Ria Vaishnavi ▫ BioBuilderClub, Andover High School, Andover, Massachusetts, United States
Reviewed on 6 May 2023; Accepted on 13 July 2023; Published on 16 October 2023
With help from the 2023 BioTreks Production Team.
With more than 170,000 cases diagnosed annually, Meningiomas are one of the most common types of brain tumors for the elderly. One reason behind the replication of cells composing Meningioma tumors is an overproduction of cerebrospinal fluid (CSF) in the brain, resulting in arachnoid cap cells absorbing too much CSF. This then leads to an overproduction in cell replication, causing a tumor to form. The formation of the tumor occurs in the Subarachnoid mater, specifically the choroid plexus where the CSF production site is found. This approach attempts to use AAV vectors to deliver carbonic anhydrase inhibitors (CAIs), which reduce CSF production at the choroid plexus, to reduce overall CSF production in patients with meningioma tumors. The vectors would deliver the CA1 gene to code a carbon anhydrase inhibitor, known as the CA1 proteins, to reduce the CSF level production in meningiomas. The vectors would be delivered by intracranial injection of Adeno-associated Viral Vectors injection. Currently, the surgical removal of meningiomas is an invasive process with both long and short-term side effects of the surgery and involves a lengthy recovery process. This study aims to mitigate the long-term effects of the surgery, provide a less invasive form of treatment, and increase the quality of life of patients with meningiomas.
Keywords: Meningioma, tumor, brain, cancer
Authors are listed in alphabetical order. Eva Holm-Anderson and Lindsey L’Ecuyer from BioBuilderClub, Andover High School, Andover, Massachusetts, United States mentored the group. Please direct all correspondence to .
Roughly 3% of those over 60 years, or more than 170,000 people, will be diagnosed with a meningioma in their life (Cleveland Clinic, 2022). Over the course of a decade, the average survival rate with a meningioma is about 60%. Though this number is relatively high, patients report having difficulties with emotions, memory, attention, and other executive functions needed for daily life (Nassiri et al., 2020). Tumors are caused by uncontrollable cell replication of abnormal or damaged cells. As of now, there is no specific reason as to what causes Meningiomas to form apart from being exposed to radiation in childhood (National Cancer Institute, 2021a). What is known, however, is that meningioma tumors are made up of arachnoid cap cells (Ogasawara et al., 2021). These cells are found in the meninges that protect the brain and spinal cord (Neurosurgery Wiki, 2018) (see Figure 1).
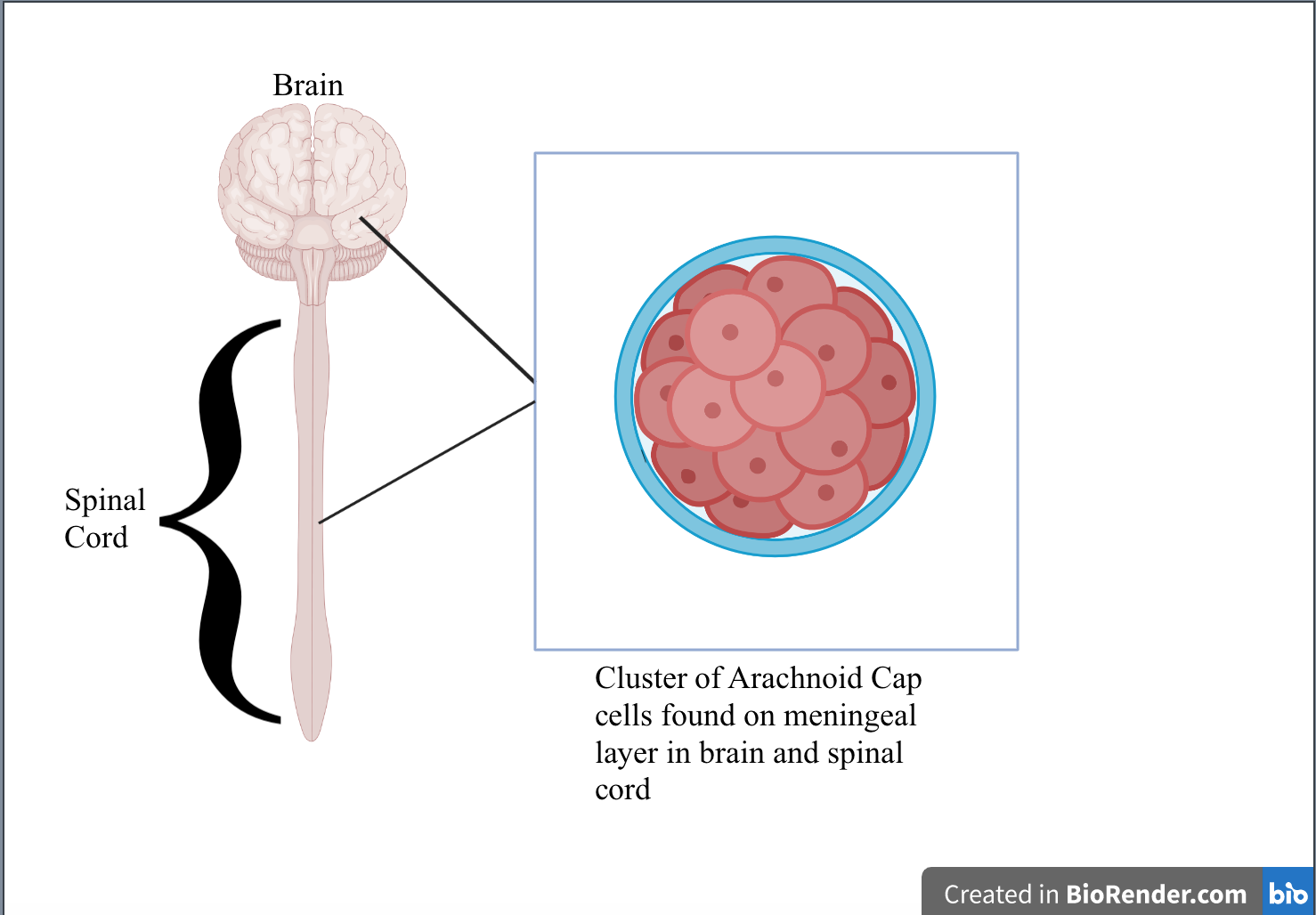
| Figure 1. Arachnoid cap cells and their location in the brain and spinal cord. |
|---|
The meningines itself can be further broken down into the following layers: the dura mater, the arachnoid mater, and the pia mater (Mayo Clinic, n.d.) (see Figure 2) The listing of these layers corresponds to their location from the outermost to the innermost layer of the brain. The Arachnoid mater can be further broken down into the following layers from the outermost (closest to the dura mater) to the innermost (closest to the pia mater), respectively: the mesothelial layer, a central layer, and a deeper layer.
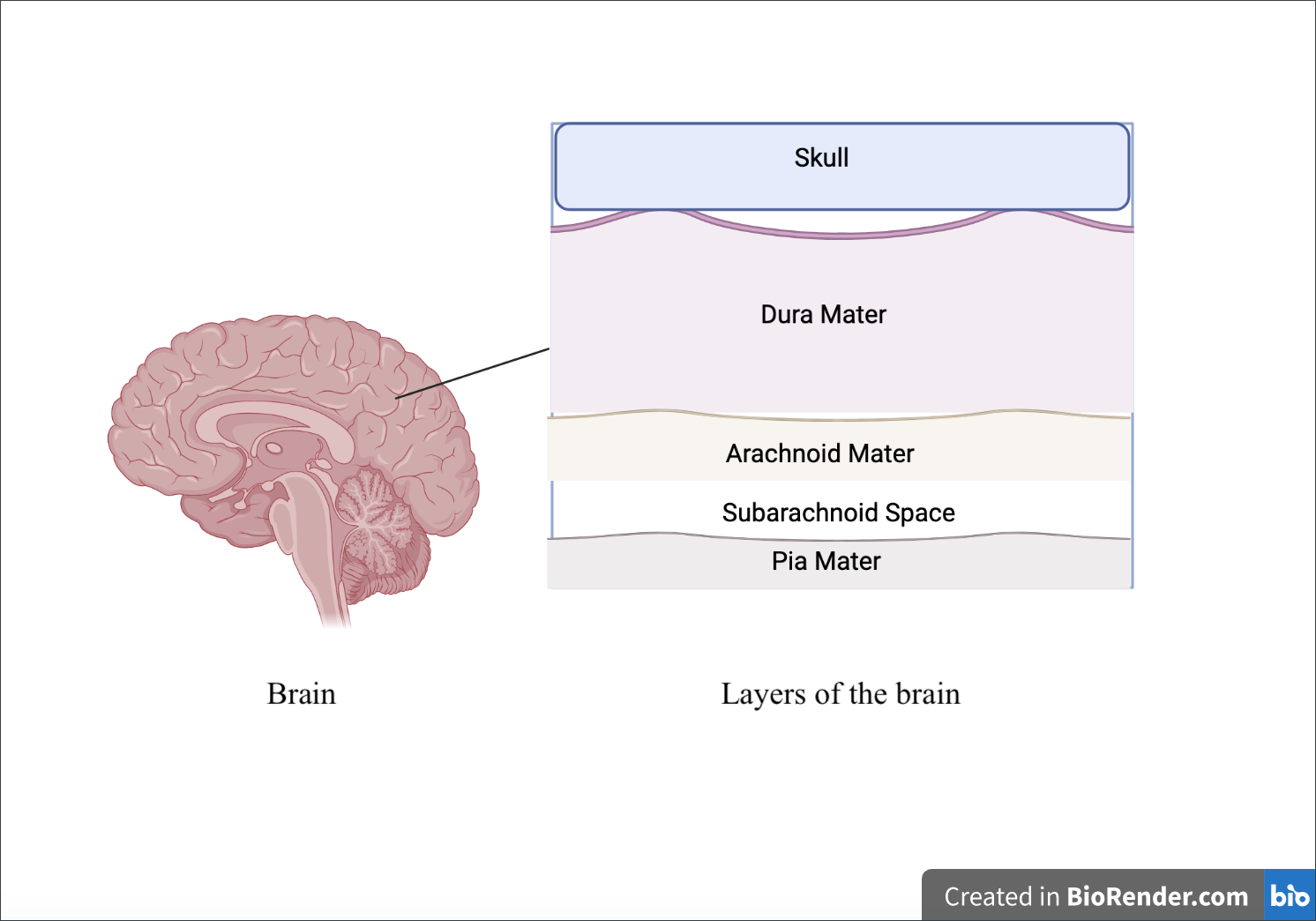
| Figure 2. Composition of layers of the brain from the Skull (closest to the surface) to Pia Mater (furthest from the surface). |
|---|
After the deepest layer of the arachnoid mater, there is the subarachnoid space which connects the deepest layer of the arachnoid mater and the Pia mater (see Figure 3) Within the Pia mater, there are choroid plexuses that produce cerebrospinal fluid (CSF) (Javed et al., 2023) Arachnoid cap cells absorb CSF, allowing them to replicate in their environment (ScienceDirect), which could be a possible cause behind the spreading of Meningioma tumors since these cells are located in the meninges covering the brain and spinal cord. (National Council Institute, 2021) CAIs are one of the ways to reduce CSF levels, thus by inserting them into the pia mater where the meningioma tumor is detected and where choroid plexuses are located would reduce the levels of CSF made while simultaneously preventing the spread of the tumor (Nelson, 2018).
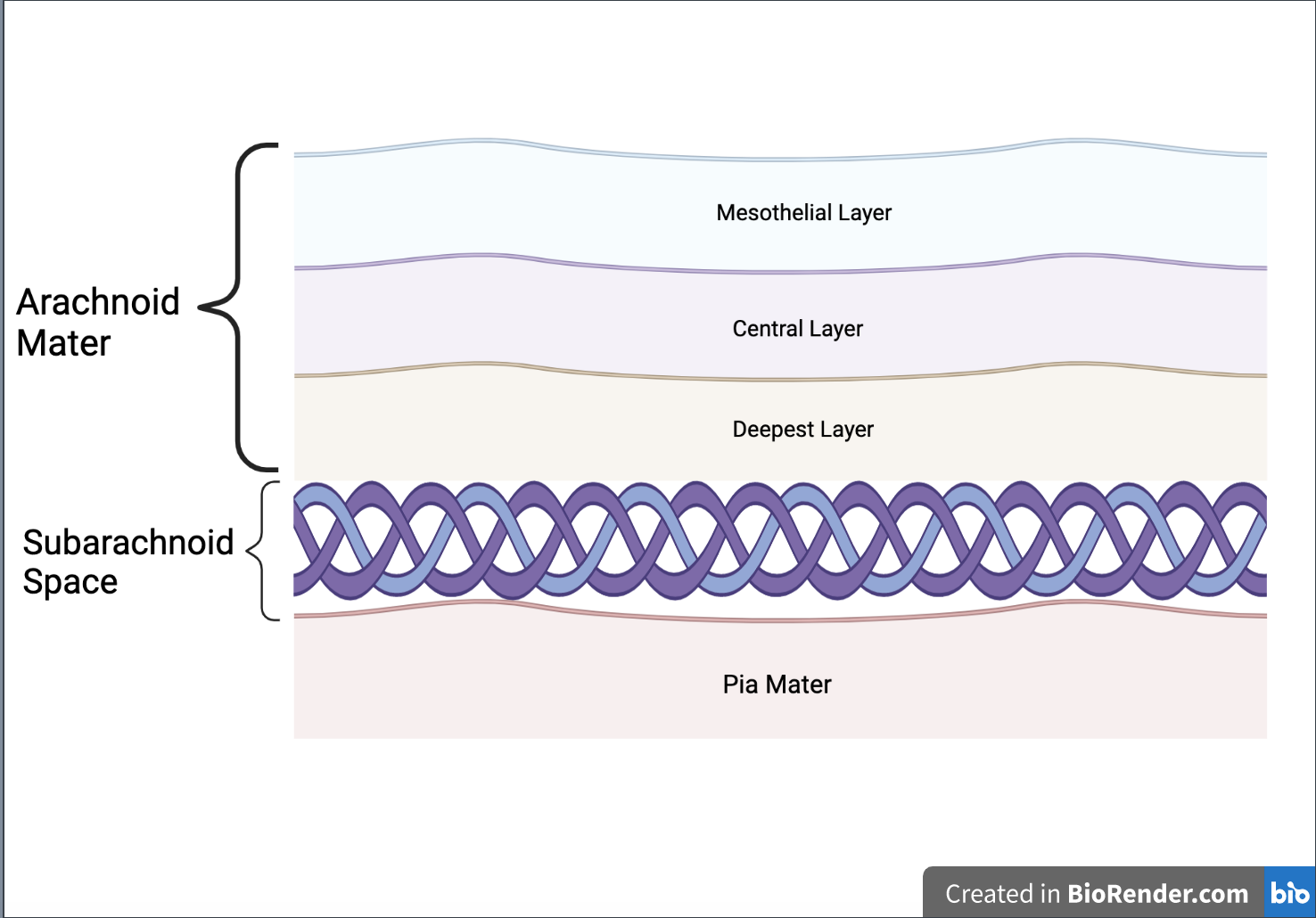
| Figure 3. Composition of the arachnoid mater, subarachnoid space, and pia mater. |
|---|
As of now, there is no known concentration of CAIs to reduce CSF levels in meningioma tumors, however, CAIs have been proven to work in other medical complications such as glucomas, in which acetazolamide was given to patients in 250mg to 1000mg per day. The greatest amount of progress shown in the quickest amount of time was 20 minutes, thus showing how a similar approach to meningiomas would be a quick and fast-acting treatment as opposed to surgery (Aslam & Gupta, 2023).
Systems level
An incision will be made to inject the AAV vector directly into the dura mater. After injection, the vector would have to cross the blood brain barrier (BBB). The BBB is a barrier that selectively regulates blood-borne molecule entry to the brain and preserves the brain’s homeostatic environment. Increased permeability of the BBB can lead to edema formation (Stamatovic et al., 2008), an excess of fluid that disrupts the brain’s homeostatic state (Scallan et al., 2010). Homeostasis is when the brain maintains a balance with its environment. Currently, there are two main approaches to delivering viral vectors across the BBB: transient disruption of brain microvasculature, endothelial tight junctions and receptor-mediated transcytosis (RMT) (Fu & McCarty, 2016).
Disrupting the brain’s microvasculature endothelial tight junctions can result in increased paracellular permeability, allowing for different molecules, or in the case of this study, a vector, to cross the membrane (Stamatovic et al., 2008) (see Figure 4 & Figure 5). Paracellular permeability refers to the transport between cells in the BBB pathway (Yu, 2017). RMT is a process regulated by a series of steps. First, a ligand will bind to a receptor triggers adsorptive endocytosis, a process by which the ligand molecule is now internalized in the cell and in a vesicle. From this point, the vesicle may proceed towards a number of locations to satisfy a number of different functions, but in RMT, the vesicle travels to the basolateral membrane, a membrane that’s part of the brain, where transcytosis, the process by which a vesicle can fuse to the membrane and release it outside into its environment (see Figure 6) (Pulgar, 2019).
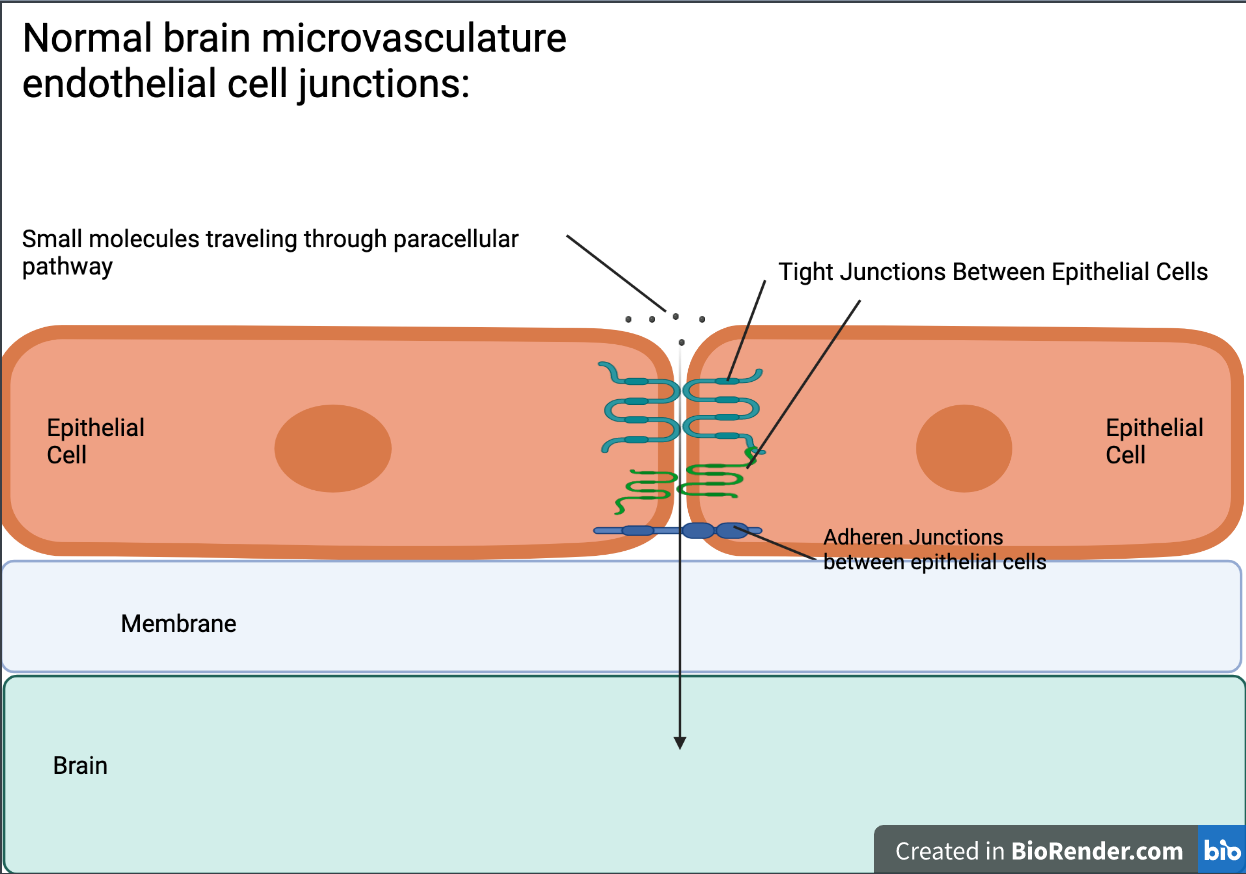
| Figure 4. Normal brain microvascular endothelial cell junctions and the pathway of small molecules traveling through the paracellular pathway. |
|---|
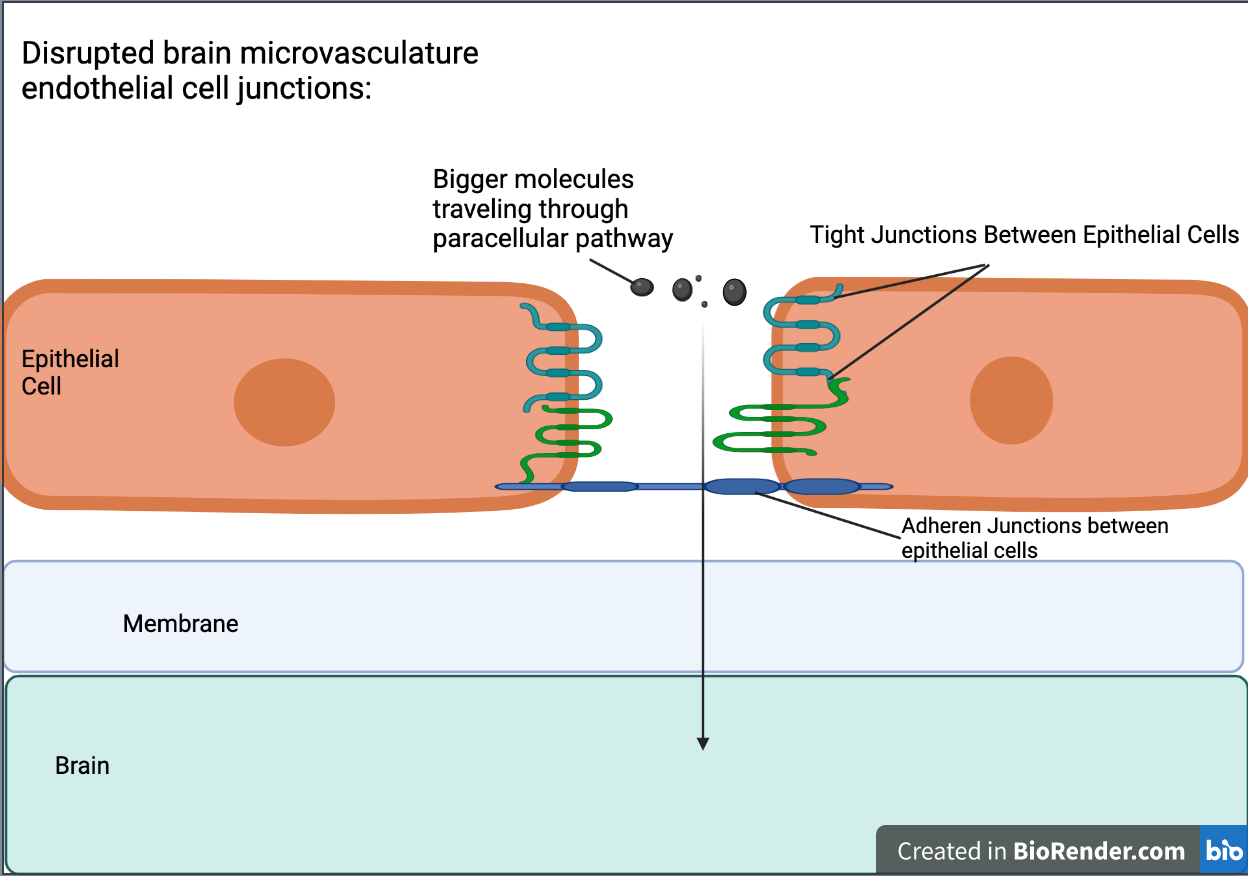
| Figure 5. Disrupted brain microvasculature endothelial cell junctions and the pathway of large molecules traveling through the paracellular pathway. |
|---|
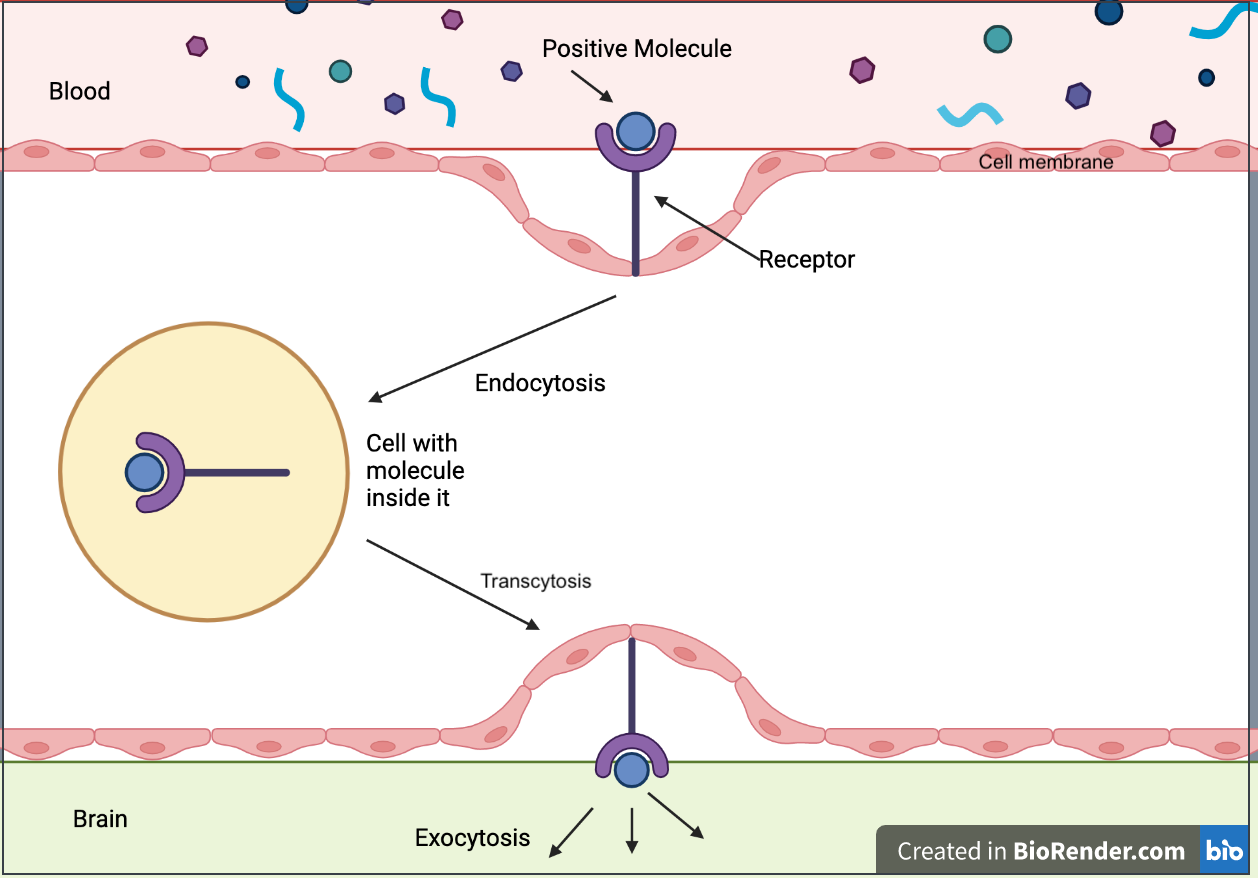
| Figure 6. Receptor-mediated transcytosis (RMT) |
|---|
This method of injection is a commonly used method of injecting AAV vectors specifically. These injections are specifically beneficial for this type of treatment due to the ability to adjust injection location, volume, rate, AAV serotype, and the promoter driving gene expression for each individual. Consequently, allowing for a personalized treatment for every Meningioma patient. (Lowery & Majewska, 2010).
Device level
In the AAV vector the open reading frame (ORF), in this case the CA1 gene, contains the DNA sequence parts necessary for coding for the CA1 protein. AAV vectors are best for this design due to their ability to transduce nondividing cells; they do not have viral genes encoding immunogenic proteins, and they have the ability to be prepared at high titers, meaning they allow for a higher degree of protection from infection over time, and they can reach long-term transgene expression in animal models (Adeno associated virus vector. Adeno Associated Virus Vector – an overview). The CA1 gene codes for the protein carbonic anhydrase 1 (CA1), a known protein in the reduction of the overproduction of CSF, carbonic anhydrases (CAs) catalyze carbon dioxide (CO2), and water (H2O) to make bicarbonate and protons whilst avoiding the creation of carbonic acid. In this process CAs will remove the proton from water, making a zinc-hydroxyl group and a separate proton. Using histidine or glutamate, the proton is transferred to external buffer molecules, effectively causing the reduction of CSF levels (National Library of Medicine, 2023a). Through the insertion of the CA1 gene, we would provide the cell with specific instructions to produce the CA1 protein to reduce CSF levels in the pia mater.
The CA1 gene is not only applicable to meningiomas but also breast cancer (National Library of Medicine, 2023a). The CA1 gene can be a potential oncogene meaning it is a gene that has the ability to become cancerous. In addition to being a potential oncogene, CA1 is vulnerable to ankylosing spondylitis. Ankylosing spondylitis is a condition in which bones in the spine fuse together, resulting in a posture causing respiratory difficulties (Mayo Clinic, 2023). The CA1 gene is versatile in its use and is active in numerous biological processes such as respiration, calcification, acid-base balance, bone resorption, and the formation of aqueous humor. CA1 has low brain regional specificity, and therefore may be used in numerous areas of the brain as well as in other biological systems such as the respiratory system. CA1 has been proven useful in suppressing augmented breaths and airway protective reflexes are involved in regular cellular respiration, without interrupting the expanding and contracting motions of the lungs and other activity of muscles.
For this method of treatment, an intracranial AAV vector Injection would be the most effective. Intracranial injections are used in a variety of neurodegenerative diseases, for example, intracranial malignancies, to inject a specific drug into a particular part of the brain (Bilsky, 2023). Before injecting the AAV vector containing the instructions for coding the CA1 protein directly into the part of the dura mater located nearest to the meningioma tumor itself, an incision large enough to administer the needle will be needed (See Figure 7). In order to safely inject the AAV vector into the dura, stereotaxic coordinates, a micropipette, and an automated pump will be needed to minimize the amount of damage done to nearby areas of the brain (Lowery & Majewska, 2010). Stereotaxic coordinates are coordinates given by imaging to help direct the injection to reach a specific part of the body (Merriam-Webster, n.d.). Though this is an invasive process by definition, there is a new revolution in minimally invasive injection treatment. A similar approach through the treatment of patients with radicular lumbar spine syndrome was seen and proved to allow rectification of pain and deficits as well as result in no major complications due to the treatment process.
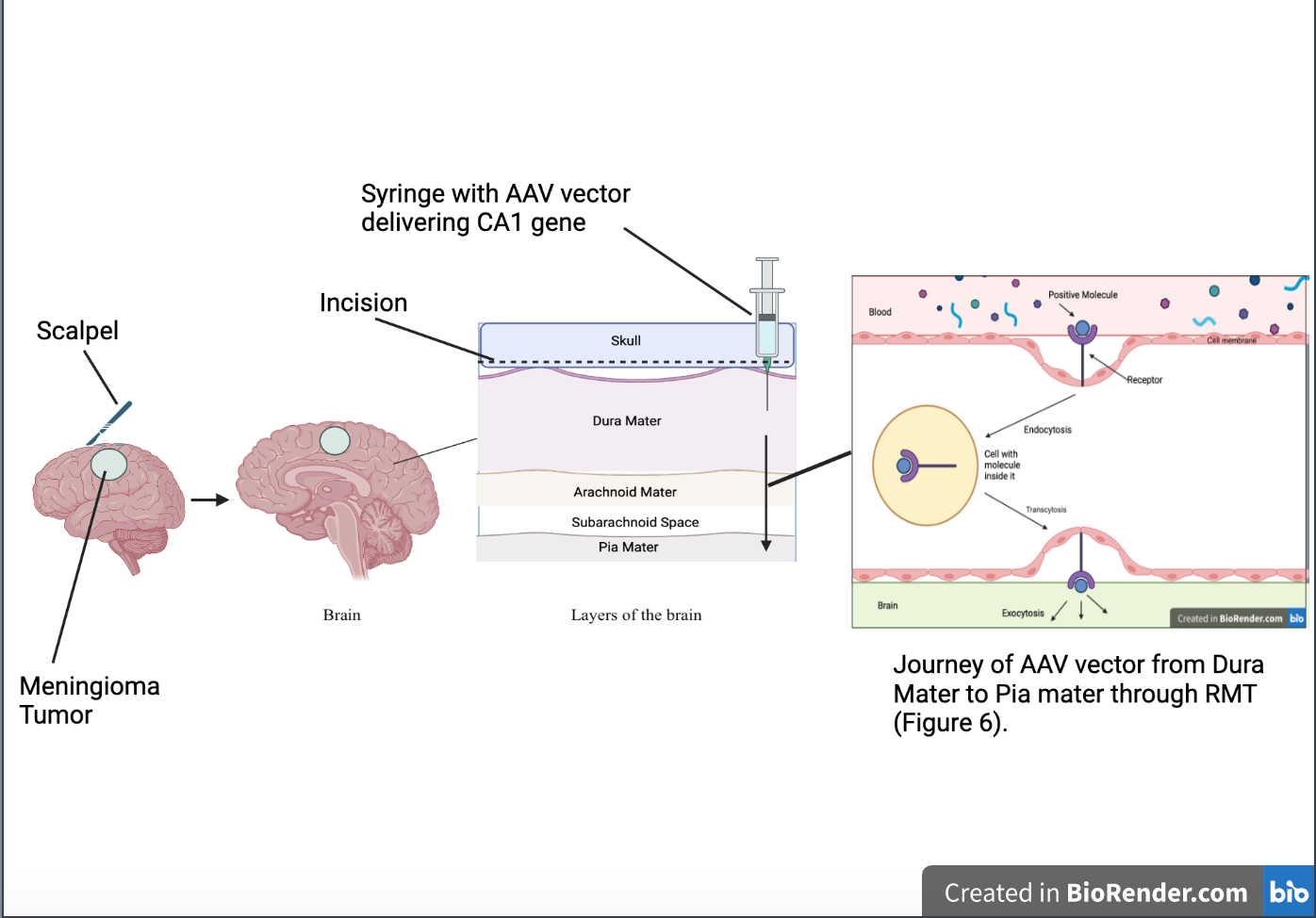
| Figure 7. Steps of treatment process. |
|---|
Note. Steps include: Step 1(Incision of brain with scalpel in area of brain where Meningioma is located) and Step 2 (Insertion of Intracranial AAV vector Injection with AAV vector traveling through different layers of brain from Dura mater to pia mater through RMT (see Figure 6).
Parts level
The vector itself is comprised of the 5’ITR, Foxj1 promoter, Kozak, its OCF, weak Rho-independent terminator and ribosome binding site (WPRE), BGH pA, 3’ ITR, ampicillin, and pUC ori (see Figure 8).
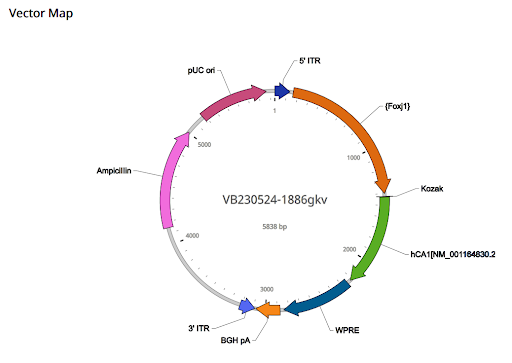
| Figure 8. Components and map of AAV vector |
|---|
5’ ITR: The 5’ITR is located on the 1-130 position, and its size is 130 bp. The 5’ITR allows for the virus to be retrieved from the plasmid and replication of the CA1 genome. It is identical to the wild-type AAV2 genome.
Foxj1 promoter: The promoter is located on the 158-746 positions and is 589 bp. The mammalian gene expression adeno-associated virus (AAV vector) would deliver the Foxj1 promoter to ependymal cells, cells that are specialized cells in the brain and spinal cord. With the Foxj1 promoter, we will be able to communicate to the cell when and where the gene should be turned on thereby giving us the ability to target the ependymal cells specifically.
Kozak: The Kozak is located on the 771 to 776 position and is 6 bp. Kozak completes the translation of the ATG start codon of the Kozak sequence. Without the translation of the start codon, the replication process would be of no use.
The ORF: The ORF is located on the 777 to 1562 position and is 786 bp. In this case, the CA1 gene contains the DNA sequence parts necessary for coding for the CA1 protein. This is a vital part of the system due to the entire system depending on the success of the CA1 genes to code the CA1 protein in order to reduce CSF levels.
WRPE: The WRPE is located on the 1593 to 2190 position and is 598 bp WRPE is a regulatory element that is a sequence in regulating gene expression. It signals the cell instructing it when to begin production of the CA1 protein encoded by the CA1 gene and when to terminate production. In this vector, WRPE also regulates the process of production of the protein, ensuring it is a controlled CA1 production process.
BGH pA: The BGH pA is located on the 2221 to 2428 position and is 208 bp. BGH pA stands for bovine growth hormone polyadenylation signal. With this, mRNA is transcribed and translated, and polyadenylation of mRNA undergoes transcription due to Pol II RNA polymerase. The polyadenylation process is in which the pre-mRNA strand is cleaved, and that strand undergoes polymerization, the process in which a poly A tail is added in, in turn making the new strand more stable and resistant to degradation in its environment.
3’ITR: The 3’ITR is located on the 2436 to 2565 position and is 130 bp. The 3’ITR allows for the virus to be retrieved from the plasmid and replication of the CA1 genome. It is identical to the wild type AAV2 genome. This ITR serves the same purpose as the 5’ITR.
pUC ori: pUC ori is located on the 4524 to 5112 position and is 589 bp. It allows for the plasmid to replicate in great quantities in Escherichia coli cells and regulates plasmid number (500-700). This allows for the plasmid to spread throughout the brain and produce enough of the CA1 protein to lower CSF levels in the Pia mater.
Ampicillin: Ampicillin is an antibiotic-resistant gene that will serve as a selectable marker, allowing for the identification and tracing of cells that have taken up the AAV vector. A successful incorporation of the vector would promote growth in Ampicillin, and those who do not successfully incorporate the vector would die. This process verifies that the vector is successful in inserting itself into the cell (Vector Builder, n.d.).
Safety
This system will be designed in a proper laboratory setting, such as a sterile surgical room, with all the necessary equipment to perform the surgical procedure and all necessary persons for a successful procedure. The operators must receive informed consent from the patient receiving the operation and follow standard ethical principles as listed in the American Medical Association code of ethics. All tools used on the patient must be sterilized, and there must be no transference of needles or repeated use of contaminated tools on patients. Tools needed for the treatment include but are not limited to a scalpel, a micropipette, an automated pump, and a syringe to ensure the procedure is performed correctly. This being said, the treatment should follow the standard procedure of an operation with appointments before the operation, a team of surgeons in the sterile surgical room, and follow-up appointments after the surgery to ensure the surgery was successful as well as mitigate any potential aftereffects, such as headaches, post-operation. The role of CAIs is to reduce the number of bicarbonate ions produced and to lower fluid flow levels between different parts of the brain (Aslam & Gupta, 2023). Carbonic anhydrase is not otherwise dangerous to humans unless suffering from hypokalemia, hyponatremia, metabolic acidosis, hyperchloremic acidosis, adrenal insufficiency, or marked renal impairment under which this treatment may cause electrolyte imbalances (Aslam & Gupta, 2023) All potentially hazardous materials, such as needles, should be handled with caution by ensuring they do not get reused and are disposed of properly after use. After surgery, all tools used must undergo the process to become sterilized once again.
In terms of a wider context, the use of gene therapy may be controversial to individuals. However, individuals will only receive this treatment if they give consent for surgeons to do so. There is controversy surrounding a variety of treatments that introduce new products into our biological system, such as vaccines with antigens, but this treatment will only be performed on people who consent to receive it. The patient will be presented with differing options of treatment (i.e., tumor removal operation, AAV vector insertion, Gamma Knife radiosurgery, and chemotherapy) and will have full control as to which treatment they wish to receive after consulting with a neurosurgeon or neurologist in a hospital.
Discussions
This method of approaching meningiomas is a sharp contrast to the normal surgical procedure (John Hopkins Medicine, n.d.-a) The surgical removal of the tumor is a lengthy and intense recovery process, including difficulty in speech, movement, and overall physical coordination (Cohen-Gadol, 2023). With this method of treatment, there is a greater likelihood that patients suffering from the tumor will be able to recuperate much quicker after this procedure, as the effects of lowering CSF levels include nausea, dizziness, and headaches (Telano & Baker, 2023).
One of the main challenges that need to be addressed is the overall lack of research in regard to this specific tumor. There could be possible side effects of the treatment unannounced until after the surgery itself is complete such as problems with balance or pain and painful sensations in limbs (Tufts Medical Center, n.d.).
Meningiomas, for the majority of the time, are benign and often treated through surgery in which the whole tumor is removed. There are also ongoing radiation therapies, such as Gamma Knife radiosurgery, which are used to decrease the size of the tumor, as well as chemotherapy used to treat Meningiomas (John Hopkins Medicine, n.d.-b) In Gamma Knife radiosurgery, gamma rays send radiation to the cells to inhibit their ability to grow. With Gamma Knife radiosurgery, patients will face hair loss, swelling, and pain, as well as fatigue as a direct result (John Hopkins Medicine, n.d.-b).
Chemotherapy is not often used as a treatment, however, patients with meningiomas whose tumor’s removal may result in greater harm due to its location being too close to the spine, hence some patients may undergo chemotherapy as opposed to surgery. Direct effects of chemotherapy include appetite loss, lack of energy, hair fallout, and infections (Moffit Cancer Center, n.d.).
Inserting CAIs would provide a more immediate form of treatment and is an alternative to those who do not wish to receive forms of radiation therapy. As databases progress with more advanced knowledge every day, the potential for improving this method of treatment can be refined after more advancements in the field have been made.
In this treatment, if the AAV is not incubated for 48-72 hours, there is a risk that the genomes will become singular-stranded (Gray et al., 2011). This means bacteria is now sensitive to DpnI digestion and differentiates from unmethylated DNA in eukaryotic cells. This means the AAV is now indigestible. leaving the plasmid at risk of the DNA being unable to be used for molecular cloning (GenScript, n.d.). If methylated by bacteria, there is also the possibility of restriction enzyme cleavage being inhibited. Restriction enzyme cleavage is the process by which enzymes bind themselves to DNA sequences and cleave (New England Biolabs, n.d.), meaning breaking the covalent bonds between sugar and phosphate molecules in nucleotides (UMASS, n.d.).
If less than normal amounts of CSF levels are in the brain, this leaves the potential for the patient to suffer from headaches and other symptoms that are often associated with post-surgery rehabilitation. Also, when CSF levels are too low, there is an opportunity for the brain itself to sag, resulting in the meninges of the brain being stretched and causing pain. If, in the case of the brain sag, the pain will subside once lying down, however, when standing or sitting in an upright position, it will be painful for the patient (John Hopkins Medicine, n.d.-c) With the head being craned abnormally due to low CSF levels, there is a chance for a patient to obtain neck issues such as irritated ligaments or spasms as a result of the strain on the neck when lying down too much.
It is necessary to monitor all vitals during the surgical procedure as well as follow up with the patient after treatment to determine if any follow-up procedures are necessary. In the case that CSF levels are lowered too much, there will have to be a second operation in which a vector is inserted containing the gene to code for CSF but shut off after the CSF levels reach a certain point. All patients should be handled with the utmost care before the surgery, during, and after to assist with improving the quality of life of the patient. The equipment necessary for this operation are standard tools found in most hospitals; therefore, surgery should not be complicated and a positive alternative to having the whole tumor removed.
Next steps
If the insertion of the CA1 gene via the AAV vector proves to be successful in regard to application and treatment, similar diseases that spread through the excess of CSF, such as hydrocephalus may be examined to determine if a similar treatment is plausible. Hydrocephalus is a neurological condition in which CSF stores itself in the ventricles of the brain, causing a build-up that can lead to the development of pressure on the tissues inside the skull. Rather than injecting the AAV vector into the Dura mater and sending the vector down an RMT, the injection may be administered directly into the ventricles of the brain. If the AAV vector inserting the CA1 gene works for numerous diseases, such as Hydrocephalus, then further studies would be necessary for deducing the possible side effects after treatment and whether or not they would be similar to those of the post AAV vector treatment operation effects.
To expand and add to the limited knowledge of meningiomas and hydrocephalus an experiment may be conducted in which the anatomical structures of the tumors with no treatment, the surgical removal of the Meningioma, the AAV vector injected brain with meningioma, and the AAV vector injected brain with Hydrocephalus. The experiment can compare the effect of this reduction in CSF level on the specific brain locations targeted and other areas of the brain involved with the systems CAIs, such as the brainstem and the respiratory system.
To adapt this study for the high school laboratory setting, samples of the brain of an organism that is no longer alive due to a meningioma which has all layers from the dura mater to the pia mater, can be obtained and tested using the AAV vector and a green fluorescent protein (GFP). It is critical that students will be able to determine CSF levels in which they can collect the CSF fluid and preserve it in a controlled environment. In the controlled environment, the sample will be cultured, and students will be able to use a syringe to deliver the AAV vector with the CA1 gene that codes for the CA1 protein, resulting in a decrease in CSF levels with the GFP. If present in the brain, the fluorescence will cause that part of the brain to glow, and from this point, students can re-measure the CSF levels post-operation.
Author contributions
I wrote and researched this paper myself whilst actively communicating with members of the club for assistance when necessary. Fellow members of my club helped me with the logistics of my paper and gave me feedback on how to improve. Research has been done proving CA IX/XII inhibitors’ ability to reduce the formation of primary tumors and metastases (Supuran, 2020).
Acknowledgements
This study was made possible due to members of the Andover High School BioBuilders Club. Through this club, I have been given the opportunity to research a personal topic of interest whilst combining it with one of my other interests, writing. I enjoyed being able to conduct research in the lab setting alongside my peers. I’d also like to thank Ms. Lindsey Le’Cuyer (Club Advisor), Ms. Eva Holm-Anderson (AP Biology Teacher), and Emily Packer (a peer of mine) for helping me throughout this process and giving me the knowledge necessary to be successful in this subject.
References
Adeno associated virus vector. Adeno Associated Virus Vector – an overview | ScienceDirect Topics. (n.d.). https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular -biology/adeno-associated-virus-vector#:~:text=AAV%20vectors%20are%20promising%20therapeutic,transgene%20expression%20in%20animal%20models
Arachnoid. Arachnoid – an overview | ScienceDirect Topics. (n.d.). https://www.sciencedirect.com/topics/immunology-and-microbiology/arachnoid
Aslam, S., & Gupta, V. (2023). Carbonic anhydrase inhibitors. StatPearls Publishing
Bilsky, M. H. (2023, May 25). Overview of intracranial tumors. MSD. https://www.merckmanuals.com/professional/neurologic-disorders/intracranial-and-spinal-tumors/overview-of-intracranial-tumors
Black, P., Kathiresan, S., & Chung, W. (1998). Meningioma surgery in the elderly: A case-control study assessing morbidity and mortality. Acta Neurochirurgica, 140, 1013-1017. https://doi.org/10.1007/s007010050209
Cleveland Clinic. (2022, September 5). Meningioma. https://my.clevelandclinic.org/health/diseases/17858-meningioma#%20
Cohen-Gadol, A. (2023, January 6). Surgical management of meningioma. https://www.aaroncohen-gadol.com/patients/meningioma/treatment/surgical-management
Cooper, G. M. (2000). The cell: A molecular approach. Sinauer Associates.
UMASS. (n.d.). DNA cleavage. https://profiles.umassmed.edu/display/129176
Dutta, S., & Goodsell, D. (2004, January). Molecule of the month: Carbonic anhydrase. RCSB PDB. https://pdb101.rcsb.org/motm/49
Fu, H., & McCarty, D. M. (2016). Crossing the blood–brain-barrier with viral vectors. Current Opinion in Virology, 21, 87-92. https://doi.org/10.1016/j.coviro.2016.08.006
GenScript. (n.d.). What is restriction enzyme digestion? https://www.genscript.com/what-is-restriction-digestion.html
Gray, S. J., Choi, V. W., Asokan, A., Haberman, R. A., McCown, T. J., & Samulski, R. J. (2011). Production of recombinant adeno‐associated viral vectors and use in in vitro and in vivo administration. Current Protocols in Neuroscience, 57(1), 4-17. https://doi.org/10.1002%2F0471142301.ns0417s57
GreenCards. (n.d.). CA2 gene: Carbonic anhydrase 2. Retrieved April 28, 2023, from https://www.genecards.org/cgi-bin/carddisp.pl?gene=CA2
Javed, K., Reddy, V., & Lui, F. (2023). Neuroanatomy, choroid plexus. StatPearls Publishing.
Johns Hopkins Medicine. (n.d.). Meningioma treatment. https://www.hopkinsmedicine.org/health/treatment-tests-and-therapies/meningioma-treatment#
Johns Hopkins Medicine. (n.d.-b). Gamma knife radiosurgery. https://www.hopkinsmedicine.org/health/treatment-tests-and-therapies/gamma-knife-radiosurgery#
Johns Hopkins Medicine. (n.d.-c). Low CSF headache. https://www.hopkinsmedicine.org/health/conditions-and-diseases/headache/low-csf-headache#
Lowery, R. L., & Majewska, A. K. (2010). Intracranial injection of adeno-associated viral vectors. Journal of Visualized Experiments, (45), e2140. https://dx.doi.org/10.3791/2140
Mayo Clinic. (2023, February 21). Ankylosing spondylitis. Mayo Foundation for Medical Education and Research. https://www.mayoclinic.org/diseases-conditions/ankylosing-spondylitis/symptoms-causes/syc-20354808#
Mayo Clinic. (n.d.). Meninges. Mayo Foundation for Medical Education and Research. https://www.mayoclinic.org/diseases-conditions/meningioma/multimedia/meninges/img-20008665
McCarthy, K. D., & Reed, D. J. (1974). The effect of acetazolamide and furosemide on cerebrospinal fluid production and choroid plexus carbonic anhydrase activity. Journal of Pharmacology and Experimental Therapeutics, 189(1), 194-201.
MedlinePlus. (2022, February 28). How does gene therapy work?. National Library of Medicine. Retrieved April 28, 2023, from https://medlineplus.gov/genetics/understanding/therapy/procedures/
Merriam-Webster. (n.d.). Stereotactic. In Merriam-Webster.com dictionary. https://www.merriam-webster.com/dictionary/stereotactic#
Moffit Cancer Center. (n.d.). Chemotherapy for meningioma. https://www.moffitt.org/cancers/meningioma/treatment/chemotherapy/
Nassiri, F., Suppiah, S., Wang, J. Z., Badhiwala, J. H., Juraschka, K., Meng, Y., Nejad, R., Au, K., Willmarth, N. E., Cusimano, M., & Zadeh, G. (2020). How to live with a meningioma: Experiences, symptoms, and challenges reported by patients. Neuro-Oncology Advances, 2(1), vdaa086. https://doi.org/10.1093/noajnl/vdaa086
National Cancer Institute. (2021a, July 21). Meningioma Diagnosis and Treatment. Retrieved April 28, 2023, from https://www.cancer.gov/rare-brain-spine-tumor/tumors/meningioma
National Cancer Institute. (2021b, October 11). What is cancer?. NIH. https://www.cancer.gov/about-cancer/understanding/what-is-cancer
National Library of Medicine. (2023a, Jun 16). CA1: Carbonic anhydrase 1: Homo sapiens (human). National Center for Biotechnology Information. https://www.ncbi.nlm.nih.gov/gene?Db=gene&Cmd=DetailsSearch&Term=759
National Library of Medicine. (2023b, Jun 21). CA3: Carbonic anhydrase 3: Homo sapiens (human). National Center for Biotechnology Information. https://www.ncbi.nlm.nih.gov/gene/761
Nelson, S. L. (2018, June 4). Hydrocephalus Medication: Carbonic anhydrase inhibitors, Loop diuretics. Medscape. https://emedicine.medscape.com/article/1135286-medication
Neurosurgery Wiki. (2018, September 13). Arachnoid cap cell. https://operativeneurosurgery.com/doku.php?id=arachnoid_cap_cell
New England Biolabs. (n.d.). Restriction enzyme cleavage: “Single-site” enzymes and “multi-site” enzymes. https://international.neb.com/tools-and-resources/feature-articles/restriction-enzyme-cleavage-single-site-enzymes-and-multi-site-enzymes
Occhipinti, R., & Boron, W. F. (2019). Role of carbonic anhydrases and inhibitors in acid–base physiology: Insights from mathematical modeling. International Journal of Molecular Sciences, 20(15), 3841. https://doi.org/10.3390/ijms2015384
Ogasawara, C., Philbrick, B. D., & Adamson, D. C. (2021). Meningioma: A review of epidemiology, pathology, diagnosis, treatment, and future directions. Biomedicines, 9(3), 319. https://doi.org/10.3390%2Fbiomedicines9030319
Pulgar, V. M. (2019). Transcytosis to cross the blood brain barrier, new advancements and challenges. Frontiers in Neuroscience, 12, 1019. https://doi.org/10.3389/fnins.2018.01019
Scallan, J., Huxley, V.H., & Korthuis, R.J. (2010). Capillary fluid exchange: Regulation, functions, and pathology. Morgan & Claypool Life Sciences. https://doi.org/10.4199/c00006ed1v01y201002isp003
Spine and Brain. (n.d.). CSF disorders. https://spineandbrain.co.uk/csf-disorders/
Stamatovic, S. M., Keep, R. F., & Andjelkovic, A. V. (2008). Brain endothelial cell-cell junctions: How to “open” the blood brain barrier. Current Neuropharmacology, 6(3), 179-192.
Supuran, C. T. (2020). Experimental carbonic anhydrase inhibitors for the treatment of hypoxic tumors. Journal of Experimental Pharmacology, 12, 603-617.
Telano, L. N., & Baker, S. (2023). Physiology, cerebral spinal fluid. StatPearls Publishing.
The Human Protein Atlas. (n.d.). CA1: Protein summary. https://www.proteinatlas.org/ENSG00000133742-CA1
Tufts Medical Center. (n.d.). Brain tumors. https://www.tuftsmedicalcenter.org/patient-care-services/Conditions-We-Treat/B/Brain-Tumors?
Vector Builder. (n.d.). Adeno-associated virus (AAV) packaging. https://en.vectorbuilder.com/products-services/service/aav-packaging.html
Xiao, G., & Gan, L. S. (2013). Receptor-mediated endocytosis and brain delivery of therapeutic biologics. International Journal of Cell Biology, 2013. https://doi.org/10.1155/2013/703545 Yu, A. S. (2017). Paracellular transport as a strategy for energy conservation by multicellular organisms?. Tissue Barriers, 5(2), 2509-2518. https://doi.org/10.1080/21688370.2017.130185
