Cavanaugh Brett, Collins Thomas, Collins Yesenia, Fugazzotto Emily, Meyyappan Sudar, and Pelekoudas Themistocles▫ Tyngsborough High School, Tyngsborough, Massachusetts, United States
Reviewed on 6 May 2023; Accepted on 13 July 2023; Published on 16 October 2023
With help from the 2023 BioTreks Production Team.
Mosquitoes are vectors of disease. Almost 725,000 people die each year due to mosquito-borne illnesses, making them one of the deadliest animals on the planet. At this time, half of the world’s population is at risk of contracting malaria. To combat this worldwide issue, a biologically generated repellent that deters mosquitoes called AliveSCENT was proposed. Potential mosquito repellent scents, including lavender, limonene, wintergreen, and jasmine, were tested. Lavender oil was shown to be effective at preventing sugar feeding of laboratory-raised mosquitoes. AliveSCENT exploits the native methylerythritol 4-phosphate (MEP) pathway of Escherichia coli in the production of linalool, a key compound in the lavender scent. First, the potential mosquito repellent scent of lavender was tested by placing various concentrations of lavender oil on cotton balls containing a 10% sucrose solution. The number of mosquito feedings after the addition of lavender oil was then recorded. Lavender oil was shown to be effective at reducing the number of mosquito sugar feedings. Next, a plasmid was designed to exploit the native MEP pathway of E. coli to produce linalool, a key compound in the lavender scent. Successfully transformed colonies were evaluated for the presence of lavender scent using a wafting test, and a small discernible difference was observed between the conditions.
Keywords: Mosquito, repellent, Aedes albopictus, mep pathway, Escherichia coli, lavender
Authors are listed in alphabetical order. Michael Sheets and Rebekah Ravgiala from Tyngsborough High School, Tyngsborough, Massachusetts, United States mentored the group. Please direct all correspondence to rebekah.ravgiala@tyngsboroughps.org; michael.sheets4@gmail.com.
One of the most common insects on the planet is also one of its most dangerous: the mosquito. Mosquitoes are responsible for endangering half of the world’s population with malaria (WHO, 2023a). Malaria is a mosquito-borne disease that can cause symptoms including an increase in heart rate, fever, diarrhea, fatigue, chills, and joint pain (Mayo Clinic, 2023). In recent years, there have been just under a quarter billion cases of malaria annually (WHO, 2023a). Malaria, along with other diseases, can be transmitted through a female mosquito’s bite. Female mosquitoes are the ones that search for blood to use its proteins to lay eggs (Centers for Disease Control and Prevention, 2020). When someone is bitten, the proboscis, an elongated mouth part of the mosquito, penetrates the skin, leaving saliva behind. If the mosquito is carrying a disease, it is transmitted through the saliva and enters the bloodstream. Since there is a disease in the blood, it can also be transmitted through interaction with contaminated blood. Hence, when a mosquito bites an infected person, it carries the infection with it, possibly spreading that infection to any person it bites thereafter (WHO, 2023a). Finding an effective repellent could reduce the number of fatalities attributed to malaria. Another notable mosquito-borne illness is dengue fever, estimated to result in 390 million infections each year; it can be spread by mosquitoes in the Aedes genus, which includes A. albopictus (WHO, 2023b). Currently, the best way to stop the spread of these diseases is to use netting, screens, coils, and other forms of chemical and physical repellents (WHO, 2023a). A deterrent that does not require the proper use of lotions or nets (a difficulty for children) or the use of fire, in the case of candles and citronella torches, would decrease these hazards. AliveSCENT is a solution to this persistent problem by using an alternative biological repellent as a mosquito deterrent. In this BioBuilder Club season, a plasmid with a linalool coding sequence and a red fluorescent protein marker for the production of lavender scent was designed, built, and tested. Lavender has been reported to be an effective mosquito repellent through tests that assessed various essential oils (Collins et al., 2022). It was also reported that lavender scent can be generated from the native methylerythritol 4-phosphate (MEP) metabolic pathway of E. coli (Collins et al., 2022). The minimal concentration of lavender scent necessary to elicit a response from mosquitoes was tested. The AliveSCENT alternative would contribute to a reduction in the spread of disease and could save countless lives.
Materials and methods
Raising mosquitoes
A colony of A. albopictus mosquitoes was raised for the purpose of this project. The mosquitoes were raised following the protocol described by Aldulaimy et al. (2021), including the established procedures for care and feeding. Mosquitoes begin their life as eggs submerged in water. After approximately five days, eggs will hatch into larvae. Larvae mature into pupae after as little as five days and begin their adult life within two to three days (Figure 1). Taking care of an actively developing mosquito colony involved daily feeding of larvae and monitoring the transformation of larvae into adult mosquitoes. Once matured, adult mosquitoes were transferred to a dedicated bug dorm (Figure 2) by aspiration. Bug dorms were equipped with a cotton ball saturated with a 10% sucrose solution upon which adult mosquitoes could feed. A net attached to the bug dorm entrance was secured in a tight knot to prevent mosquitoes from escaping. The process of transferring the maturing mosquitoes and feeding them was repeated daily for the duration of the experimentation.
| Figure 1. A general depiction of the mosquito life cycle from eggs to adults. |
|---|
Testing mosquitoes
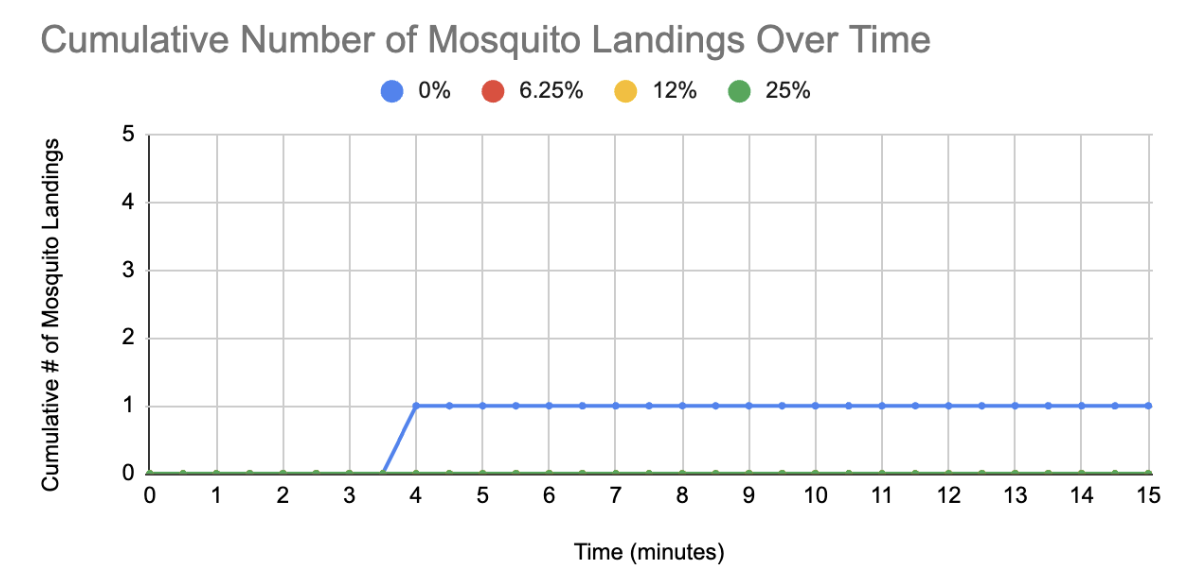
On the day of experimentation, 25 female mosquitoes were aspirated and transferred into a second bug dorm (Figure 2). Female mosquitoes were easily distinguishable from males without the use of a microscope because females are larger and have thinner antennae (Figure 3). Mosquitoes were starved 24 hours prior to experimentation to increase the likelihood that the hungry mosquitoes would land on a sucrose/lavender-treated cotton ball. If starved mosquitoes do not land on the cotton ball, it could be inferred that they were repulsed by the lavender present. Lavender was tested at concentrations of 0% (negative control), 6.25%, 12.5%, and 25% (see Table 1). A cotton ball was treated with 10 mL of each lavender concentration and placed into the testing chamber containing the 25 female mosquitoes. A camera was used in place of direct observations made by team members to avoid interference with native mosquito behavior. A timer was in view for the 15-minute testing duration so an accurate count of mosquito landings and the associated time could be recorded. The team rewatched the video and recorded how many mosquitoes landed on the cotton ball every 30 seconds for 15 minutes. The mosquitoes were then given a rest period of 10 minutes before the next lavender concentration was tested.
Table 1. Essential oil solution preparation to a final volume of 10 mL, which completely saturated the cotton ball. Each concentration contained a 10% sucrose solution to get mosquitoes to land on the cotton ball when lavender was added. If the mosquitoes did not land on the cotton ball, it demonstrates they were repelled by lavender even with the presence of the sucrose solution.
| 25% Lavender | 12.5% Lavender | 6.25% Lavender | 0% (control) | |
|---|---|---|---|---|
| 10% Sucrose | 5 mL | 5 mL | 5 mL | 10 mL |
| Lavender | 1.25 mL | 0.625 mL | 0.3125 mL | — |
| Distilled water | 3.75mL | 4.375 mL | 4.6875 mL | — |
Bacterial transformation
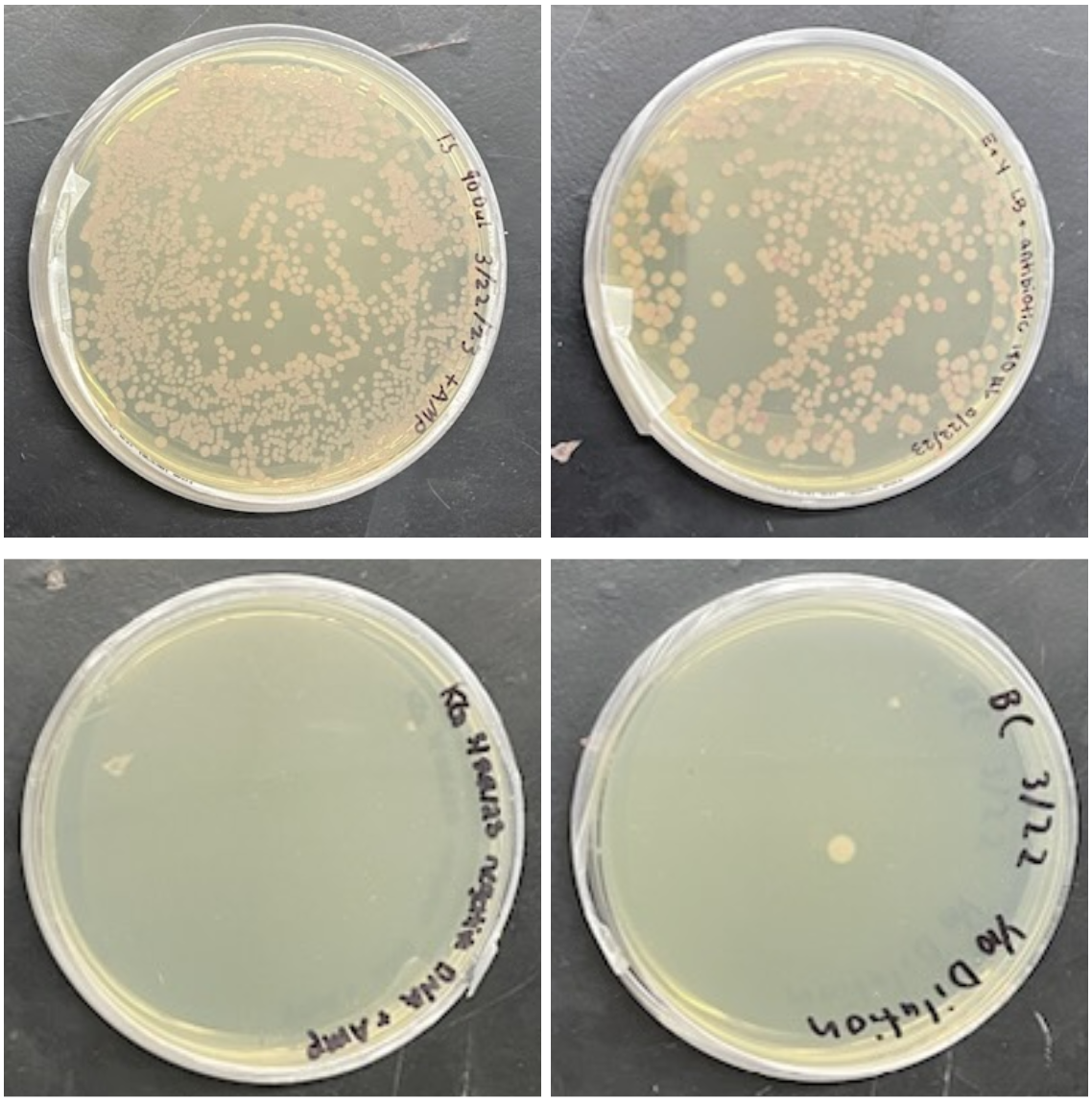
Plasmid DNA was delivered on ice from VectorBuilder and stored in a – 80°C freezer until use. A high-efficiency transformation protocol from New England Biolabs using C3013I E. coli was utilized in the transformation. The cells were thawed, and 50 µL were transferred into 4 Eppendorf tubes labeled 0 ng, 18 ng, 180 ng, and 900 ng. To the tube labeled 0 ng, an additional 1 µL of sterile water was added. To the tube labeled 180 ng, 1 µL of the 180 ng/µL stock DNA plasmid was added. To the tube labeled 900 ng, 5 µL of the 180 ng/µL stock DNA was added. A 1:10 dilution of the 180 ng/µL stock DNA was prepared (1 µL of DNA mixed with 9 µL of sterile water). To the tube labeled 18 ng, 1 µL of this 1:10 dilution was added. All tubes were inverted to mix. The mixtures were placed on ice for 30 minutes, heat shocked at 42°C for 10 seconds, and placed back on the ice for another 10 minutes. 950 µL of room temperature SOC medium was transferred to each tube and subsequently incubated on a roller at 37°C for an hour. After incubation, 180 µL of SOC was combined with 20 µL of each mixture. For each mixture, a 50 µL sample was then spread on an LB/amp/chl agar plate. The plates were then incubated for 72 hours at room temperature.
IPTG induction and scent tests
Liquid overnight cultures were grown using colonies picked from the 180 ng plate at 37°C and ambient temperature, followed by IPTG induction.
Four 50 mL conical tubes were filled with 15 mL of LB/amp/chl broth. The broth was prepared to 100 µg/mL and 25 µg/mL ampicillin and chloramphenicol final concentrations, respectively. The tubes were labeled ambient (-) IPTG, ambient (+) IPTG, 37°C (-) IPTG, and 37°C (+) IPTG. The tubes were flicked to mix. A colony from the 180 µL plate was picked with a sterile tip and ejected into each broth. Samples were vortexed, and the caps were loosened. Culture tubes were incubated overnight at their designated temperatures. The following day, the OD600 was measured in each tube to confirm that cultures were growing at a similar rate. 7mL of culture was transferred to labeled culture tubes. 70 µL of 100mM IPTG was added to the ambient (+) IPTG and the 37°C (+) IPTG tubes. 7 hours later, the OD600 was re-measured. Each E. coli culture was evaluated for the presence of a spicy or floral scent in a blind smell test. Seven volunteers were gathered to smell by wafting each of the cultures and rated the intensity of the lavender scent on a scale of 0 (least like lavender) to 5 (most like lavender). Between smelling each culture, each participant smelled a scented cleanser (ground coffee beans).
Safety protocols
Cell cultures
Throughout all investigations, proper safety precautions were taken. The cultures and chemicals used in the bacterial transformation were BSL1, and unlikely to cause disease. Team members were outfitted with proper personal protective equipment, including lab-grade safety glasses, lab coats, and nitrile gloves. Contaminated materials were disposed of according to required environmental health and safety standards.
Mosquitoes
Mosquitoes were hatched and raised in an isolated environment. No risk of disease transmission was possible because the mosquitoes were raised from eggs and did not come into contact with other disease-carrying animals. Mosquitoes used during experimentation were humanely cared for and were routinely fed a 10% sucrose solution. At the conclusion of experimentation, mosquitoes for experimentation were euthanized by freezer.
Results
Mosquito experiment
Lavender was tested at 0% (negative control), 6.25%, 12.5%, and 25% concentrations. Each concentration was placed on a cotton ball and inserted into a bug dorm containing 25 female mosquitoes to test its repellent effectiveness. Every 30 seconds for 15 minutes, the number of mosquitoes that landed on the cotton ball was recorded. When the 0% lavender concentration was tested, one mosquito landed on the cotton ball. However, for all other concentrations (6.25%, 12.5%, and 25%), no mosquitoes landed on the cotton ball (Figure 4). This, along with preliminary experimentation in previous years (Aldulaimy et al., 2021; Collins et al., 2022), concluded that lavender is a likely candidate. These results also demonstrate that the amount of lavender scent required to repel mosquitoes is small and could realistically be produced by E. coli.
Bacterial transformation
A bacterial transformation was performed as described in the materials and methods section. The AliveSCENT plasmid designed in the previous publication (Collins et al., 2022) was modified, built, and then transformed into E. coli. Modifications included changing the backbone of the plasmid from the Addgene pBbE5a-RFP plasmid to the pET bacterial recombinant protein vector from VectorBuilder. An RFP gene was added to the vector since it did not contain one. The results of the transformation were positive.
No colonies were observed on the negative control. One colony was observed on the petri dish with the 1/10 dilution (18 ng/µL). The remaining plates each showed tremendous growth and red fluorescence. A total of 291 colonies were observed on the 180 ng/µL plate, while 840 colonies were observed on the 900 ng/µL plate (Figure 5). Colony counts were compared to the concentration of DNA used. The graph below shows a direct relationship between increasing DNA concentration and the number of colonies (Figure 6). Note. In the Petri dish with the 18ng/ µL dilution, there was one colony present. There were zero colonies for the Petri dish that was negative for the plasmid. In the 180 ng/µL and 900 ng/µL Petri dishes, 291 and 840 colonies were present, respectively.
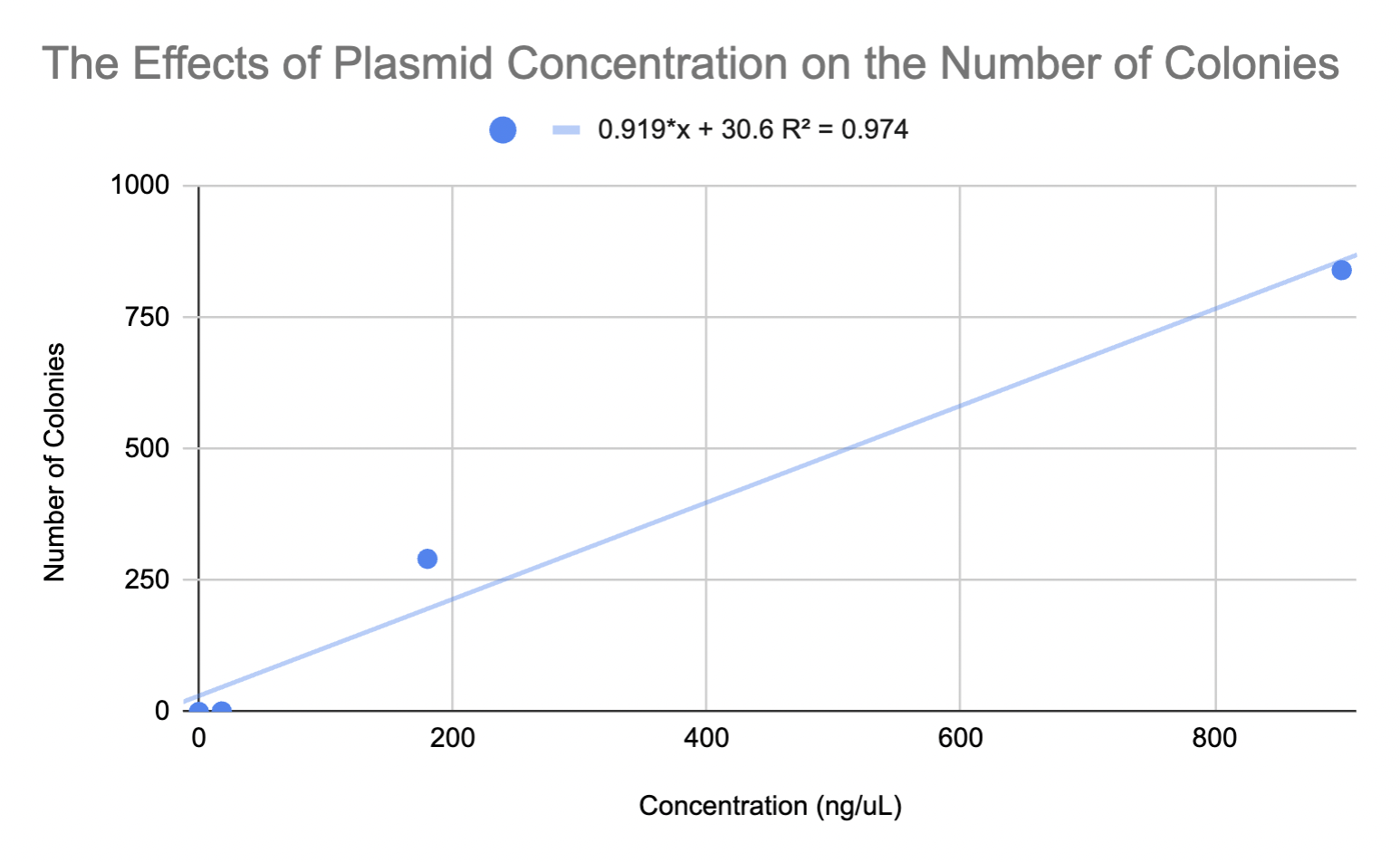
| Figure 6. Depicts the average number of colonies that were observed at varying concentrations based on the team’s four tested dilutions. |
|---|
IPTG induction and scent tests
A blind smell test was performed on liquid overnight cultures to determine whether there was a detectable difference between cultures that underwent IPTG induction and those that did not. The optical density at 600 nm was measured before and after a seven-hour incubation period to measure the growth of the cultures (Table 2). The optical density increased over time, proving that growth occurred evenly among all cultures.
| Time (hours) | Ambient (-) | Ambient (+) | 37°C (-) | 37°C (+) |
|---|---|---|---|---|
| 0 | 38.8 | 37.9 | 41.1 | 39.9 |
| 7 | 50.0 | 51.1 | 59.7 | 50.7 |
The smell test revealed a small discernible scent difference between the cultures induced with IPTG and those without (Table 3). Although volunteers smelled a different scent in the ambient IPTG induced cultures, it was neither spicy nor floral, as the scent of lavender has been described. Anecdotally, one volunteer said that the control culture tubes smelled better than the experimental cultures for both ambient and 37°C. It should be noted that the smell could have been more distinct had it not been overpowered by the natural odor of E. coli in all samples.
| Volunteer | Ambient (-) | 37°C (-) | Ambient (+) | 37°C (+) |
|---|---|---|---|---|
| 1 | 2 | 2 | 0 | 3 |
| 2 | 0 | 1 | 0 | 2 |
| 3 | 0 | 0 | 0 | 1 |
| 4 | 0 | 0 | 0 | 1 |
| 5 | 0 | 0 | 0 | 1 |
| 6 | 0 | 0 | 0 | 1 |
| 7 | 0 | 0 | 0 | 0 |
Note. Two of the cultures were ambient, meaning they were both incubated at 37°C and at room temperature. Contrarily, the other two cultures were only incubated at 37°C. Positive cultures were induced in IPTG, while negative cultures do not.
Note. Seven volunteers were used in the experiment to smell and report the strength of lavender on a scale from 0 to 5. If volunteers reported a zero, they did not smell lavender in the culture. If the volunteers reported a five, they smelled a significant amount of lavender in the culture.
Discussions
These experiments aimed to determine the effectiveness of lavender scent as a mosquito repellent and produce this scent in E. coli. A method found to achieve this goal would be used to construct AliveSCENT: a biologically inspired mosquito deterrent. Generating a mosquito repellent by exploiting bacteria could provide another solution to mosquito-borne illne sses, a worldwide health issue.
The objective of this project was to use the native MEP pathway to convert glucose into (s)-linalool, a compound found in lavender essential oil (see Figure 5). This paper documents the progress made on AliveSCENT over the last four years. Previous experiments were conducted to determine whether lavender scent is a suitable candidate for mosquito deterrence. To further test this conclusion, lavender was tested for its effectiveness at minimum concentrations. A previous study conducted by the team tested the repellent properties of lavender essential oil at concentrations of 100%, 50%, and 25%. While planning for linalool to be produced in E. coli, the team did not know how strong the scent would be. Thus, an experiment was conducted to test the efficacy of the scent at concentrations of 25%, 12.5%, and 6.25%.
It should be noted that while the population size was small for this experiment, the results did show that the mosquitoes were deterred from their food source once it was treated with lavender essential oil at any of the aforementioned concentrations. We remain optimistic that these results indicate the validity of linalool as a mosquito repellent. However, we recognize that future research should be conducted using larger sample sizes and more trials. In future experiments, a better control could be 5 mL of the 10% sugar solution mixed with 5 mL of distilled water. In this way, every cotton ball would have the same amount of sugar solution. There is a possibility that the presence of twice as much sugar solution in the control could have influenced the results. The team redesigned their previous plasmid using the platform VectorBuilder. They added geraniol diphosphate synthase, linalool synthase, and a red fluorescent protein part to the pET bacterial recombinant protein vector (Figure 7). The team then synthesized this plasmid and inserted it into E. coli. After the bacteria plates were incubated at room temperature, colonies with a distinct red hue were observed. This indicated that the transformation had been successful, as the bacteria were expressing a red fluorescent protein. Additionally, a small smell test was conducted using the transformed bacteria. The results were inconclusive. It was found that there was a small discernible scent difference between bacteria that had taken up the plasmid and those that had not, but it could not be determined by everyone that this difference was due to the production of linalool. The natural odor of E. coli was mostly overpowering, making it difficult to differentiate between the samples.
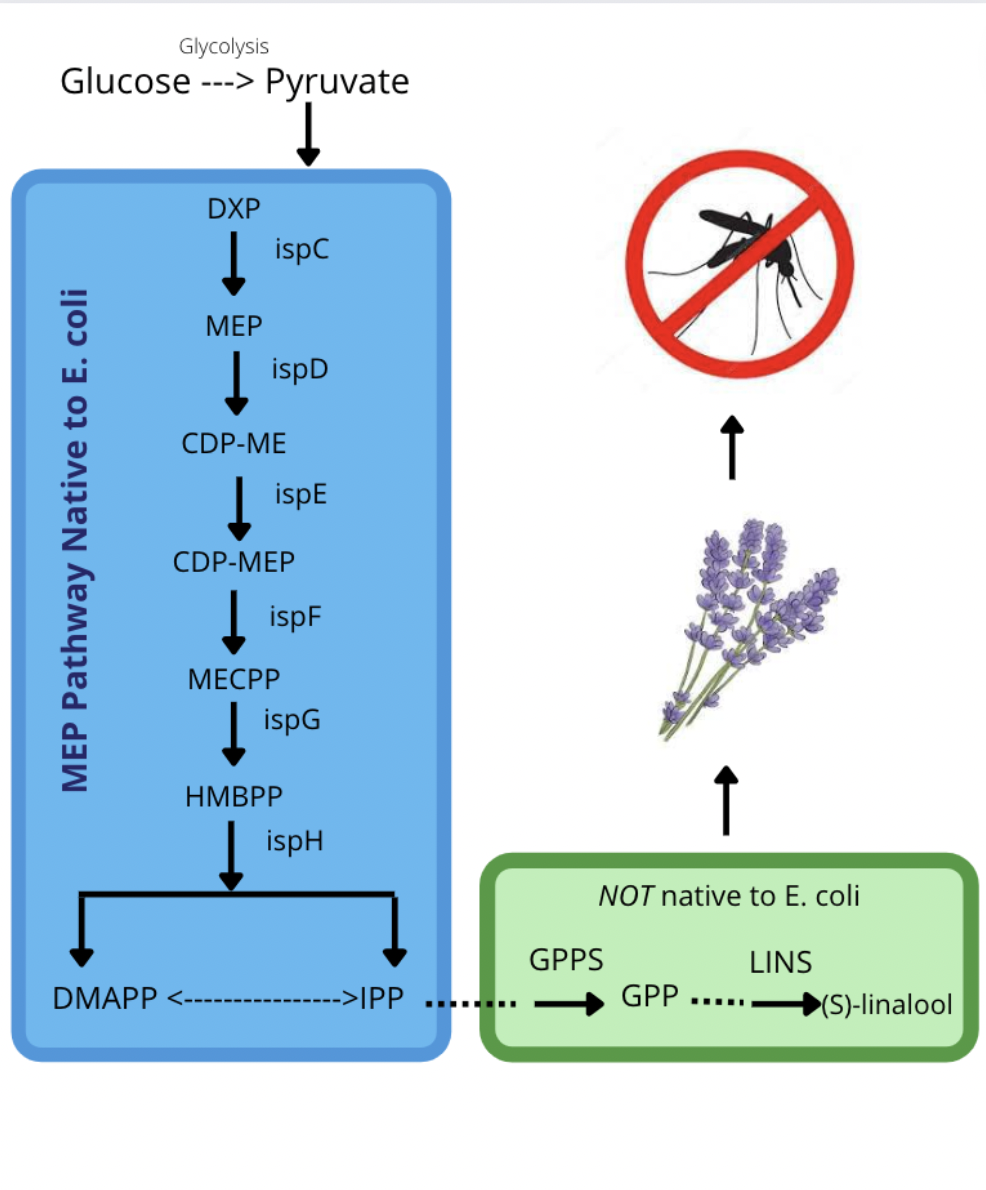
| Figure 7. General overview of AliveSCENT. Glucose would be converted to pyruvate by glycolysis. |
|---|
Future work would involve testing the bacteria samples for their repellent properties in a similar fashion to the mosquito experiments described in this paper. The possibility of inserting the plasmid into E. coli that have been genetically modified to produce a weaker odor should also be investigated.
It is also important to consider that a different species of bacteria could be more suitable for this particular project, but the biological pathways of these species would need to be researched.
Note. This would feed into the native MEP pathway (shaded blue) of E. coli and then be converted to dimethylallyl pyrophosphate (DMAPP) and isopentenyl pyrophosphate (IPP). This would be converted into (S)-linalool after adding a plasmid containing GPPS and LINS (shaded green). The (S)-Linalool compound would emit a spicy floral scent resembling lavender.
Next steps
Over the course of four years, the team has accomplished quite a bit with respect to the AliveSCENT project. However, there is still more work to do. Future testing should be conducted with mosquitoes to further support the repellency effects of lavender scent and its potential public health benefits. These tests should have larger sample sizes, multiple trials using various concentrations, and use additional mosquito species. Unfortunately, time and space have limited the team in this regard.
The possibility of isolating (S)-linalool from the transformed bacteria should also be pursued. This compound should be tested for its repellent properties in a similar fashion to the test conducted on lavender essential oil with mosquitoes, which has been outlined in this paper.
The transformation efficiency of the bacterial transformation should be calculated and improved for future transformations. Larger amounts of bacteria should be transformed for the purpose of continued testing.
The smell test conducted with the bacteria cultures should also be conducted with more volunteers to produce a larger sample size and more accurate results. Additionally, it is very possible that the effects of the transformation could not be fully appreciated, as the natural odor of E. coli overpowered the scent of lavender. Future experiments should include transformation using a strain of indole-deficient E. coli, which has been genetically modified to produce less natural bacteria smell, so it does not interfere with the lavender scent detection. Research should also be conducted to determine if the team should consider another species of bacteria entirely. Biological pathways of other bacteria should be researched in the event that they could also be exploited to produce a lavender scent, and preliminary design work could begin to consider what a commercial product utilizing the AliveSCENT mosquito repellent approach might look like.
Author contributions
T.C. conducted background research. Y.C. and E.F. designed the experimental protocols. Y.C. and E.F. conducted nursery assembly and mosquito care. Y.C., E.F., performed the mosquito experiment. B.C., Y.C., T.C., E.F., S.M., and T.P. performed the bacterial transformation and smell tests. Y.C. and E.F. wrote and edited the abstract. Y.C. created and developed the video. Y.C. collected and created the manuscript graphics. B.C., Y.C., T.C., E.F., S.M., and T.P. wrote the manuscript, which B.C., Y.C., T.C., and E.F. edited. Y.C. designed the plasmid. Y.C., T.C, and E.F. compiled references.
Acknowledgments
The Tyngsborough High School BioBuilder Club would like to give thanks to our mentor, Michael Sheets, whose guidance has been an invaluable resource. He also helped with the construction of the plasmid and guided the team through the bacterial transformation. We would also like to extend our thanks to Dr. Natalie Kuldell, who allowed our BioBuilder Club to use the Learning Lab to conduct numerous hands-on experiments, including the bacterial transformation described in this paper. She also gave us the opportunity to attend the BioBuilder Final Assembly, where we learned about many other teams and their amazing experiments. We would also like to thank Dr. Wagman. He generously donated the original mosquito eggs, taught us how to raise them, and helped us to design an experimental protocol for the mosquito experiments. The team also recognizes Hiroko Kaczmarek for developing and testing the Optical Density and Smell Test protocols. Thank you to BioMade for making the team’s plasmid synthesis a reality. It should also be recognized that Sophia Lundberg gave the team permission to use her art in this paper (see Figures 1 and 3), as did Atena Jasim (see Figure 5). Additionally, the team would like to acknowledge Nathaniel Marino for his contribution in helping to edit the manuscript. The BioBuilder team would also like to extend their gratitude towards the administrative staff at Tyngsborough High School for assisting the club with planning our fantastic field trips and for their continued support throughout the years. Lastly, a huge thank you to our advisor Dr. Rebekah Ravgiala for the countless hours spent on research, experiments, field trips, and fundraising to keep the club running every year.
References
Aldulaimy, M., Angelo, E., Collins, Y., Cote, G., Fugazzotto, E., Lundberg, S., Patel, M., Puma, S., & Riley, C. (2021, October 25). AliveSCENT: Evaluating the potential use of limonene as a mosquito repellent. BioTreks. https://biotreks.org/e202115/
Centers for Disease Control and Prevention. (2020, March 5). What is a mosquito. Retrieved April 26, 2023, from https://www.cdc.gov/mosquitoes/about/what-is-a-mosquito.html
Collins, Y., Fugazzotto, E., Jasim, A. & Tirado D. (2022, October 25). AliveSCENT: Evaluating the potential use of various essential oils as a mosquito repellent. BioTreks. https://biotreks.org/e202201/
Mayo Clinic. (2023, February 9). Diseases & conditions: Malaria: Symptoms and causes. https://www.mayoclinic.org/diseases-conditions/malaria/symptoms-causes/syc-20351184
World Health Organization. (2023b, March 17). Dengue and severe dengue. https://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue World Health Organization. (2023a, March 29). Malaria.https://www.who.int/news-room/fact-sheets/detail/malaria