Rebecca Rubiano. Siwen Yang, Yinmeng Li ▫ Western Reserve Academy, Hudson OH
Reviewed on 6 May 2023; Accepted on 13 July 2023; Published on 16 October 2023
With help from the 2023 BioTreks Production Team.
Harmful algae blooms negatively affect aquatic ecosystems and communities that rely on those bodies of water for drinking water. Many algal blooms contain cyanobacteria which produce harmful toxins. Many algal blooms, like those in Lake Erie, contain the cyanobacteria Microcystis aeruginosa which produces the toxin microcystin-LR. This toxin disrupts cellular pathways and inhibits the removal of phosphates. As a result, the toxin causes liver and kidney damage in humans which can lead to death. To combat the harmful effects of microcystin-LR produced from algal blooms, our group intends to target the toxin, microcystin-LR, in the aquatic environment. Our previous design of an enzyme spray containing mlrA, mlrB, mlrC, mlrE, and mlrF will be sprayed in affected ecosystems to degrade the toxin. These enzymes will be produced by inserting the enzyme genes isolated from Sphingomonas sp. and Sphingopyxis sp. into Escherichia coli which will mass produce the enzymes. In this study, our group grew Sphingomonas sp. PS-60 in an attempt to isolate the mlrA gene.
Keywords: microcystin-LR, mlrA, Sphingomonas sp. PS-60, algal blooms
Authors are listed in alphabetical order. Beth Pethel from Western Reserve Academy, Hudson OH mentored the group. Please direct all correspondence to .
Harmful algal blooms (HABs), which have been documented for centuries, affect many ecosystems (hab.whoi.edu, 2022). Lake Erie has warm, shallow waters and is surrounded by agricultural lands which create large amounts of fertilizer run-off into Lake Erie. These characteristics provide the perfect environment for algal growth (Ohio Sea Grant College, 2022). Algal blooms establish dangerous habitats by consuming oxygen, blocking sunlight, and producing detrimental toxins. Thus, algal blooms impact human health activity in Lake Erie’s surrounding communities by impacting the biological functions of domestic animals through drinking water containing cyanobacteria and increasing the chance of liver tumors in humans (Butler et al., 2009). In Toledo, Ohio, algal blooms in Lake Erie and the Maumee River poison the community that sources its water directly from the lake and the river (Rogener, 2020). In 2014, a combination of heavy winds and an HAB prompted citywide orders to pause the use of all forms of tap water for three days. The harsh winds caused waves of water to directly flow into water treatment facilities, raising the level of the algal blooms in Lake Erie to a quantity the World Health Organization deems toxic for humans. This halt in access to water was a significant burden on the eleven million people who depend on Lake Erie water (Ohio Sea Grant College, 2022).
Algal bloom toxicity originates from Microcystin-LR (MC-LR). Released by M. aeruginosa when its cell walls burst during death, MC-LR is a cyclic heptapeptide made up of seven amino acids: D-alanine (D-Ala), L-amino acid (L-Leu), D-Methylaspartic acid (D-Me Asp), L-amino acid (L-Arg), β amino acid (Adda), D-Glutamic acid (D-Iso Glu), and N-Methyl Dehydroalanine (Moha) (Cyanosite, n.d.). The stable structure of MC-LR makes it resistant to common chemical breakdown mechanisms such as hydrolysis or oxidation (Butler et al., 2009; Cyanosite, n.d.). MC-LR denatures enzymes, known as protein phosphatases, inhibiting them from performing common steps in many biological pathways. For example, it disrupts the process of removing phosphates from amino acids like serine and threonine, thus causing a build-up of phosphorylated protein, which induces cell apoptosis and, accordingly, liver cell death (Yoshida et al., 1997).
Sphingomonas bacteria, which reside primarily in soil and water, produce a set of enzymes that degrade MC-LR—MlrA, MlrB, MlrC, MlrD, MlrE, and MlrF—to obtain a carbon source (Savola, 2016). MlrA linearizes the cyclic nature of MC-LR through hydrolysis, breaking the peptide bond between β amino acid (ADDA) and L-arginine (L-Arg) to form a linearized product (Savola, 2016). Following this breakdown, MlrB, a serine protease, breaks apart the bond between the Leucine (L-Leu) and D-alanine amino acids to form a tetrapeptide (Massey & Yang, 2020). MlrC, a metalloprotease, further degrades the tetrapeptide into amino acids (Savola, 2016). MlrD aids in the transport of microcystin-LR into the cell (Massey & Yang, 2020). The specific functions of MlrE and MlrF are unknown, but they are critical to microcystin-LR degradation (Metzloff, 2019a; 2019b). We inserted genes for enzymes MlrA, B, C, E, and F into five different DH5α E. coli cultures optimized for plasmid production (MCLAB, n.d.; Figure 1). We did not include MlrD because this enzyme imports the toxin into a cell. Instead, the transformation will include the enzymes produced within DH5α E. coli to break down the microcystin toxin.
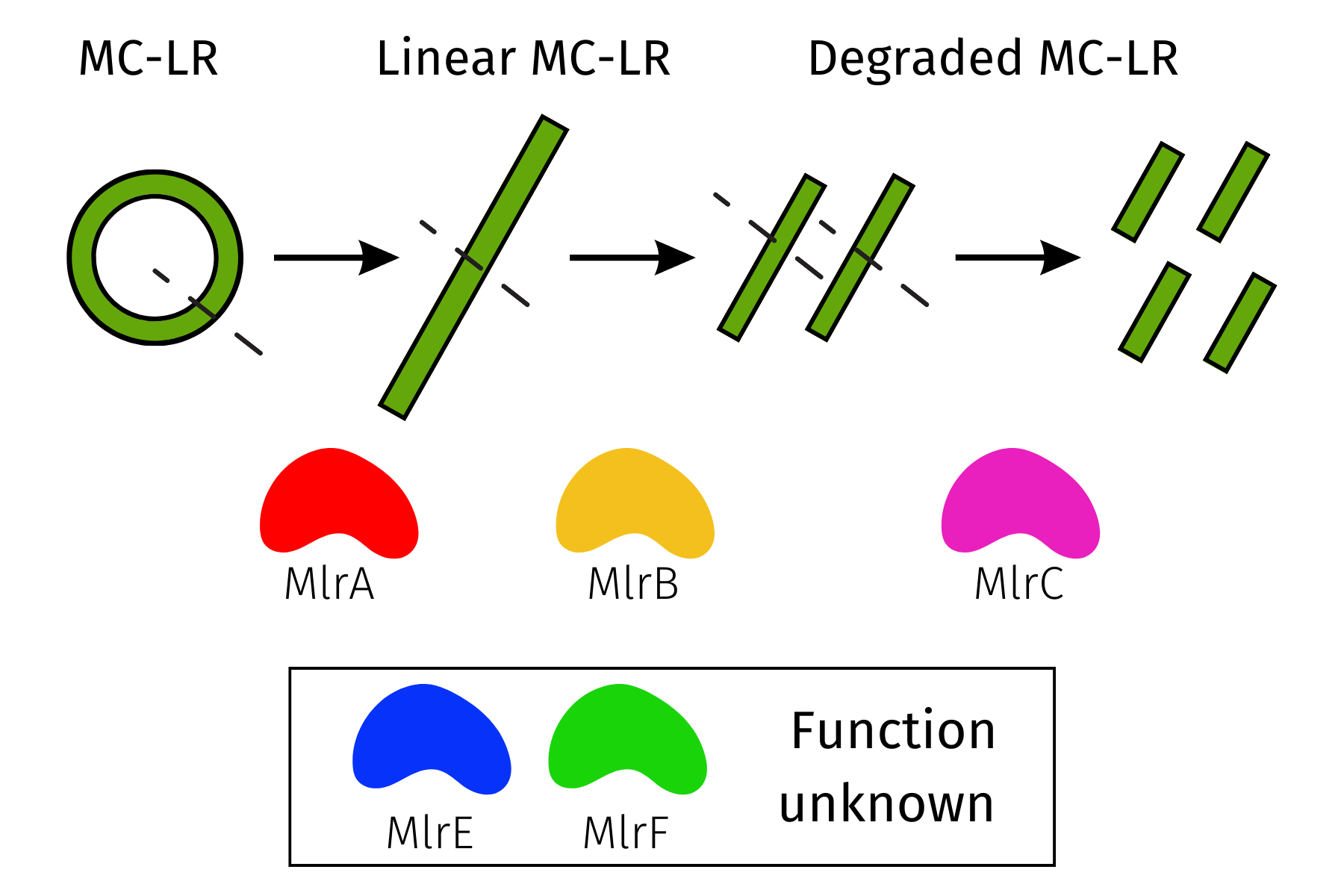
| Figure 1. The breakdown of microcystin-LR through MlrA, MlrB, and MlrC enzymes. The function of MlrE and MlrF enzymes is unknown. |
|---|
The project aims to mass-produce and purify the enzymes, allowing us to test them for optimal enzymatic activity against the toxin. We envision using these enzymes to directly remediate toxin-producing algal blooms and bolster filtration mechanisms in water treatment facilities. As past experiments observed successful heterologous expression of MlrA in E. coli (Lee et al., 2006), we designed our first experiment to transform MlrA from Sphingomonas sp. PS-60 into E. coli. The experiment will determine the presence of the MlrA gene in Sphingomonas sp. PS-60. Unfortunately, we were unable to isolate the MlrA gene from Sphingomonas sp. PS-60. Instead, we designed our experiment to transform MlrB from Sphingosinicella microcystinivorans Y2 into E. coli.
Materials and methods
Overview
The final goal of our project was to extract the genes encoding for the Microcystin-degrading enzymes—mlrA, mlrB, mlrC, mlrE, and mlrF––and move them into DH5α E. coli, which we tested for its ability to decompose toxins released by algal blooms. Our experiments aim to test the effects of one of the five MC-degrading enzymes on disintegrating microcystin-LR.
To begin this process, we attempted to confirm the chosen bacterial strain contained the MlrA MC-degrading enzyme. The ideal choices of bacteria include Sphingopyxis sp. USTB-05, Novosphingobium sp. THN1, and Sphingomonas sp. ACM-3962, which have been tested for containing MC-biodegrading gene cluster mlrABCD by Wei et al. (2021). However, we were neither able to find a site to purchase any of these bacteria nor obtain them from a laboratory that has worked on them in the past. Instead of locating a specific strain, we focused on finding a strain within the desired genus.
Since the majority of MC-degrading bacteria isolated from Lake Taihu, China, belong to Sphingomonas and Sphingopyxis (Wang et al., 2021), we decided to use Sphingomonas sp. PS-60 for our initial testing of MC-degrading enzymes. Since this bacterium is still mostly unexplored by scientists, we want to begin with checking that the mlrA gene exists within the bacteria, since there is a higher chance that other MC-degrading enzymes are contained in this bacterium if we are able determine that the mlrA gene is contained in this bacterium.
First Experiment
Growing Sphingomonas sp. PS-60
Once we received bacteria numbered ATCC 31461, we followed the directions provided by ATCC to rehydrate the bacteria. We allowed the bacteria to grow in a tube with 6 mL of nutrient broth and a 194 mL flask of nutrient broth overnight in a 30°C shaking incubator. We used Carolina Biological nutrient broth which is composed of beef extract and pancreatic digest of gelatin. Upon visible turbidity, we streaked 2 nutrient agar plates using the flask and 2 nutrient agar plates using the tube. Using isolated colonies from those plates, we streaked two additional plates on nutrient agar.
DNA Extraction and Purification
We followed the DNA Purification procedure for gram-negative bacteria, Thermo Scientific GeneJET Genomic DNA Purification Kit #K072 (Ravintheran et al., 2019). Purification included centrifugation, digestion, proteinase K digest, vortexing, RNase A, vortexing, incubation, Lysis solution, vortexing, 50% ethanol, vortexing, purification column, wash buffer I, centrifugation, wash buffer II, centrifugation, elution buffer, incubation, storing the purified DNA at -20 ℃ (Ravintheran et al., 2019).
PCR
We attempted to obtain the gene mlrA from the Sphingomonas sp. PS-60 DNA that we extracted. We used the following mlrA primers: Forward: 5′-GACCCGATGTTCAAGATACT-3′ and Reverse: 5′-CTCCTCCCACAAATCAGGAC-3′ (Zhang et al., 2017). All primers were purchased from ThermoFisher. We created our PCR mix (Table 1) with the end goal of a 50µl sample, labeled one PCR tube as Sphingomonas mlrA and the other as a negative control, and proceeded to add 25µl of taq PCR Master Mix to each tube.
e202307t01
| Component | Volume | Concentration (µmol/µl) |
|---|---|---|
| Taq PCR Master Mix | 25 µl | 2.5 units Taq DNA Polymerase. 1x QIAGEN PCR Buffer*. 200 μM of each dNTP |
| Forward 10x primer mix | 1 μl | 0.4 μM |
| Reverse 10x primer mix | 1 μl | 0.4 μM |
| Template DNA | 5 μl | 0.1μg/ul |
| RNAse free water | 18µl | – |
| Total | 50 μl | – |
To achieve the ratio of 0.2µM of each primer in the sample, we added 1µl of the forward 10x primer mix and 1µl of the reverse primer mix. We added 5µl of template DNA and 18µl of RNAse free water to the PCR tube labeled Sphingomonas mlrA. We added 23µL of RNAse free water negative control. We briefly centrifuged the two tubes and placed them in the PCR machine (BIO RAD T100 Thermal Cycler) and ran the following PCR program listed in Table 2.
| Step | Time | Temperature |
|---|---|---|
| Initial Denaturing | 3 minutes | 94℃ |
| 3-Step Cycling: | ||
| Denaturing | 45 seconds | 94℃ |
| Annealing | 45 seconds | 50℃ |
| Extension | 1 minute | 72℃ |
| Number of Cycles | 34 | |
| Final Extension | 10 minutes | 72℃ |
DNA Gel Electrophoresis
We analyzed our PCR products using gel electrophoresis. We used 12.5uL per sample and a control with 4uL of minipcr 1kb DNA ladder. We ran the gel for 30 minutes at 90 volts.
BLASTn
We performed a BLASTn alignment of the full mlrB sequence and that of S. microcystinivorans Y2 with the default settings of BLASTn.
Results
Growing Sphingomonas sp. PS-60
Following supplier guidelines, our liquid cultures reached turbidity after two days. Similarly, our streaked plates appeared dark brown with a haze around the colonies after three days (Figure 2). After two days, the plates appeared more orange yellow with the color appearing darker closer to the initial streak (Figure 3).
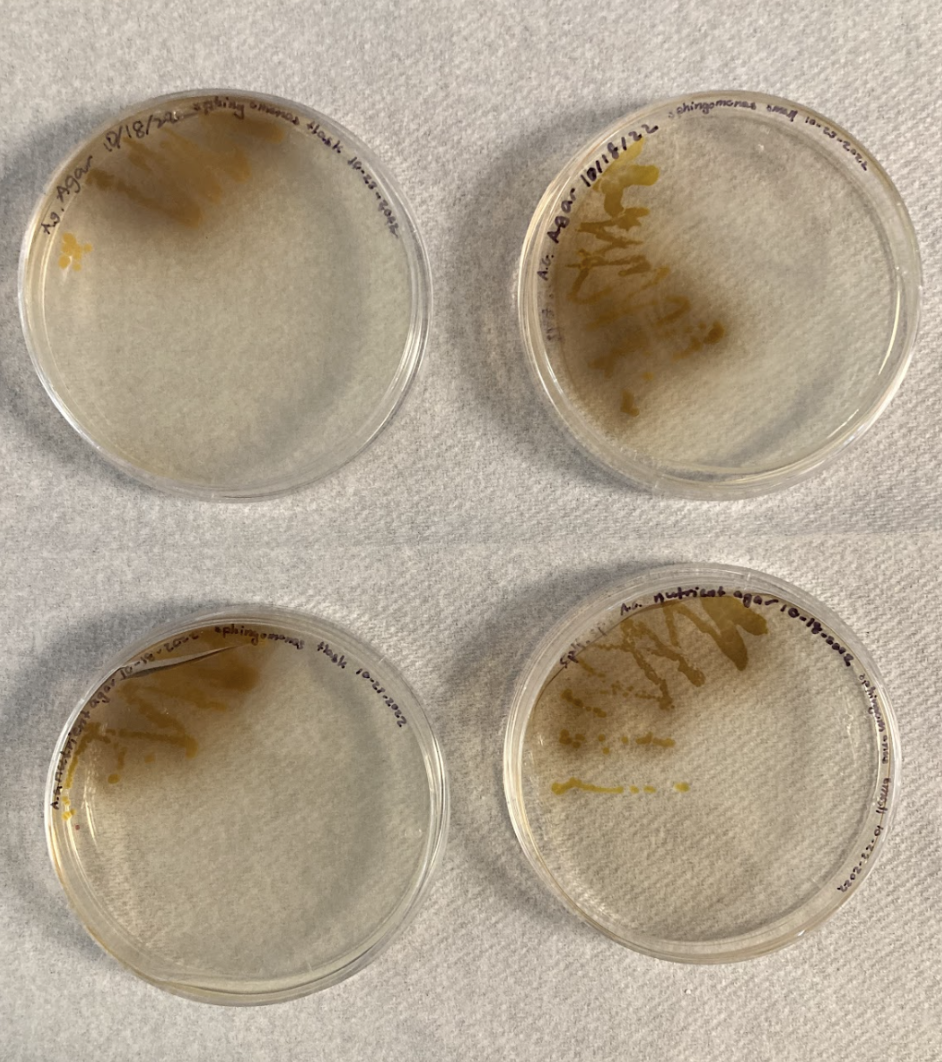
| Figure 2. An image of streaked plates shows the dark brown substance produced by Sphingomonas sp. PS-60 after four days of incubation. |
|---|
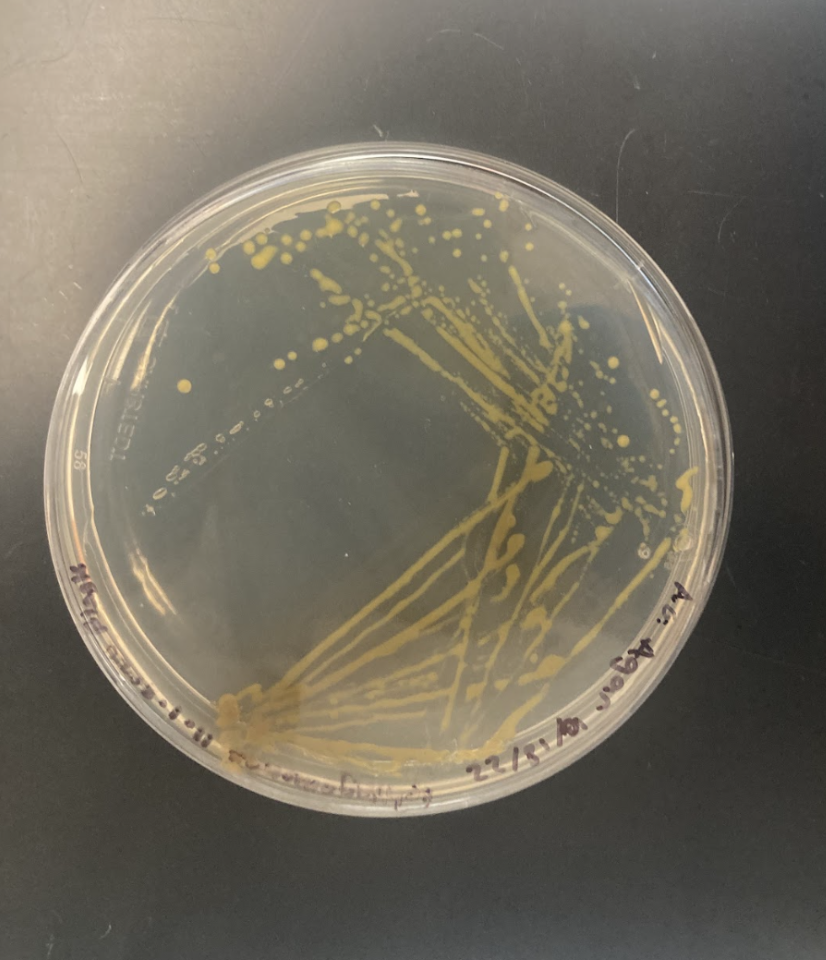
| Figure 3. After two days, the colonies on this streaked place appeared a dark yellow. The original streak appeared a darker orange yellow while the most isolated colonies appeared yellow. |
|---|
PCR and Gel Electrophoresis
As seen in Figure 4, no bands were present. After repeating DNA extraction, PCR, and gel electrophoresis no bands appeared in the negative control or sample lanes. Since there were no visible bands in the bacterial or negative control lane in any of our three trials, this suggests a high possibility that Sphingomonas sp. PS-60 does not contain the mlrA gene.
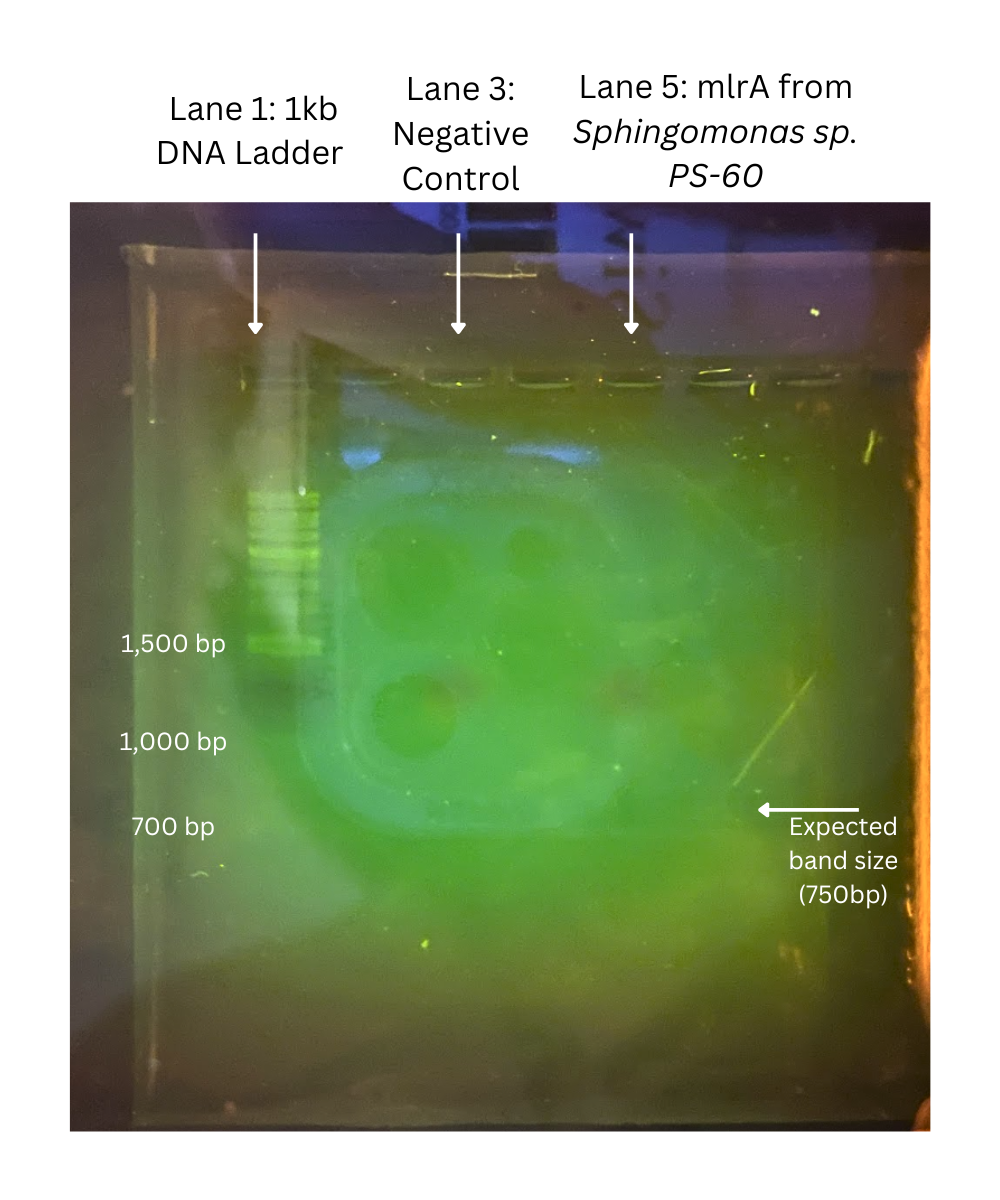
| Figure 4. An image of the electrophoresis gel shows that only the ladder appeared. There are no bands in the negative control lane or the lane with bacterial DNA. |
|---|
BLASTn Results
According to BLASTn results (Figure 5), there is an 88% identity between the genome sequence of Sphingosinice microcystinivorans Y2 and the DNA sequences for the mlrB gene, which gives some promise in the presence of the mlrB gene in S. microcystinivorans Y2 (Altschul et al., 1990).
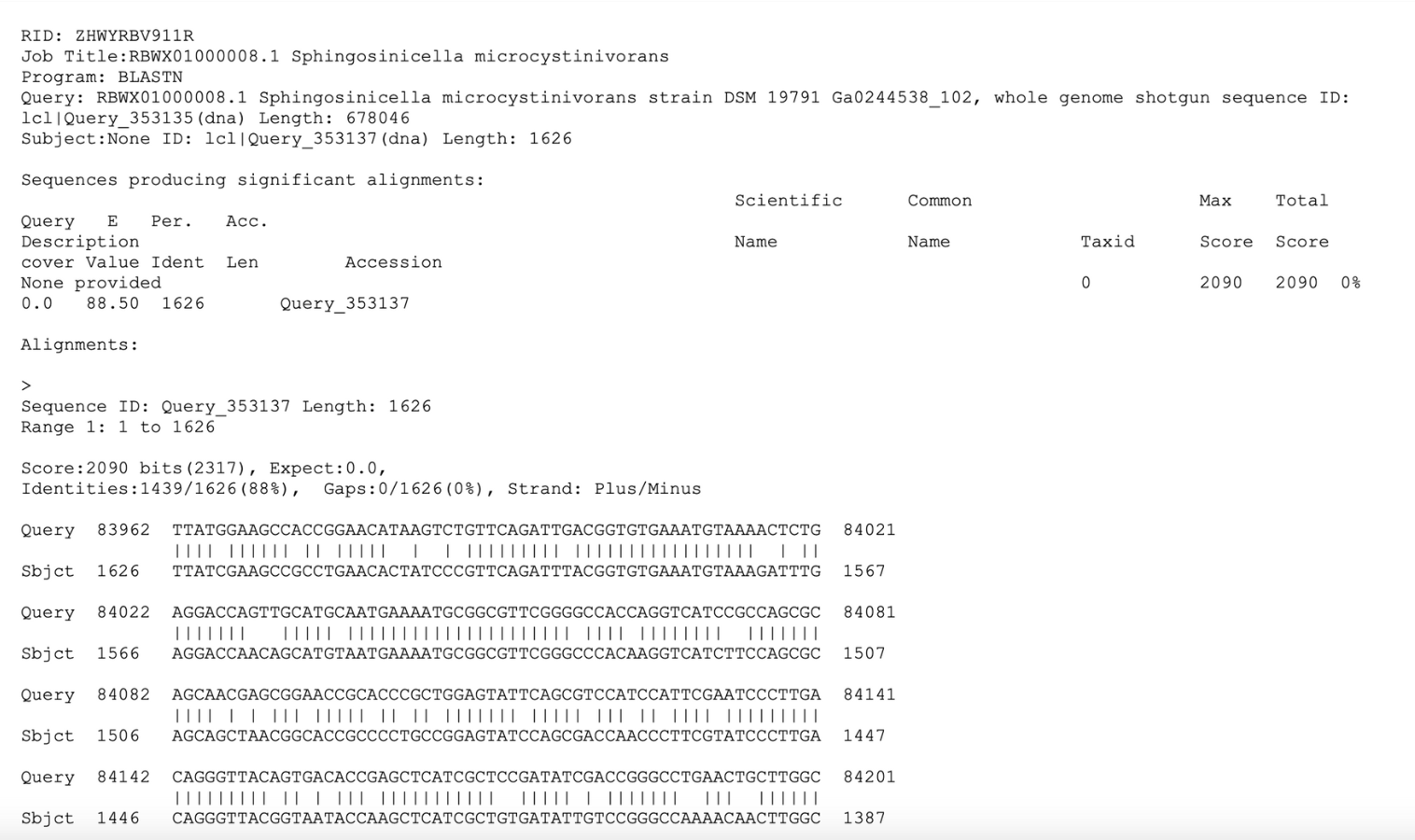
| Figure 5. The BLASTn result and sequence alignment for mlrB and Sphingosinicella microcystinivorans Y2. |
|---|
Discussion
The negative result of our gel electrophoresis has several implications. First, the DNA extraction estimated that we harvest up to 2×10^9 bacterial cells. Due to limited resource availability, we weren’t able to measure the concentration of our bacterial culture using either fluorescence techniques (e.g. nanodrop or Qubit) or a particle counter. Therefore, we roughly estimated the amount of liquid culture, around 1 μL, for purification. In turn, we likely used insufficient DNA samples for PCR, resulting in very low concentrations for starting DNA in our PCR (Life Technplogies, n.d.). However, since we conducted the experiment three times with different amounts of DNA samples, insufficient DNA samples might not be the most probable cause of a negative result for our PCR.
Primer design could be another source of error. We took the primer sequence for Sphingomonas PS-60 from Zhang et al. (2017), which analyzed the roles of mlrA to mlrD from Sphingopyxis sp. in MC-LR degradation, because Sphingopyxis sp. is a gram-negative bacterium from the same genus as our chosen bacteria. However, the primers for Sphingopyxis sp. could be unsuitable for Sphingomonas PS-60, which can cause either the synthesis of an unrelated gene fragment or no reaction at all as no parts of the primer could bind with the bacterial sequence. This source of error remains under investigation, but it could be a potential factor that we need to examine further in future experiments. Moreover, the purity of extracted DNA may also negatively influence the test result of electrophoresis (Purity – Does, n.d.). Inhibitors and proteins left in purified nucleic acids will lead to the inhibition of enzymatic reactions, including PCR and ligation.
Next steps
Upon discovering that Sphingomonas sp. PS-60 may not contain the genes necessary for our design, we found that the bacteria S. microcystinivorans Y2 may instead contain the genes we seek to isolate. In future experiments, we intend to grow S. microcystinivorans Y2, which we have obtained from ATCC. Once we receive the bacteria, we will grow it, then extract and purify the DNA, and eventually, run a PCR to amplify and isolate the mlrB gene.
Author contributions
This project is based off Western Reserve Academy’s 2022 Biobuilder Team’s project “Mass-producing microcystin-LR-degrading enzymes for algal bloom toxic remediation”. All authors contributed to the research, writing, and proofreading of this paper. R.R. was the principal designer and creator of the video.
Acknowledgements
We appreciate Western Reserve Academy for its support through resources and opportunities. We are especially grateful for Dr. Beth Pethel who introduced us to and cultivated our passion for synthetic biology. Additionally, our research would not have been possible without her guidance during labs.
References
Altschul, S. F., Gish, W., Miller, W., Myers, E. W., & Lipman, D. J. (1990). Basic local alignment search tool. Journal of Molecular Biology, 215(3), 403-410. https://doi.org/10.1016/S0022-2836(05)80360-2
Butler, N., Carlisle, J. C., Linville, R., & Washburn, B. (2009). Microcystins: A brief overview of their toxicity and effects, with special reference to fish, wildlife, and livestock. California Environmental Protection Agency, Sacramento, 5.
Cyanosite. (n.d.). Microcystins. https://www-cyanosite.bio.purdue.edu/cyanotox/toxins/microcystins.html
Lee, P. A., Tullman-Ercek, D., & Georgiou, G. (2006). The bacterial twin-arginine translocation pathway. Annual Review of Microbiology, 60, 373–395 https://doi.org/10.1146/annurev.micro.60.080805.142212
Life Technologies. (n.d.). Troubleshooting DNA agarose gel electrophoresis. https://bio.davidson.edu/molecular/tips/trblDNAgel.html
Massey, I. Y., & Yang, F. (2020). A mini review on microcystins and bacterial degradation. Toxins, 12(4), 268. https://doi.org/10.3390/
Metzloff, A. (2019a, October 16). Part:BBa_K2960010. iGEM. http://parts.igem.org/wiki/index.php?title=Part:BBa_K2960010
Metzloff, A. (2019b, October 16). Part:BBa_K2960011. iGEM. http://parts.igem.org/wiki/index.php?title=Part:BBa_K2960011
MCLAB. (n.d.). Dh5-Alpha Competent E. coli. https://www.mclab.com/Dh5-Alpha-Competent-E.-Coli.html
Ohio Sea Grant College. (2022). Fact sheet: Harmful algal blooms and the Toledo event. Glslcities. https://glslcities.org/wp-content/uploads/2015/05/Fact-Sheet-HAB-and-Toledo.pdf
QIAGEN. (n.d.). Purity – Does my sample contain contaminants?. https://www.qiagen.com/us/knowledge-and-support/knowledge-hub/technology-and-research/sample-quality-control/qc-parameters/purity-does-my-sample-contain-contaminants
Ravintheran, S. K., Sivaprakasam, S., Loke, S., Lee, S. Y., Manickam, R., Yahya, A., Croft, L., Millard, A., Parimannan, S., & Rajandas, H. (2019). Complete genome sequence of Sphingomonas paucimobilis AIMST s2, a xenobiotic-degrading bacterium. Scientific Data, 6(1), 280. https://doi.org/10.1038/s41597-019-0289-x
Rogener, M. (2020, August 226). Study identifies causes of Toledo’s unprecedented 2017 Maumee River algal bloom. National Centers for Coastal Ocean Science. https://coastalscience.noaa.gov/news/study-identifies-causes-of-toledos-unprecedented-2017-maumee-river-bloom/
Savola, P. (2016, October 12). Part:BBa_K1907001: Microcystinase (MlrA) for S. cerevisiae. http://parts.igem.org/Part:BBa_K1907001
U.S. National Office for Harmful Algal Blooms. (n.d.). Historical occurrence of habs. https://hab.whoi.edu/about/historical-occurrence-of-habs/
Vanderbilt University Medical Center. (n.d.). Basics of biosafety level. https://www.vumc.org/safety/bio/biosafety-basics-level-1
Wang, S., Yang, S., Zuo, J., Hu, C., Song, L., Gan, N., & Chen, P. (2021). Simultaneous removal of the freshwater bloom-forming Cyanobacterium microcystis and cyanotoxin microcystins via combined use of algicidal bacterial filtrate and the microcystin-degrading enzymatic agent, mlra. Microorganisms, 9(8), 1594. https://doi.org/10.3390/microorganisms9081594
Wei, J., Huang, F., Feng, H., Massey, I. Y., Clara, T., Long, D., Cao, Y., Luo, J., & Yang, F. (2021). Characterization and mechanism of linearized-microcystinase involved in bacterial degradation of microcystins. Frontiers in Microbiology, 12, 646084. https://doi.org/10.3389/fmicb.2021.646084
Yoshida, T., Makina, Y., Nagata, S., Tsutsumi, T., Yoshida, F., Sekijima, M., Tamura, S., & Ueno, Y. (1998). Acute oral toxicity of microcystin-LR, a cyanobacterial hepatotoxin, in mice. Natural Toxins, 5(3), 91-95. https://doi.org/10.1002/1522-7189(1997)5:3%3C91::AID-NT1%3E3.0.CO;2-H Zhang, J., Lu, Q., Ding, Q., Yin, L., & Pu, Y. (2017). A novel and native microcystin-degrading bacterium of Sphingopyxis sp. isolated from lake Taihu. International Journal of Environmental Research and Public Health, 14(10), 1187. https://doi.org/10.3390/ijerph14101187

Be the first to comment