., Chen, J. S., & Lee, W. T. (1986). Insecticide resistance in diamondback moth. Diamondback Moth Management: Asian Vegetable Research and Development Center, 359. VectorBuilder. (n.d.). Plant gene expression Agrobacterium binary vector. https://en.vectorbuilder.com/resources/vector-system/pPBV_Exp.html

