Jittanan Tirawatthanaprapha, Napai Pattamajintathamrong, Pakpol Kunopasvorakul, Worathas Wongsatidporn, Chanyanath Chinthammit, Aiyada Ieumwananonthachai, Nicha Visuthranukul, Pawarisa Teemaethawee ▫ Medical Research Student’s Association of Patumwan Demonstration School, Bangkok, Thailand
Reviewed on 6 May 2023; Accepted on 13 July 2023; Published on 16 October 2023
With help from the 2023 BioTreks Production Team.
The Lamiaceae species are widely used amongst other herbs in traditional medicine as they exhibit antimicrobial properties. With increasing interest and protrusion of material science in everyday lives, the antibacterial properties of various plants extracts were tested on four materials including silver, aluminum, glass, and polyethylene. These materials were selected based on their prevalence in everyday objects and their ability to withstand high temperature in the autoclaving process. Crude extracts from the Lamiaceae spp.; Ocimum basilicum, Salvia rosmarinus, Thymus vulgaris, and Origanum vulgare were obtained using water as a solvent. Serial dilution was performed on three plant pathogenic bacteria commonly found in contaminated water: Erwinia carotovora, Salmonella typhimurium, and Pseudomonas aeruginosa. The diluted bacteria and crude extracts were applied onto the materials that were cut into 2.54 cm x 2.54 cm. After leaving the materials to dry in a sterile environment, the surviving bacteria were quantified by stamping on agar plates. Then, the number of colonies were counted after an incubation period of two days using ImageJ. The four Lamiaceae extracts were found to possess inhibitory effects on bacterial growth. When the testing materials varied, inhibition of bacterial growth in metallic surfaces and acceleration of bacterial growth on non-metallic surfaces were observed.
Keywords: Herbal extract, Lamiaceae extract, microbial properties, metallic and non-metallic materials
Authors are listed in alphabetical order. Somkiat Phornphisutthimas from Srinakharinwirot University, Bangkok, Thailand and Jason Ping Zhang from the Medical Research Students’ Association of Patumwan Demonstration School, Bangkok, Thailand mentored the group. Please direct all correspondence to .
Historically, herbs have played a significant role in medicine as they can be naturally cultivated and are accessible to most of the population (Carmona & Pereira, 2013). Concerns are often raised about the efficacy and safety of traditional remedies involving herbs as they are not strictly regulated like mainstream pharmaceuticals (Firenzuoli & Gori, 2007). However, studies have shown that herbs exhibit less side effects than synthetic drugs or chemical drugs that produce reactive oxygen species (ROS) (Parham et al., 2020). An excessive or unmonitored amount of ROS can lead to the disruption of cell homeostasis and is linked to pathologies ranging from autoimmune diseases to oxidative-stress related cancer (Kim et al., 2016; Patel, et al., 2017)
The four herbs from the Lamiaceae family, or the mint family, were chosen prior due to its antimicrobial properties in essential oils and common appearance in culinary settings, as it creates distinct flavors and aromas in food (Carovic-StanKo et al., n.d.). O. basilicum essential oil is mainly composed of methyl cinnamate, linalool, β-elemene, and camphor (Suppakul, et al., 2003). The herb, along with its main constituents, were found to exhibit antimicrobial properties to a range of gram-positive and gram-negative bacteria. Ranging from most to least, the S. rosmarinus essential oil is composed of p-cymene, linalool, γ-terpinene, thymol, β-pinene, α-pinene, and eucalyptol (Ozcan & Chalchat, 2008). A study found that it exhibits antifungal properties. The antimicrobial properties of T. vulgaris is linked to its high concentrations of carvacrol, thymol, and phenols (Parham et al., 2020). Finally, the essential oil of O. vulgare which also exhibits antibacterial properties is mainly composed of carvacrol, β-fenchyl alcohol, thymol, and γ-terpinene (Teixeira et al., 2013). Both S. rosmarinus and T. vulgaris have shown the potential to replace synthetic food preservatives as natural food preservatives in a recent study (Tzima et al., 2015).
Material science is a rapidly growing field with a wide-ranging impact in our daily lives, especially in the age of globalization. Each material has unique properties based on its atomic arrangement, therefore, use varies substantially (Kakani, 2004). A study showed that on plain inanimate surfaces, bacteria can persist ranging from hours to months (Kramer et al., 2006). Upon investigation, there is an increasing interest in developing materials with antimicrobial properties, particularly in healthcare settings where risk of infection is high (Elbehiry et al., 2022). In light of these developments, we aimed to investigate the potential antimicrobial activity of Lamiaceae extracts and their integration to material science.
The main objective of our research was to test the efficacy of Lamiaceae extracts in inhibiting bacterial growth on different materials commonly used in daily life, such as food packaging, including: silver, aluminum, glass, and polyethylene (PET). The materials were selected based on their prevalence in everyday objects and their resistance to the extreme conditions of the autoclaving process (Dar & Nayik, 2022). Furthermore, silver is found to exhibit antimicrobial properties due to its positive-charge ions that bind to certain microorganisms, destroying it in the process (Nielsen, n.d.).
Through our research, we aim to explore potential microbial-associated properties of Lamiaceae extracts and their application with common materials. We hope our research findings will serve as a valuable resource for the further development and optimization of herbal extracts and materials, with possible applications to healthcare systems in modern pharmaceuticalogy and other industries. Additionally, extensive knowledge and techniques of synthetic biology can be utilized to obtain desirable antimicrobial traits of the Lamiaceae properties. For instance, through synthetic biology methods, β-Caryophyllene, which possess antimicrobial properties, can be derived from Lamiaceae species including rosemary, oregano, and Thyme (Vassiliou et al., 2023).
Materials and methods
Preparation of Lamiaceae solution
The herbs (O. basilicum, S. rosmarinus, T. vulgaris, and O. vulgare) were prepared individually with a net weight of 30 grams. The leaves were grounded using a mortar and pestle into coarse grains. The extract was made by adding water to the grain by the ratio of 1 g/mL. Then, the solution was filtered through a cotton-plugged syringe. Finally, the solution was sterilized by an autoclave at a temperature of 121 degrees Celsius for 15 minutes.
Bacterial culture
Nutrient broth (NB) (Wick & Hansen, 2011a) was prepared for bacterial culture. Nutrient agar (NA) (Wick & Hansen, 2011b) was prepared for the disc diffusion method and solutions/surfaces conditions test. The composition of NB and NA is shown in Table 1.
Table 1
| Ingredient of NB | 1 L of LB |
|---|---|
| Distilled Water | 1 L |
| Beef Extract | 1g |
| Yeast Extract | 2g |
| Peptone | 5g |
| Sodium Cloride (NaCl) | 5g |
| Ingredient of NA | 1 L of NA |
| Distilled Water | 1 L |
| Beef Extract | 1g |
| Yeast Extract | 2g |
| Peptone | 5g |
| Sodium Cloride (NaCl) | 5g |
| Agar | 15g |
Table1. Table reporting the composition of Nutrients broth (NB) and the composition of Nutrients agar (NA) with the volume of one liter.
Bacterial preparation by sub-culturing with nutrients broth
100 mL of bacteria including E. carotovora, S. typhimurium, P. aeruginosa was prepared with 300 mL of nutrients broth via micropipette into the flask. After that, we used the orbital shaker to spin the bacterial culture for 48 hours with 250 rpm so it surpasses the log phase and reaches the stationary phase where the growth of bacteria remains constant.
Antimicrobial activity by disc diffusion methods
A stock solution of 1 g/mL was used with a sterile disc 6 mm in diameter. 10 μL of extracted solution was then added to the disc. When the disc dried, it was placed onto the agar plate to measure the clear zone.
The comparison of solutions/surfaces conditions
Serial dilution was performed to lessen bacteria concentration in order to minimize errors in data summarizing by decreasing the chance of overlapping bacterial colonies. Bacteria was diluted to 10-4 CFU/mL. The same concentration was used to test the comparison of solutions/surfaces. The surfaces of aluminum, silver, glass, and plastic were cut into pieces of 2.54 cm x 2.54 cm and sterilized by autoclaving.
Data were collected on the number of colonies formed on the agar using the imprinting method with a two-day incubation period (Giannella, 1996; EHA Consulting Group, n.d.; Santander & Biosca, 2017). At the beginning, each petri dish was divided into four quadrants to maximize space efficiency. ‘Empty,’ acts as a negative control with the agar undisturbed (upper left -). ‘Solution-only,’ to check for the contamination of the solution (upper right – first quadrant). ‘Bacteria-only,’ to examine the antimicrobial effect of the materials alone (lower left – third quadrant); and ‘Bacteria-and-solution,’ to examine how both the solutions and surfaces used in conjunction affect the growth of bacteria (lower right – fourth quadrant).
10 μL of extract solution and bacteria solution were added to the first quadrant and the third quadrant , respectively. 5 μL of bacteria solution and 5 μL of extract solution was mixed and added to the fourth quadrant. No solutions were added to the second quadrant (Figure 1).

| Figure 1. Picture of a petri dish divided into 4 quadrants corresponding to the explained paragraph. |
|---|
As preparation, the solutions were added using a micropipette onto the material and then spread using a spreader glass (Figure 2), whilst applying uniform pressure on the surface in correspondence to the labeled quadrants. Then, the prepared surface was inoculated via imprinting onto the culture media. The imprinted material was removed using tape to preserve the characteristic of the agar. After the inoculation, the petri dish was incubated for two days. The image was taken via iPhone 14th generation with the light switched off to decrease the shadow and self-reflection from the light. The colony was counted via ImageJ, an image processing program developed by the National Institutes of Health which is widely used among research involving counting colonies (Cai et al., 2011).

| Figure 2. Process of bacterial inoculation on the petri dish with the glass spreader. |
|---|
Results
Extracts of the Lamiaceae spp. and bacterial growth
As shown by the table (Table 2), clear zones appeared on the plate when O. basilicum, S. rosmarinus, T. vulgaris, and O. vulgare were tested with P. aeruginosa, showing that Lamiaceae spp. involved in this experiment have the ability to inhibit the growth of P. aeruginosa.
Table 2
| Radius of Clear Zone presented by Extracts and Bacteria | |||
| Herb Extract | 1 g/ml | ||
| P. Aeruginosa | E. Carotovora | S. Typhimurium | |
| O. basilicum | 1 | – | – |
| S. rosmarinus | 1.7 | – | – |
| T. vulgaris | 1 | – | – |
| O. vulgare | 1.7 | – | – |
Table 2. Table illustrating the radius of clear zones when herb extracts including Ocimum basilicum, Salvia rosmarinus, Thymus vulgaris, and Origanum vulgare were tested with Pseudomonas aeruginosa, Erwinia carotovora, and Salmonella typhimurium. The clear zones were presented when the extracts were tested on P. aeruginosa but cannot be found when combining the extracts with E. carotovora, and S. typhimurium.
‘Antimicrobial Activity by Disc Diffusion Methods.’ Interpreting this figure, ‘1 mm’ indicates the radius of the clear zone.
Metallic surfaces
However, the opposite was observed when the extracts were tested with E. carotovora and S. typhimurium. The interactions did not form a clear zone. Thus, we suspected that either the microorganisms required different environmental factors such as the amount of time and temperature they were kept in, or the extracts of Lamiaceae spp. used in this experiment do not have an effect on the selected bacterial species.
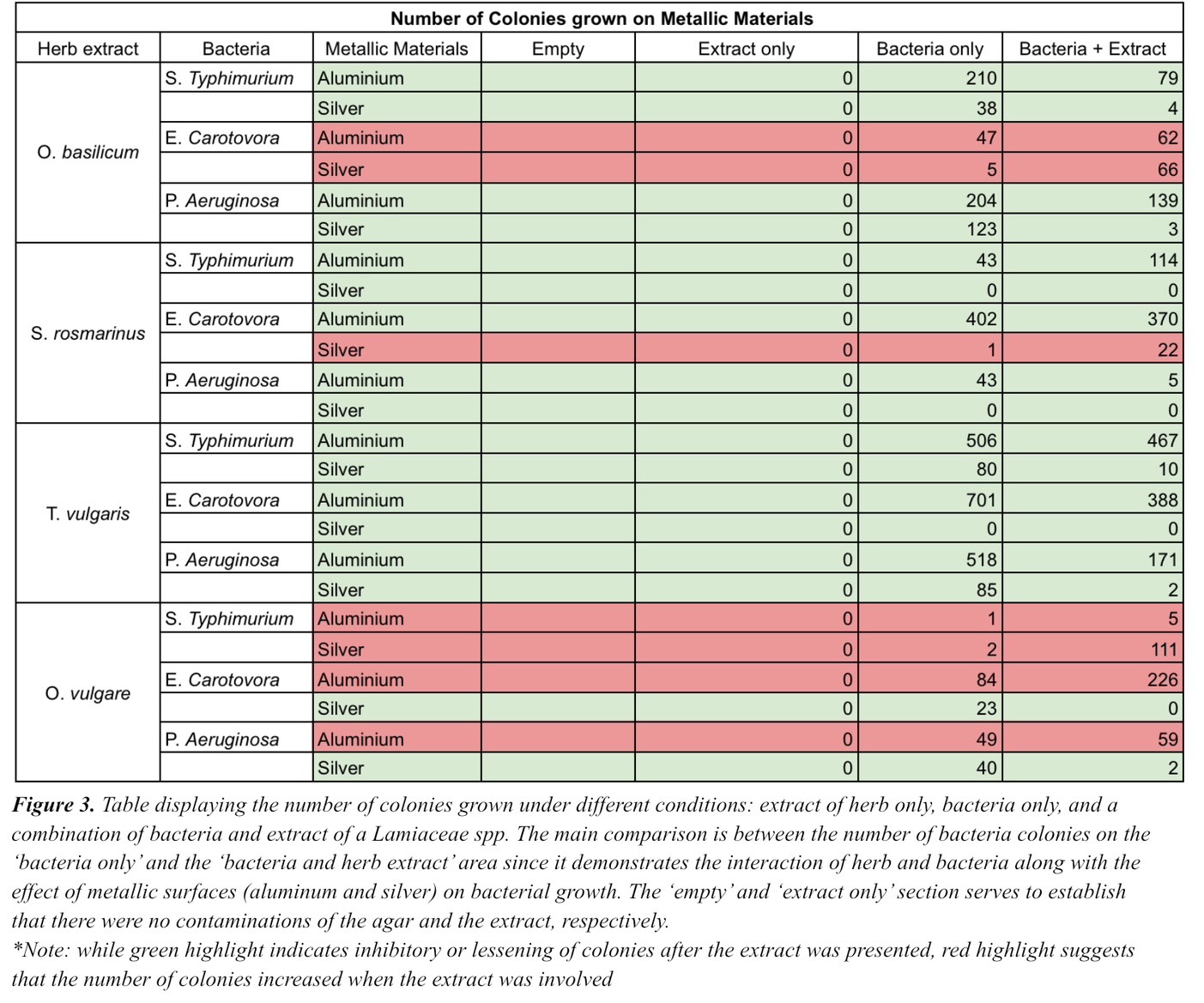
Table 3. Table displaying the number of colonies grown under different conditions: extract of herb only, bacteria only, and a combination of bacteria and extract of a Lamiaceae spp. The main comparison is between the number of bacteria colonies on the ‘bacteria only’ and the ‘bacteria and herb extract’ area since it demonstrates the interaction of herb and bacteria along with the effect of metallic surfaces (aluminum and silver) on bacterial growth. The ‘empty’ and ‘extract only’ section serves to establish that there were no contaminations of the agar and the extract, respectively.*Note: while green highlight indicates inhibitory or lessening of colonies after the extract was presented, red highlight suggests that the number of colonies increased when the extract was involved.
The metallic surfaces including aluminum and silver, treated with serially diluted bacteria and extracted herb, were stamped on the agar and left in the incubator for two days to test the relationship with bacteria—P. Aeruginosa, E. Carotovora, and S. Typhimurium—and to investigate the effect Lamiaceae spp. had on bacterial growth when they were applied in conjunction to metallic surfaces. As shown in Figure 3, despite increasing the number of colonies in some cases, a combination of metallic surfaces and Lamiaceae spp. proved to be effective in reducing the number of bacteria colonies grown in majority. Thus, bacterial growth can be inhibited by establishing metallic surfaces together with extracts of Lamiaceae spp.
Non-metallic surfaces
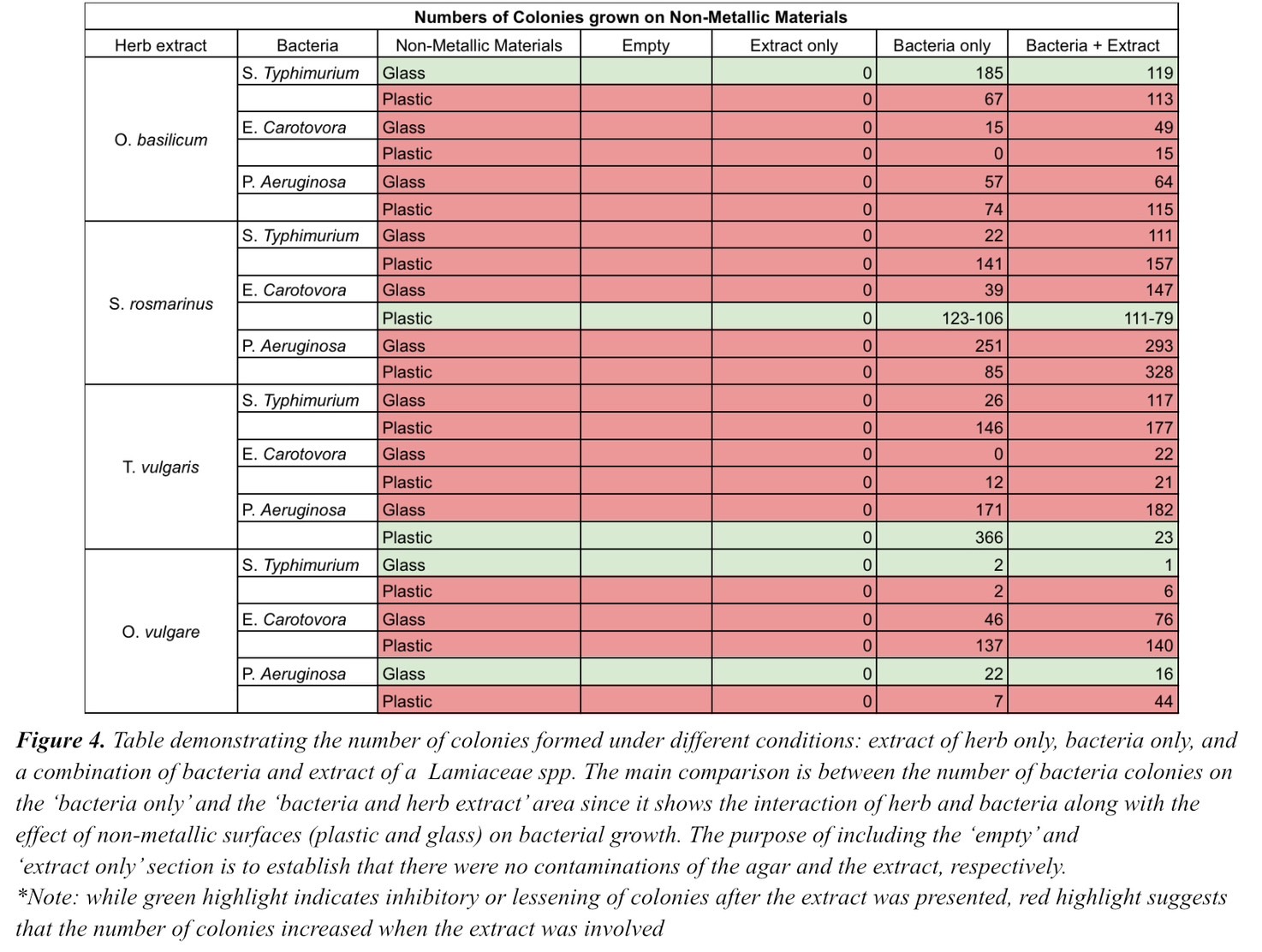
Non-metallic surfaces including plastic and glass were also being tested using the same procedures as the metallic surfaces. However, unlike the combination of metallic surfaces and Lamiaceae spp. extracts, when these non-metallic surfaces were used in conjunction with the extracts, the number of colonies appeared to increase. This suggests that when Lamiaceae spp. are applied on non-metallic surfaces, they either accelerate the bacterial growth or do not have any effect.
Discussions
We encountered several challenges that affected the accuracy and reproducibility of our results during the course of our research. One significant challenge was the potential for human error during various stages of the experiment, including sample preparation, inoculation, and colony counting. Contamination of samples that involved the growth of microorganisms was also a concern that may have affected the reliability of our results. Another challenge was the limited time available to work with our prepared solutions, as we hypothesized that their microbial-associated properties could degrade over time. In order to ensure that the lowest number of risk factors were present, we had to prepare new solutions every seven days. This posed a challenge as the process of extracting the herbs was time-consuming, which increased the likelihood of experimental discrepancy. Lastly, the equipment used in our experiments such as agar plates and glass instruments were fragile and prone to damage or malfunction, which could affect the reproducibility of our results. Precise measurement of volumes was often difficult, further contributing to possible inconsistencies.
Table 4. Table demonstrating the number of colonies formed under different conditions: extract of herb only, bacteria only, and a combination of bacteria and extract of a Lamiaceae spp. The main comparison is between the number of bacteria colonies on the ‘bacteria only’ and the ‘bacteria and herb extract’ area since it shows the interaction of herb and bacteria along with the effect of non-metallic surfaces (plastic and glass) on bacterial growth. The purpose of including the ‘empty’ and ‘extract only’ section is to establish that there were no contaminations of the agar and the extract, respectively.*Note: while green highlight indicates inhibitory or lessening of colonies after the extract was presented, red highlight suggests that the number of colonies increased when the extract was involved.
To address several challenges, we encountered during our research, there are potential improvements that could be implemented. To reduce contamination while working with microorganisms, a laminar flow cabinet should be used during processes that involve dexterity to minimize as many risks as possible. However, due to several limitations including budget restrictions, we could not implement this policy. Another potential improvement could involve developing a new process that preserves the antimicrobial activity of our solutions, while still ensuring that they are sterile. We could possibly explore alternative methods for sterilization that are less prone to degrade the properties of our herb extracts. Finally, changing the solvent used in our experiment could attract more solute and increase the efficiency of the extraction process. For instance, hexane could be used instead of water, as it is less polar and could yield a higher quantity of non-polar compounds (Reichardt & Welton, 2011).
Next steps
Due to the significant time required and financial restrictions, we were unfortunately able to perform our experiment once. Although each procedure was carefully planned and executed, conducting more trials would increase the consistency of the results and strengthen the reliability of the conclusion. Therefore, to further develop our project, more trials could be performed. Furthermore, other herbs such as echinacea, sage, or clove, which are known to be antimicrobial compounds, can also be explored using the same procedures.
Author contributions
W.W. and N.P. conducted background research. W.W. designed experiment. C.C., A.I., and P.T. produced the video. P.T. edited the video. All authors performed the experiment. P.K., J.T., and N.P. wrote the abstract which W.W. edited. P.K., J.T., W.W., and N.P. wrote and reviewed the manuscript. All authors collected and analyzed the data. N.V. constructed the manuscript graphics. N.V. completed the references.
Acknowledgements
We would like to express our heartfelt gratitude and utmost respect for Assist. Prof. Dr. Somkiat Phornphisutthimas, our mentor, for his unwavering support and guidance throughout this project. We also greatly appreciate the Alumni of Patumwan Demonstration School’s Medical Research Students Association, our seniors, Ajarn Jason Ping Zhang who introduced us toBiotreks, and Ajarn Sakkarin Achimar for the crucial initial guidance respectively.”
References
Santander, R. D., & Biosca, E. G. (2017). Erwinia amylovora psychrotrophic adaptations: Evidence of pathogenic potential and survival at temperate and low environmental temperatures. PeerJ, 5, e3931. https://doi.org/10.7717%2Fpeerj.3931
Cai, Z., Chattopadhyay, N., Liu, W. J., Chan, C., Pignol, J., & Reilly, R. M. (2011). Optimized digital counting colonies of clonogenic assays using ImageJ software and customized macros: Comparison with manual counting. International Journal of Radiation Biology, 87(11), 1135–1146. https://doi.org/10.3109/09553002.2011.622033
Carmona, F., & Pereira, A. M. S. (2013). Herbal medicines: Old and new concepts, truths and misunderstandings. Revista Brasileira de Farmacognosia, 23(2), 379-385. https://doi.org/10.1590/S0102-695X2013005000018
Carović-StanKo, K., PeteK, M., Grdiša, M., Pintar, J., Bedeković, D., Ćustić, H., & Satovic, Z. (2016). Medicinal plants of the family Lamiaceae as functional foods–A review. Czech Journal of Food Sciences, 34(5), 377-390. https://doi.org/10.17221/504/2015-CJFS
Dar, A. H., & Nayik, G. A. (Eds.). (2022). Nanotechnology interventions in food packaging and shelf life.
EHA Consulting Group. (n.d.). What is Pseudomonas aeruginosa?. https://www.ehagroup.com/resources/pathogens/pseudomonas-aeruginosa/
Elbehiry, A., Marzouk, E., Abalkhail, A., El-Garawany, Y., Anagreyyah, S., Alnafea, Y.,Almuzaini, A. M., Alwarhi,W., Rawway, M., & Draz, A. (2022). The development of technology to prevent, diagnose, and manage antimicrobial resistance in healthcare-associated infections. Vaccines, 10(12), 2100. https://doi.org/10.3390%2Fvaccines10122100
Firenzuoli, F., & Gori, L. (2007). Herbal medicine today: Clinical and research issues. Evidence-based Complementary and Alternative Medicine, 4, 37-40. https://doi.org/10.1093/ecam/nem096
Giannella, R. A. (1996.). Salmonella. In S. Baron. (Ed.), Medical Microbiology. University of Texas Medical Branch. NCBI. Retrieved April 30, 2023, from https://www.ncbi.nlm.nih.gov/books/NBK8435/
Kramer, A., Schwebke, I., & Kampf, G. (2006). How long do nosocomial pathogens persist on inanimate surfaces? A systematic review. BMC Infectious Diseases, 6(1), 1-8. https://doi.org/10.1186%2F1471-2334-6-130
Kim, S. H., Kim, K. Y., Yu, S. N., Park, S. G., Yu, H. S., Seo, Y. K., & Ahn, S. C. (2016). Monensin induces PC-3 prostate cancer cell apoptosis via ROS production and Ca2+ homeostasis disruption. Anticancer Research, 36(11), 5835-5843. https://doi.org/10.21873/anticanres.11168
Kakani, S. L. (2004). Material science. New Age International Publishers.
Nielsen, K. (n.d.). Silver. A powerful weapon against microbes. Coloplast. https://www.coloplast.com/products/wound/articles/silver-a-powerful-weapon-against-microbes/
Özcan, M. M., & Chalchat, J. C. (2008). Chemical composition and antifungal activity of rosemary (Rosmarinus officinalis L.) oil from Turkey. International Journal of Food Sciences and Nutrition, 59(7-8), 691-698. https://doi.org/10.1080/09637480701777944
Parham, S., Kharazi, A. Z., Bakhsheshi-Rad, H. R., Nur, H., Ismail, A. F., Sharif, S., RamaKrishna, S., & Berto, F. (2020). Antioxidant, antimicrobial and antiviral properties of herbal materials. Antioxidants, 9(12), 1309. https://doi.org/10.3390/antiox9121309
Patel, R., Rinker, L., Peng, J., & Chilian, W. M. (2017). Reactive oxygen species: The good and the bad. In C. Filip & E. Albu (Eds.), Reactive oxygen species (ROS) in living cells. https://doi.org/10.5772/intechopen.71547
Reichardt, C., & Welton, T. (2011). Solvents and solvent effects in organic chemistry. John Wiley & Sons.
Sun, D., Babar Shahzad, M., Li, M., Wang, G., & Xu, D. (2014). Antimicrobial materials with medical applications. Materials Technology, 30(sup6), B90-B95. https://doi.org/10.1179/1753555714Y.0000000239
Suppakul, P., Miltz, J., Sonneveld, K., & Bigger, S. W. (2003). Antimicrobial properties of basil and its possible application in food packaging. Journal of Agricultural and Food Chemistry, 51(11), 3197-3207. https://doi.org/10.1021/jf021038t
Teixeira, B., Marques, A., Ramos, C., Serrano, C., Matos, O., Neng, N. R., Nogueira, J. M., Saraiva, J. A., & Nunes, M. L. (2013). Chemical composition and bioactivity of different oregano (Origanum vulgare) extracts and essential oil. Journal of the Science of Food and Agriculture, 93(11), 2707-2714. https://doi.org/10.1002/jsfa.6089
Tzima, K., Makris, D., Nikiforidis, C. V., & Mourtzinos, I. (2015). Potential use of rosemary, propolis and thyme as natural food preservatives. The Korean Journal of Nutrition, 1(6). https://doi.org/10.13188/2469-4185.1000002
Vassiliou, E., Awoleye, O., Davis, A., & Mishra, S. (2023). Anti-inflammatory and antimicrobial properties of Thyme oil and its main constituents. International Journal of Molecular Sciences, 24(8), 6936. https://doi.org/10.3390/ijms24086936
Wick, R., & Hansen, M. A. (2011b, August 5). Nutrient agar. BugwoodWiki. https://wiki.bugwood.org/Nutrient_agar Wick, R., & Hansen, M. A. (2011a, August 5). Nutrient broth. BugwoodWiki. https://wiki.bugwood.org/Nutrient_broth