Napasjutha Kongsonthana andWorathas Wongsatidporn
Patumwan Demonstration School, Bangkok, Thailand
Reviewed on 7 May 2022; Accepted on 25 June 2022; Published on 15 October 2022
With help from the 2022 BioTreks Production Team.
Recent developments in spectroscopic technologies, which exploit light-matter interaction to identify samples’ species, have extended the frontier of diagnosis and treatment in medicine. In particular, surface-enhanced Raman spectroscopy (SERS), which integrates nanotechnology with Raman spectroscopy, has increased the biomarker detection sensitivity to the point that detection of a single molecule is possible. This capability could be used to detect and diagnose diseases earlier, which entails increased survival chances and better disease progression prevention. This paper aims to highlight the potential of SERS in the field of medicine; it discusses the fundamental SERS mechanism, its application in diagnosing both infectious and non-communicable diseases, and provides a perspective on its current and potential application of SERS in medicine.
Keywords: Surface-enhanced Raman spectroscopy, medical applications, early diagnosis
Authors are listed in alphabetical order. Jason Ping Zhang mentored the group. Please direct all correspondence to jason.zha@satitpatumwan.ac.th.
In modern medicine, the ability to diagnose illnesses correlates strongly with the ability to detect and identify biomarkers.Prominent developments in the field of communicable diseases have been focused on increasing the quantity of biomarkers to reach detectable levels through processes such as growing cell cultures and Polymerase Chain Reaction (PCR). However, there is still room for improvement. For example, on average, it takes 24 hours for PCR results (Cleveland Clinic, 2021) and up to five days for bacterial culture tests (MedlinePlus, 2020). This is problematic, particularly for patients in time-sensitive conditions. Without an immediate diagnosis, those patients would be prescribed broad-spectrum antibiotics, contributing to antibiotic resistance, which threatens the lives of both the patients and others. Early detection is a vital step to an increased chance of successful treatment and alleviating symptoms of diseases. Not only that, treating diseases in their early stages requires fewer resources, which potentially saves cost and increases resource usage efficiency. It is apparent that there is a lot to be gained in developing a more sensitive analytical approach; one promising candidate, Surface-enhanced Raman spectroscopy (SERS), shall be discussed in this paper.
Surface-enhanced RAMAN spectroscopy (SERS)
SERSis an analytical method based on Raman spectroscopy with the addition of metallic nanostructure on the substrate surface, enhancing Raman scattering—the underlying phenomenon behind Raman spectroscopy. The surface enhancement exploits the Localized Surface Plasmon (LSP)—a phenomenon that occurs when light interacts with structures with lower dimensions than its wavelength, inducing strong electron oscillations (Badshah et al., 2020). In brief, the extent of surface enhancement is heavily dependent on the nanostructure (i.e., SERS active sites and electronic mechanism enhancement factor) (Stiles et al., 2008).
Indeed, the development of SERS has been enabled by developments in nanotechnology–not only in pursuing higher enhancement factors but also in expanding our understanding of SERS. Previously, SERS was a qualitative phenomenon replicated by imprecise rudimentary structures such as metallic thin film; advances in nanofabrication have allowed for greater control of the nanoarchitectures responsible for LSP (Stiles et al., 2008). This has narrowed down the variables in studying the phenomenon and increased our understanding, enabling future developments in various fields, particularly in medicine.
Mechanism
Raman spectroscopy exploits the different fingerprints of molecules owing to the unique characteristics of their chemical bonds, which affects the inelastic scattering of photons. It follows a fundamental principle of light-matter interaction: When light interacts with matter, the frequency of scattered radiation differs from that of incident radiation. Inelastic scattering of incident radiation attributes to the Raman effect and is unique for different samples. The Raman spectrum is constructed following the alteration in frequency of light, which occurs when incident radiation collides with molecules in a sample and is analyzed to identify the sample species (Bumbrah et al., 2016). In simple words, analyte species could be determined via a Raman shift (change in frequency observed in scattered light) of certain analytes. Raman signals are naturally weak. Given the low number of scattered photons, detection of a change in a signal can be difficult. As such, the SERS method is employed to amplify Raman signals through the use of metallic nanoparticles, typically gold (Au) or silver (Ag), on substrate surfaces (Fig.1) to better determine the species of the analyte.
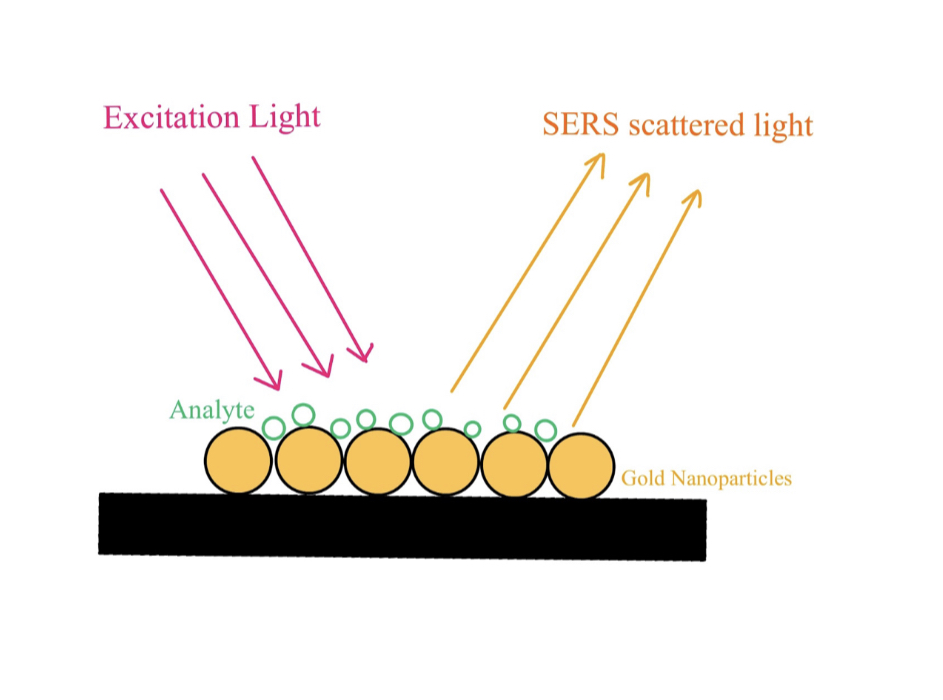
| Figure 1: Raman signal enhancement through Au nanoparticles |
|---|
Although fundamentals behind gravity might not be fully understood, we built countless inventions that utilize gravity. Likewise, the exact underlying mechanism of LSP, which enables SERS, is still a matter of debate. Even so, SERS has paved the way for a new frontier in biosensors with its demonstrated capability in single molecule detection (Pérez-Jiménez et al., 2020; Mayer & Hafner, 2011). There are a myriad of usages with such capability, but among them, the field of medicine is undoubtedly one of the most prominent.
Application in medicine
SERS application in detection of biomarkers of infectious diseases
Infectious diseases are caused by organisms, such as bacteria, viruses, fungi, or parasites. They can be transmitted from person to person, vectors, and in contaminated environments. Globally, infectious diseases are a major cause of death, particularly in low-income countries and among young children (Baylor College of Medicine, n.d). In 2019, three of the ten leading causes of death were infectious diseases, with lower respiratory infections being the most fatal (World Health Organization, 2020). Other common infectious diseases include HIV, influenza, sexually transmitted diseases, measles, and pneumonia.
HIV
Human Immunodeficiency virus (HIV) is one of the most severe infectious diseases that could be fatal if not treated. HIV refers to retroviruses that attack the body’s immune system, which could lead to Acquired Immunodeficiency Syndrome (AIDS), a life-threatening stage of HIV infection. In the status quo, there is virtually no permanent cure for the disease (Centers for Disease Control and Prevention, 2021). However, early diagnosis and treatment lead to increased survival chances. In 2016, Fu and his colleagues were able to detect HIV-1 viral strain through the utilization of a SERS-based lateral flow assay. Ramen reporter-labeled gold nanoparticles (AuNPs) were used as nanotags for detecting HIV-1 DNA, a biomarker, in lateral flow strips and quantitative analysis of low-concentration reagents were achieved. They were able to target and detect HIV-1 DNA under the limit of 0.24 pg/mL, which was at least 1,000 times more sensitive than other detection assays (Fu et al., 2016).
Influenza
Another disease-causing virus that SERS-based assay and aptasensor technology was able to detect was the influenza virus. In 2019, Kukushkin and his colleagues developed the SERS-based aptasensor, which was able to recognize multiple influenza virus strains, including H1, H3, and H5 hemagglutinin subtypes, using RHA0385 aptamers. Primary aptamers, influenza viruses, and secondary aptamers were arranged in a sandwich model to achieve high sensitivity. Change in signal was observed within primary and secondary aptamers. Normally, about 10-1 – 10-3 hemagglutination units (HAU) per sample are required for aptamer-based approaches. In comparison, the limit of SERS detection was 104 virus particles per sample, 10-4 HAU per sample, which was significantly lower than the conventional technique. Moreover, the time required for the whole analysis was only 12 minutes (Kukushkin et al., 2019).
SERS application in detection of Biomarkers of non-communicable diseases
Non-communicable diseases (NCDs), or chronic diseases cannot be transmitted from one person to another and tend to last for a long period. According to the World Health Organization, NCDs kill approximately 41 million people per year and more than fifty percent of mortalities are from low to middle-income countries (World Health Organization, 2021). Examples of serious NCDs include cardiovascular diseases, cancer, respiratory diseases, diabetes, and Alzheimer’s disease. Detection, screening, and treatment are key components to dealing with NCDs.
Cancer
In recent years, the usage of exosomes as biomarkers for cancer detection has gained rising popularity. It contains significant information about the pathological process of a disease. While normal people have approximately 2000 trillion exosomes, patients with cancer have about 4000 trillion exosomes (Zhong et al., 2021), as tumor cells can secrete more exosomes than normal cells. Thus, detection of cancer-related exosomes is helpful in the rapid screening of patients suspected of having cancer. A study by Zhang et al. (2019) has demonstrated the potential of SERs for rapid, label-free exosome detection. In their study, SERS technology was employed to detect different types of cancer cells based on their unique raman phenotypes. Gold nanoparticle colloids were exploited to enhance Raman signals in their experiment. They reached sensitivity and specificity greater than 95% and an accuracy of 96.7% for classifying the eight exosomes collected from different cell lines (Zhang et al., 2019). Moreover, in 2019, Moisoiu et al. (2019) utilized SERS for urine analysis to distinguish between patients with breast cancer and healthy patients. The study employed surface-enhanced Raman scattering (SERS) enhanced using silver nanoparticles for analyzing urine from breast cancer patients and controls. Using linear discriminant analysis (PCA-LDA), a sensitivity of 81%, a specificity of 95%, and an overall accuracy of 88% were achieved (Moisoiu et al., 2019).
Alzheimer’s disease
Alzheimer’s disease (AD) is the most common type of dementia and is regarded as another fatal chronic disease found commonly in people aged 65 or older. AD is a neurodegenerative disease that causes progressive loss of memory and cognitive skills, mood swings, and language disorders (Centers for Disease Control and Prevention, 2020). Three primary disease markers that are detectable via SERs analysis are tau protein, β-amyloid peptide (Aβ), and cysteine (Lin et al., 2020). In 2020, Park et al. (2020) introduced a more efficient way of measuring SERS spectra of tau protein and β-amyloid peptide using carboxylic-acid-functionalized and graphitic nanolayer-coated three-dimensional SERS substrate (CGSS), which allows secondary structural change to be analyzed with an enhancement factor of 5.5 x 105 (Park et al., 2020).
More recently, tears have been used as AD biomarkers. In a study by Cennamo et al. (2020), SERS analysis of tears collected from subjects with AD, ascertained mild-cognitive symptoms, and healthy subjects showed differences in various spectral regions, specifically protein-concentrated regions. Alteration in tear composition provided information about the pathological process, which allowed classification of healthy, AD, and mild-cognitive symptoms to be performed (Cennamo et al., 2020).
Perspective
As basic health infrastructure develops globally, a trend emerges: the number of deaths caused by non-communicable diseases such as stroke, ischaemic heart disease, and Alzheimer’s disease has been on the rise, with stroke and ischaemic heart disease increasing at a concerning rate. At the same time, deaths from communicable diseases are decreasing (World Health Organization, 2020). Despite that, since communicable diseases are still very destructive in developing nations, and major causes of death worldwide are incurable, chronic diseases, there is a demand for a more sensitive test for detecting both communicable and non-communicable diseases at earlier stages than the status quo has never been more paramount.
Mobile, potentially low-cost yet highly sensitive test kits
With the existence of portable Raman spectrometers, the advantages of SERS could be brought to areas where conventional high-sensitivity tests could not be performed. Wildlife disease screening—where biological samples would typically be shipped to a lab for testing and are at risk of transportation failure—was one of the cases with a high demand for portable diagnostic probes. Moreover, recent developments in SERS substrate have proved the potential of manufacturing SERS substrate at relatively low cost. Since the SERS signal is greatly dependent on LSPR on the surface of metallic nanostructures, namely gold and silver, to achieve a highly stable SERS signal, a complicated and time-consuming substrate fabrication process involving expensive instruments and specially-trained professionals is presupposed. This limits SERS’ application to the lab and significantly hinders its practice in the real-world context. However, current findings on SERS substrates synthesis using alternative fabrication techniques have provided the promise of cost-and-time-effective substrates. For instance, a simple, low-cost and less time-consuming Ag-Pt based SERS substrate was successfully fabricated using a galvanic placement method; equipment and reagents used in the method were readily available in the market (Wang et al., 2018). In addition, the initial cost of Raman spectrometers would not be problematic in the long run, as they could be reused multiple times investigating outbreaks or screening a community entails large scale testing. SERS-based tests could upgrade rural area disease testing where lateral flow tests are typically used; offering higher sensitivity with similar costs.
Lowering costs of SERS substrate
SERS substrates do not undergo chemical change with testing, suggesting that SERS substrates could be reused. In fact, there has been a success at a laboratory scale in reusing SERS substrates (Das et al., 2019; Oliveira et al., 2021; Zu et al., 2020). However, that potential is still unutilized as all production-scale SERS on the market (Ocean Insight, Nanova Q-SERS, P-SERS Metrohm, SERSitive, SLIMECO, Power SERS, and ONSPEC) are predominantly single-use disposables, and those that aren’t possesses low enhancement factor. More research on substrate cleaning could potentially be disruptive to the industry, particularly in terms of price. Furthermore, in principle, substrate cleaning could be done without sophisticated protocols as nanostructures supporting SERS are typically metals while the analytes are typically biological. Selective cleaners could be used to clean SERS substrates. An alternative for cleaning more structurally complex molecules off the substrate, such as carbohydrates,could be to use enzymes. Furthermore, in the case of proteins, the addition of heat triggering denaturation could facilitate enzyme breakdown—in this case, the breakdown product would not need to be thoroughly cleaned, as it would have a different Raman fingerprint compared to its original molecule.
The authors propose a plasma treatment protocol which has also been used to remove organic contamination on surfaces with great success (Banerjee et al., 2010). However, due to the variation of analytes, use cases, and SERS substrates’ material and structure—the cleaning protocol is most likely unique for each of the variations. Fortunately, with plasma treatment, various parameters could be tweaked to enhance the selectivity of the removal process, for example, the type of gas, power, and duration.
The authors acknowledge that the reusability of SERS substrates might not be as popular in literature compared to the pursuit of higher sensitivity levels, a unique advantage of SERS, which holds massive commercial potential. Nevertheless, the reusability potential that SERS substrate possesses is still very interesting as it is inevitable to tackle cost as the technology develops.
Challenges
Currently, the production of SERS substrates is the main challenge to bringing the technology to the masses. As more sophisticated nanostructures are harder to produce on a large scale, the cost and production time of higher sensitivity substrates are not yet practical commercially. In addition, developing a single-molecule-detection SERS substrate requires a more advanced Raman spectrometer, which costs a lot more. For example, in Thailand, there are only a couple—mostly with limited access behind high-end laboratories—hindering the development speed.
Conclusion
SERS has illustrated high potential in medicine, particularly in disease diagnosis. It has shown its potency as both a stand-alone and SERS-integrated diagnostic assay. A variety of biomarkers on which SERS methods are applicable widens the room for disease diagnosis and proves the potential of SERS serving as a non-invasive diagnostic method. On top of that, substrates used in SERS have exhibited a high potential of being low-cost and reusable. When practical, this potential would essentially aid low to middle-income countries, which tend to have high mortality rates from both communicable and non-communicable diseases and cannot access high-cost diagnostic techniques, especially for populations in remote or rural areas.
Considering these characteristics of SERS, SERS has distinctive competitive advantages compared to other diagnostic methods. However, at the present, the literature on SERS has mostly been proof-of-a-concept. Therefore, its application in clinical settings has to be further investigated.
Next steps
More research needs to be done on the production of large scale nanopatterns. Among the various approaches to nanofabrication, convective deposition stands out as its cost is relatively low. However, advanced equipment, especially the motor, is very costly, thus further investigation into this might be out of reach for high school students.
Another approach could be to look more into the reusability potential of the SERS substrate. If the substrates could be reused multiple times, a large quantity might not be needed. With this approach, there is a need for research on the optimal protocol for substrate cleaning for the various different analytes. This is a more plausible step for high school students, as substrates should not be hard to secure from a lab, as typically, they would be thrown away.
Author contributions
W.W. and N.K. both planned out the paper and brainstormed perspectives. W.W. was mainly responsible for the following sections: abstract, introduction, Surface-enhanced Raman spectroscopy, perspective, challenges, and next steps. N.K. was mainly responsible for the sections of mechanism, application in medicine and conclusion.
Acknowledgements
We would like to express gratitude to Dr. Jason Ping Zhang who has been assisting us throughout the whole writing process and our cute pets for emotional support. Also, we would like to thank Patumwan Demonstration School’s Medical Research Students Association for introducing BioTreks to us.
References
Badshah, M. A., Koh, N. Y., Zia, A. W., Abbas, N., Zahra, Z., & Saleem, M. W. (2020). Recent developments in plasmonic nanostructures for metal enhanced fluorescence-based biosensing. Nanomaterials, 10(9), 1749. https://doi.org/10.3390/nano10091749
Banerjee, K. K., Kumar, S., Bremmell, K. E., & Griesser, H. J. (2010). Molecular-level removal of proteinaceous contamination from model surfaces and biomedical device materials by air plasma treatment. Journal of Hospital Infection, 76(3), 234–242. https://doi.org/10.1016/j.jhin.2010.07.001
Bumbrah, G. S., & Sharma, R. M. (2016). Raman spectroscopy – Basic principle, instrumentation and selected applications for the characterization of drugs of abuse. Egyptian Journal of Forensic Sciences, 6(3), 209–215. https://doi.org/10.1016/j.ejfs.2015.06.001
Cennamo, G., Montorio, D., Morra, V. B., Criscuolo, C., Lanzillo, R., Salvatore, E., Camerlingo, C., Lisitskiy, M., Delfino, I., Portaccio, M., & Lepore, M. (2020). Surface-enhanced Raman spectroscopy of tears: Toward a diagnostic tool for neurodegenerative disease identification. Journal of Biomedical Optics, 25(8), 1-12. https://doi.org/10.1117/1.jbo.25.8.087002
Centers for Disease Control and Prevention. (2021, June 1). About HIV/AIDS. Centers for Disease Control and Prevention. Retrieved April 27, 2022, from https://www.cdc.gov/hiv/basics/whatishiv.html
Centers for Disease Control and Prevention. (2020, October 26). Alzheimer’s disease and related dementias: What is alzheimer’s disease? Centers for Disease Control and Prevention. Retrieved May 5, 2022, from https://www.cdc.gov/aging/aginginfo/alzheimers.htm
Baylor College of Medicine, Department of Molecular Virology and Microbiology. (n.d) Introduction to infectious diseases. https://www.bcm.edu/departments/molecular-virology-and-microbiology/emerging-infections-and-biodefense/introduction-to-infectious-diseases
Das, D., Senapati, S., & Nanda, K. K. (2019). Rinse, repeat: An efficient and reusable SERS and catalytic platform fabricated by controlled deposition of silver nanoparticles on cellulose paper. ACS Sustainable Chemistry & Engineering, 7(16), 14089–14101. https://doi.org/10.1021/acssuschemeng.9b02651
Fu, X., Cheng, Z., Yu, J., Choo, P., Chen, L., & Choo, J. (2016). A SERS-based lateral flow assay biosensor for highly sensitive detection of HIV-1 DNA. Biosensors and Bioelectronics, 78, 530–537. https://doi.org/10.1016/j.bios.2015.11.099
Kukushkin, V. I., Ivanov, N. M., Novoseltseva, A. A., Gambaryan, A. S., Yaminsky, I. V., Kopylov, A. M., & Zavyalova, E. G. (2019). Highly sensitive detection of influenza virus with SERS aptasensor. PLoS One, 14(4), e0216247. https://doi.org/10.1371/journal.pone.0216247
Lin, T., Song, Y.L., Liao, J., Liu, F., & Zeng, T.T. (2020). Applications of surface-enhanced Raman spectroscopy in detection fields. Nanomedicine, 15(30), 2971–2989. https://doi.org/10.2217/nnm-2020-0361
Mayer, K. M., & Hafner, J. H. (2011). Localized surface plasmon resonance sensors. Chemical Reviews, 111(6), 3828–3857. https://doi.org/10.1021/cr100313v
Moisoiu, V., Socaciu, A., Stefancu, A., Iancu, S. D., Boros, I., Alecsa, C. D., Rachieriu, C., Chiorean, A. R., Eniu, D., Leopold, N., Socaciu, C., & Eniu, D. T (2019). Breast cancer diagnosis by surface-enhanced Raman scattering (SERS) of urine. Applied Sciences, 9(4), 806. https://doi.org/10.3390/app9040806
Oliveira, M. J., Cunha, I., de Almeida, M. P., Calmeiro, T., Fortunato, E., Martins, R., Pereira, L., Byrne, H. J., Pereira, E., Águas, H., & Franco, R. (2021). Reusable and highly sensitive SERS immunoassay utilizing gold nanostars and a cellulose hydrogel-based platform. Journal of Materials Chemistry B, 9(36), 7516–7529. https://doi.org/10.1039/d1tb01404h
Cleveland Clinic. (2021, August 24). COVID-19 and PCR testing. Cleveland Clinic. Retrieved April 27, 2022, from https://my.clevelandclinic.org/health/diagnostics/21462-covid-19-and-pcr-testing
Park, H. J., Cho, S., Kim, M., & Jung, Y. S. (2020). Carboxylic Acid-Functionalized, Graphitic Layer-Coated Three-Dimensional SERS Substrate for Label-Free Analysis of Alzheimer’s Disease Biomarkers. Nano Letters, 20(4), 2576–2584. https://doi.org/10.1021/acs.nanolett.0c00048
Pérez-Jiménez, A. I., Lyu, D., Lu, Z., Liu, G., & Ren, B. (2020). Surface-enhanced Raman spectroscopy: Benefits, trade-offs and future developments. Chemical Science, 11(18), 4563–4577. https://doi.org/10.1039/d0sc00809e
Stiles, P. L., Dieringer, J. A., Shah, N. C., & van Duyne, R. P. (2008). Surface-enhanced Raman spectroscopy. Annual Review of Analytical Chemistry, 1(1), 601–626. https://doi.org/10.1146/annurev.anchem.1.031207.112814
NIH National Institute on Aging. (2021, July 8). What is alzheimer’s disease? U.S. Department of Health and Human Services. Retrieved April 27, 2022, from https://www.nia.nih.gov/health/what-alzheimers-disease
MedlinePlus. (2020, July 30). Bacteria culture test. U. S. National Library of Medicine. Retrieved April 27, 2022, from https://medlineplus.gov/lab-tests/bacteria-culture-test/
Wang, T., Zhou, J., & Wang, Y. (2018). Simple, low-cost fabrication of highly uniform and reproducible SERS substrates composed of Ag–Pt nanoparticles. Nanomaterials, 8(5), 331. https://doi.org/10.3390/nano8050331
World Health Organization. (2020, December 9). The top 10 causes of death. World Health Organization. Retrieved May 16, 2022, from https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death
World Health Organization. (2021, April 13). Non communicable diseases. World Health Organization. Retrieved April 27, 2022, from https://www.who.int/news-room/fact-sheets/detail/noncommunicable-diseases
Xu, J., Cheng, C., Shang, S., Gao, W., Zeng, P., & Jiang, S. (2020). Flexible, reusable SERS substrate derived from ZIF-67 by adjusting LUMO and HOMO and its application in identification of bacteria. ACS Applied Materials & Interfaces, 12(44), 49452–49463. https://doi.org/10.1021/acsami.0c15754
Zhang, P., Wang, L., Fang, Y., Zheng, D., Lin, T., & Wang, H. (2019). Label-free exosomal detection and classification in rapid discriminating different cancer types based on specific Raman phenotypes and multivariate statistical analysis. Molecules, 24(16), 2947. https://doi.org/10.3390/molecules24162947
Zhong, Y., Li, H., Li, P., Chen, Y., Zhang, M., Yuan, Z., Zhang, Y., Xu, Z., Luo, G., Fang, Y., & Li, X. (2021). Exosomes: A new pathway for cancer drug resistance. Frontiers in Oncology, 11. https://doi.org/10.3389/fonc.2021.743556



