Owen Averell, Fernando Bermudez, Matthew Hernandez, Samuel Pinkerton, and Brant Satterly ▫ Western Reserve Academy, Hudson Ohio, United States
Reviewed on 6 May 2023; Accepted on 13 July 2023; Published on 16 October 2023
With help from the 2023 BioTreks Production Team.
Droughts will further the natural disasters in some parts of the planet as global warming continues to advance. According to the United Nations, climate change has been found to be responsible for a 29 percent increase in the number and duration of droughts in the last 20 years. Droughts are a major environmental stress factor that affects plant growth and productivity thus damaging the first trophic level of the food chain and causing imbalances throughout the ecosystem. Varied cuticle wax amounts alter the rates of permeability through the cuticle, thus affecting the plant’s defense against drought. Wax that comes out of the cuticle in the plant trap water and also reflects the sun rays lowering the temperature of the plant. To enhance the tolerance of the crop to drought, we have designed a genetic modification strategy to increase cuticular wax to reduce water loss thus maintaining plant hydration during a period of drought. This project consists of overexpressing genes WDS1, WAX2, and KCS16, which affect two different pathways of wax production. These two pathways are acyl-reduction and the decarbonylation pathway. The former produces alkanes, aldehydes, ketones, and secondary alcohols, while the latter produces primary alcohols and wax esters. The design utilizes three different plasmid constructs to overexpress each gene individually in three versions of the model plant Arabidopsis thaliana. These plasmid constructs utilize the inducible promoter rd29a, which is induced by drought stress to overexpress each gene. Each plasmid will be introduced into the plant through Agrobacterium-mediated transformation. Agrobacterium tumefaciens are bacteria that infect a plant transferring its DNA into the host cell, enabling easy transformation. This transformation method moves the plasmid construct from the A. tumefaciens to the embryo of A. thaliana through a wound site. This design holds the potential to increase the plant’s resistance to drought stress and improve crop production, contributing to sustainable food security solutions in water-limited environments. With our project, we can transform the future of plant-drought resistance.
Keywords: Drought resistance, wax, Arabidopsis thaliana, climate change, Agrobacterium-mediated transformation
Authors are listed in alphabetical order. Beth Pethel from Western Reserve Academy, Hudson Ohio, United States mentored the group. Please direct all correspondence to .
Climate change is a driving factor behind droughts. In areas where climate change has lowered precipitation rates, such as the Southwest of the United States, the prolonged lack of rain or snow causes droughts (Vilonen et al., 2022, p. 1). However, climate change is not the sole reason why droughts exist; rather, it exacerbates the issue. The additional artificial greenhouse gasses released by factories, cars, and other industrial areas that cause climate change increase the average temperature in the atmosphere and the sea. This causes extreme weather patterns such as more severe storms and hurricanes and lower precipitation rates. (Vilonen et al., 2022, p. 1). The irregular weather patterns, in addition to less precipitation and an increase in sun exposure, leads to the loss of native plants and crops (Ha et al., 2020, p. 1). Human intervention attempts are often not an adequate substitute for rain because plants demand a larger quantity of water than artificial irrigation can sustainably handle. Unlike other natural disasters, such as hurricanes and earthquakes, droughts are unpredictable as they do not follow a clear pattern as to when they occur or will end. Therefore, water management during droughts relies on assumptions, creating risks and water insecurity (Vicente-Serrano et al., 2022, p.1).
In the dry season of 2022, a drought caused the water levels of the Yangtze River in China to drop, which left 220,000 people in the Hubei province in need of drinking water and destroyed 6.9 million hectares of farmland (Schiefelbein, 2022) During a drought, farming becomes more challenging because of the low moisture in the ground which leads to the aforementioned reduced crop yields (Winkler et al., 2017). In these abnormally dry conditions, irrigating fields becomes more expensive and arduous, causing a fall in crop yields, leading to food shortages. Higher food prices will also negatively affect populations, as they make food unaffordable for disadvantaged people.
Climate change and droughts also have damaging effects on native plant species, which impacts ecosystem diversity. For example, a study found that the indigenous plant life was greatly affected in drought-stricken European forests, with tree wood volume being lower on record than previous years (Archaux & Wolters, 2006)
The native plant species impacted by drought also have a command of biodiversity in their original environment keeping it in check (Archaux & Wolters, 2006). The loss of native and endangered plant species’ additional negative impacts include imbalances in the life that inhabits them. Without native plants, invasive plant species may replace and devastate these ecosystems (Archaux & Wolters, 2006). For example, commonly known as the “Tree of Heaven,” Ailanthus altissima Swingle (family Simaroubaceae) is an invasive plant species that was found to have an innate defense against drought, giving it a competitive advantage over native plants (Filippou et al., 2014).
In most plant species, around 90% of water absorbed through the roots is lost through transpiration (Jones, 1992). Transpiration, the evaporation of water through the stems, leaves, and other parts of the plant, is responsible for 97-99.5% of water loss in a plant. The permeability of the cuticle affects the transpiration rate (Kane et al., 2020, p. 1) and plants with a lower permeability rate lose the scarce water they have collected before and during a drought at lower rates. To lower the permeability rate, some plants modify their cuticle through gene regulation, so the structure becomes less permeable in response to droughts (Chen et al., 2020). As a result, those plants retain more water in their systems. One of the main factors affecting cuticular permeability in plants is the amount and type of wax present in the cuticle (Riederer & Schreiber, 2001, p. 1). Studies have shown that higher amounts of wax in the cuticle leads to better defense of the plant against drought as it can lower the loss of water through transpiration (Al-Abdallat et al., 2014).
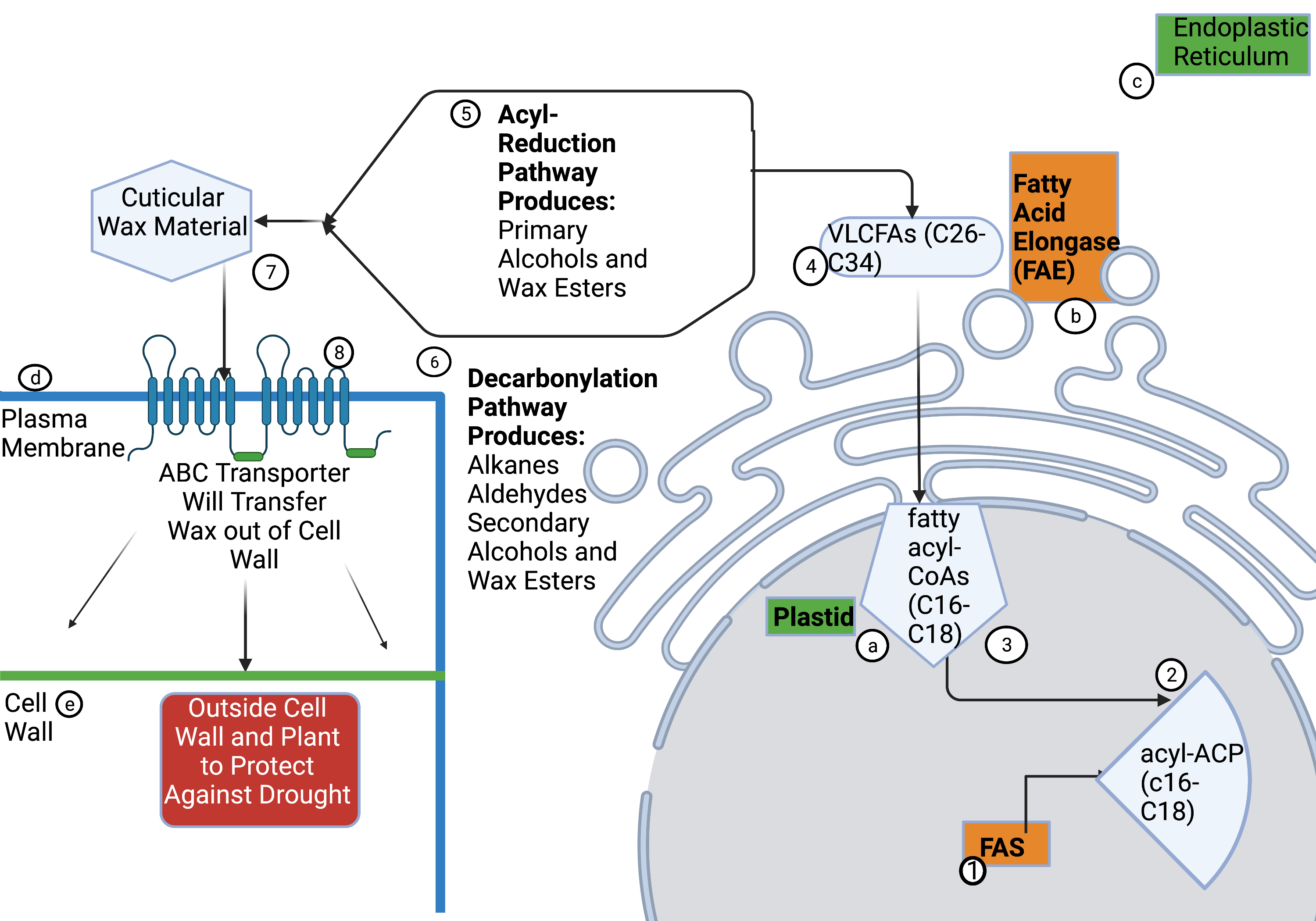
As shown in Figure 1, the process of cuticular wax production begins in the food storage/manufacturing organelle or plastid of the endoplasmic reticulum in epidermal cells where fatty acyl-CoAs (C16 and C18) are elongated to wax precursors of very long-chain fatty acids (VLCFAs) with C26 to C34 chains (Shepard & Griffith, 2006). VLCFAs are fatty acids with more than eighteen carbon atoms. The fatty acyl-CoAs are extended by the fatty acid elongase complex(b) through a repeating reaction process (Kihara, 2012). Many of the compounds for wax synthesis are derived from VLC-acyl-CoA.The wax components are formed through processing of VLCFA by different pathways, including acyl-reduction and decarbonylation (Shepard & Griffith, 2006). The decarbonylation pathway produces alkanes, aldehydes, secondary alcohols, and ketones, while the acyl-reduction pathway produces primary alcohols and wax esters (Shepard & Griffith, 2006). They are moved from the endoplasmic reticulum, where the ABC transporter moves the materials in their biosynthetic pathways to where they will be mixed to make the material. (Xue et al., 2017). The ABC transporter moves the cuticular wax material across the plasma membrane, where it can be further transported to the surface through the cell wall (Xue et al., 2017). These parts can be combined at the cell wall to create wax that is useful to plants for drought and water protection. Improving the cuticular wax production in plants is crucial for improving their drought defense.
Our project focuses on increasing wax production in the cuticle of plants by overexpression of three specific genes, WAX2, KCS16, and WSD1; all three are vital in the production of different components in the cuticular wax (Abdullah et al., 2021; Dhanyalakshmi et al., 2019; Hegebarth et al., 2017). This project anticipates that the overproduction of these three genes will increase the level of cuticular wax, therefore creating increased resistance to droughts (Abdullah et al., 2021). WAX2 is arguably the most critical gene to manipulate due to its proven role in regulating cuticular wax biosynthesis and its impact on drought tolerance in plants (Chen et al., 2003). It is responsible for the synthesis of ketones, alcohols, aldehydes, alkanes and the biosynthesis of the cuticular membrane (Dhanyalakshmi et al., 2019). KCS16 regulates the production of VLCFA’s as well as 24C fatty acid chains (Hegebarth et al., 2017). Finally, WSD1 creates wax esters through biosynthesis (Abdullah et al., 2021). Since wax is made from a combination of alcohols, wax esters, and fatty acid chains, overexpressing each gene is critical for the experiment’s success. These genes will provide insight into which parts of the pathway, when overexpressed, will provide protection against drought, allowing us to make the proper combinations of these genes for future designs.
In order to overexpress the desired genes, a specific promoter had to be found. Due to our project’s focus on drought tolerance, we selected the rd29A promoter. The cis-acting region of the rd29A promoter contains three dehydration-responsive elements, which will cause our genes to overexpress during periods when the plant suffers stress from drought (Yamaguchi-Shinozaki et al., 1992). This promoter functions in almost all organs and tissues during water stress. This promoter would be inserted into a Pcambia1300 binary expression vector by replacing the CaMV-35S promoter with the rd29A promoter (Kasuga et al., 2004). We will introduce each of the three proposed genes into a separate Pcambia1300 vector, creating three different types of plasmid structures. After construction, the plasmids will be inserted into the Agrobacterium through electroporation, and subsequently introduced into the plants using Agrobacterium-mediated transformation. Both methods are well studied and effective ways of delivering the plasmid into the cell (den Dulk-Ras & Hooykaas, 1995; Gelvin, 2003).
Systems level
The Agrobacterium mediated transformation (Figure 2) will be used to overexpress the cuticle wax production A. thaliana. This method involves Agrobacterium cells being inserted into a wound site of a plant. There, DNA derived from the bacterium can integrate itself with the plant’s genetic material. (Hwang et al., 2017). This function is achieved through two genetic elements present on the tumor-inducing (Ti) plasmid: transfer DNA (T-DNA) and virulence (vir) region. The vir region is responsible for the processing and transferring of T-DNA into the plant, resulting in the transformation. (Chang et al., 1994). When the Agrobacterium detects the plant’s vir genes, the bacteria’s virulence genes begin transferring and inserting foreign DNA. When VirA detects appropriate virulence in the plant, it activates VirG via phosphorylation. VirG then transforms into a transcriptional protein, allowing the Ti plasmid to begin creating the Vir genes that are not continuously generated in the cell. Once the plasmid generates VirD1 and VirD2, they aid in the cutting and pasting of the plasmid’s transmitted DNA (Hwang et al., 2017). VirD2 will take the piece of DNA from the Agrobacterium cell and transport it across the cell membranes in a manner similar to bacterial conjugation. Once in the plant cell, the DNA is integrated into the plant genome. Instead of producing bacterial tumors, the plant will spread the modified gene across the cells.
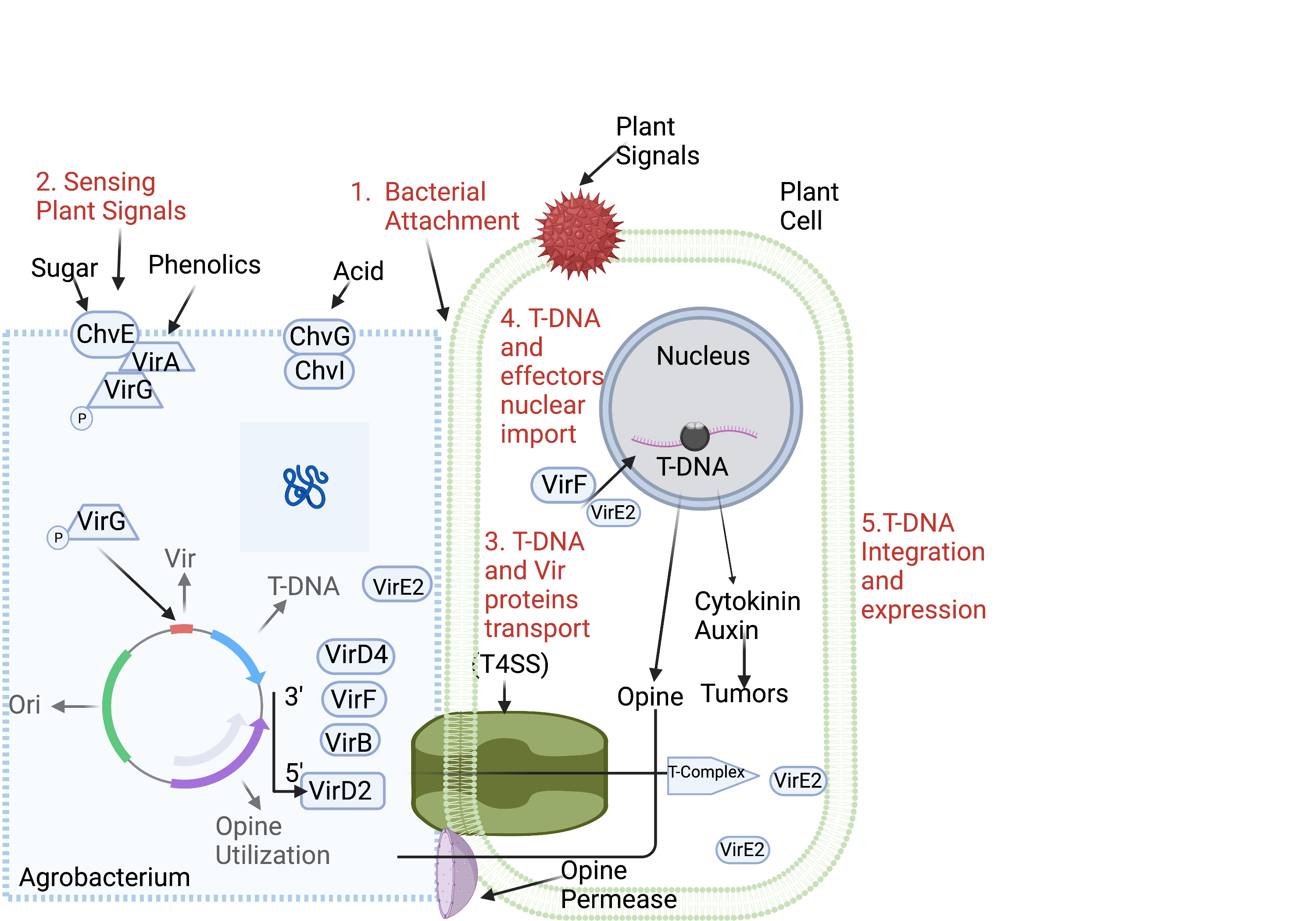
| Figure 2: Agrobacterium-mediated transformation. Overview of the process of Agrobacterium infecting the plant cell and transferring its DNA. Different steps of the process are indicated in red. |
|---|
Embryonic cells will be used for Agrobacterium-mediated transformation due to their rapid growth, which facilitates the spread of the integrated genetic material. To introduce DNA into embryonic cells, seeds will be wounded and, subsequently, infected with the Agrobacterium containing the changed plasmid. PCR will be used to test for a successful transformation. Agrobacterium-mediated floral dip transformation was confirmed to be a reliable approach in A. thaliana transformation in a similar experiment. (Chang et al., 1994).
Device level
To introduce the final Pcambia1300 plasmid constructs into A. tumefaciens strain GV3101, we will use a technique called electroporation. Electroporation involves applying a high-voltage electrical pulse to the bacterial cells in the presence of the plasmid DNA, which allows for the uptake of the DNA by the cells (den Dulk-Ras & Hooykaas, 1995). After electroporation, we will select the transformed bacterial colonies by growing them on kanamycin-containing media. Kanamycin resistance was conferred by the nptII gene on the plasmid, which allows the successfully transformed bacteria to grow.
Parts level
Our design theoretically could enable A. thaliana to combat drought by yielding an increase in its wax levels through overexpression of the genes WAX2, KCS16, and WDS1. All of these genes are involved in the synthesis of the protective wax coating. We will use the vector Pcambia1300 as the Ti-Plasmid. The Pcambia1300 plasmid is a binary vector commonly used for plant transformation studies. Downstream of the T-border (R) we will introduce the promoter rd29a in place of the CAMV35S promoter, which helps control the expression of these genetically modified genes during drought periods. Following the promoter there is a hygromycin selectable marker which will allow us to select the successful transformations in A. thaliana. Next is the Multiple Cloning Site (MCS) which will allow for the insertion of one of our selected genes. The plasmid also contains a selectable marker gene (nptII) for kanamycin resistance downstream of the T border (L) which will be used to select successful transformations in the Agrobacterium.
Safety
A. thaliana, the model plant for genetic engineering and experimentation, will be the plant for testing our transformation. The safety concerns surrounding Agrobacterium-mediated transformation extends from the lab to the outside world. For our project, transforming A. thaliana for increased wax cuticle production may have adverse effects. In the lab, however, the concerns over safety are limited as the precautions minimize any hazards. The Agrobacterium is kept in a sterile environment where the air is cleaned, and the researchers will be in protective clothing as well to keep an uncontaminated space (Que, 2018).
Genetically modified (GM) plants pose a risk when put back into their ecosystems, and by modifying A. thaliana safety risks for other plants and animals in the ecosystem come into concern (Hagedorn, 2000). Genetic engineering could alter activity of different metabolic pathways in plants, leading to the release of new toxins into the natural environment, which could pose a threat to wildlife (Hagedorn, 2000).
Discussion
Once the Agrobacterium mediated transformation is successful, each plant will be grown in a controlled environment. After the plants have fully developed, they will be exposed to a period of drought to activate our added genes, WAX2, WDS1, and KCS16. We will collect leaves from A. thaliana plants subjected to different gene modification strategies. The extracted waxes will be further analyzed to determine the composition of wax components using techniques such as gas chromatography (GC) (Jetter & Schäffer, 2001). The wax content and composition data obtained from the different gene modification strategies can be statistically analyzed to determine the significance of the differences between them. The successful ones will be combined in a new vector system to further increase the cuticular wax production potential.
We plan to implement these gene changes in species of plants that are native to regions where drought is becoming common. However, there may be some drawbacks from having a higher amount of wax. A thicker cuticle, while increasing drought resistance, will not allow the plant to absorb as much CO2 due to the lower transpiration rate, thus interrupting optimal photosynthesis and leading to irregular plant growth (Taiz et al., 2014). We need to consider other issues this may pose, such as a different composition of wax on the plant’s surface, which could impact the properties of the wax layer and its ability to protect the plant from environmental stresses or have other negative effects on metabolism. We will conduct further research and tests to investigate the potential adverse effects of the overexpression of WAX2, WDS1, and KCS16 on plants.
While its structure and amount varies between species, the protective waxy coating is present in all plants. For example, WAX2 may not be present in every plant, but a similar gene will. In every plant, there is a gene that is responsible for wax production. This modification can be implemented in multiple ways, allowing crops to grow in harsher conditions. Farmers would no longer need to worry about drought seasons or the number of crops they plant. These genetically modified crops will resist drought conditions that would otherwise kill them. This could lead to increased production of crops as well as more flexibility in agriculture.
Next steps
If all transformations mentioned are successful, and all of the genes are proven to be effective at increasing wax amounts in the cuticle and reducing the cuticle’s permeability, a new vector system could be designed that will allow the insertion of all three proposed genes into the same embryo. This vector system should be first tested in A. thaliana. If this is successful, it could be further tested in crop plants that are affected by drought. To determine the effect of a new vector on wax production, leaves could be cut into small pieces and cuticular waxes could be extracted using a non-polar solvent such as hexane or chloroform, dried under a stream of nitrogen gas, and weighed using a microbalance to determine the total wax content.
Author contributions
F.B. proposed the idea and developed it. F.B., M.H., S.P., O.A. and B.S. researched and wrote the background. M.H. and S.P. worked on Agrobacterium transformation. F.B. designed the plasmid. O.A. and F.B. wrote devices and parts. B.S. and M.H. wrote safety. S.P., F.B., and O.A. wrote discussions. B.S. and F.B. worked on the citations. M.H. made the images. O.A. edited the video and runs our instagram. F.B. and S.P. proofread.
Acknowledgements
This entire project has been through the work and diligence of our five members. However, there is still one other person, who without them none of this would have been possible. This person is our teacher and mentor Dr. Pethel who has been instrumental in helping to guide us throughout our year of research. Although all of our work has been independent, in times of doubt or confusion about how to continue, she was there to offer a helping hand or a quick lesson. Dr. Pethel has helped us to understand the process for the research paper and we are infinitely grateful to have had someone so great to help us.
References
Abdullah, H. M., Rodriguez, J., Salacup, J. M., Castañeda, I. S., Schnell, D. J., Pareek, A., & Dhankher, O. P. (2021). Increased cuticle waxes by overexpression of wsd1 improves osmotic stress tolerance in Arabidopsis thaliana and Camelina sativa. International Journal of Molecular Sciences, 22(10), 5173. https://doi.org/10.3390/ijms22105173
Al-Abdallat, A. M., Al-Debei, H. S., Ayad, J. Y., & Hasan, S. (2014). Over-expression of SlSHN1 gene improves drought tolerance by increasing cuticular wax accumulation in tomato. International Journal of Molecular Sciences, 15(11), 19499-19508 . https://doi.org/10.3390/ijms151119499
Archaux, F., & Wolters, V. (2006). Impact of summer drought on forest biodiversity: What do we know?. Annals of Forest Science, 63(6), 645-652. https://doi.org/10.1051/forest:2006041
Chang, S. S., Park, S. K., Kim, B. C., Kang, B. J., Kim, D. U., & Nam, H. G. (1994). Stable genetic transformation of Arabidopsis thaliana by Agrobacterium inoculation in plants. The Plant Journal, 5(4), 451-610. https://doi.org/10.1046/j.1365-313X.1994.05040551.x
Chen, M., Zhu, X., Zhang, Y., Du, Z., Chen, X., Kong, X., Sun, W., & Chen, C. (2020). Drought stress modify cuticle of tender tea leaf and mature leaf for transpiration barrier enhancement through common and distinct modes. Scientific Reports, 10 (1), 6696. https://doi.org/10.1038%2Fs41598-020-63683-4
Chen, X., Goodwin, S. M., Boroff, V. L., Liu, X., & Jenks, M. A. (2003). Cloning and characterization of the WAX2gene of arabidopsis involved in cuticle membrane and wax production. The Plant Cell, 15(5), 1170-1185. https://doi.org/10.1105%2Ftpc.010926
Dhanyalakshmi, K. H., Soolanayakanahally, R. Y., Rahman, T. R., Tanino, K. K., & Nataraja, K. N. (2019). Leaf cuticular wax, a trait for multiple stress resistance in crop plants. In A. de Oliveira, (Ed.), Abiotic and biotic stress in plants. IntechOpen. https://doi.org/10.5772/intechopen.84565
den Dulk-Ras, A., & Hooykaas, P. J. J. (1995). Electroporation of Agrobacterium tumefaciens. In G. E. Morris (Ed.), Plant gene transfer and expression protocols (pp. 63-76). Springer. https://doi.org/10.1385/0-89603-328-7:63
Filippou, P., Touchagier, P., Skotti, E., & Fotopoulos, V. (2014). Proline and reactive oxygen/nitrogen species metabolism is involved in the tolerant response of the invasive plant species Ailanthus altissima to drought and salinity. Environmental and Experimental Botany, 97, 1-10. https://doi.org/10.1016/j.envexpbot.2013.09.010
Jetter, R., & Schäffer, S. (2001). Chemical composition of the Prunus laurocerasus leaf surface. Dynamic changes of the epicuticular wax film during leaf development. Plant Physiology, 126(4), 1725-1737. https://doi.org/10.1104/pp.126.4.1725
Gelvin, S. B. (2003). Agrobacterium-mediated plant transformation: The biology behind the “gene-jockeying” tool. Microbiology and Molecular Biology Reviews, 67(1), 16-37. https://doi.org/10.1128/MMBR.67.1.16-37.2003
Ha, T. T. V., Fan, H., & Shuang, L. (2020). Climate change impact assessment on northeast china’s grain production. Environmental Science and Pollution Research, 28(12), 14508-14520. https://doi.org/10.1007/s11356-020-11602-5
Hagedorn, C. (2000, February). Scientific basis of risks associated with transgenic crops. Virginia State University. https://www.sites.ext.vt.edu/newsletter-archive/cses/2000-02/risks.html
Hegebarth, D., Buschhaus, C., Joubès, J., Thoraval, D., Bird, D., & Jetter, R. (2017). Arabidopsis ketoacyl‐CoA synthase 16 (KCS16) forms C36/C38 acyl precursors for leaf trichome and pavement surface wax. Plant, Cell & Environment, 40(9), 1761-1776. https://doi.org/10.1111/pce.12981
Hwang, H. H., Yu, M., & Lai, E. M. (2017). Agrobacterium-mediated plant transformation: biology and applications. The Arabidopsis Book, 15, e0186. https://doi.org/10.1199%2Ftab.0186
Jones, H. G. (1992). Plants and microclimate: A quantitative approach to environmental plant physiology (2nd ed.). Cambridge University Press.
Kane, C. N., Jordan, G. J., Jansen, S., & Mcadam, S. A. M. (2020). A permeable cuticle, not open stomata, is the primary source of water loss from expanding leaves. Frontiers in Plant Science, 11, 774 https://doi.org/10.3389/fpls.2020.00774
Kasuga, M., Miura, S., Shinozaki, K., & Yamaguchi-Shinozaki, K. (2004). A combination of the Arabidopsis DREB1A gene and stress-inducible rd29A promoter improved drought- and low-temperature stress tolerance in tobacco by gene transfer. Plant and Cell Physiology, 45(3), 346-350. https://doi.org/10.1093/pcp/pch037
Kihara, A. (2012). Very long-chain fatty acids: Elongation, physiology and related disorders. The Journal of Biochemistry, 152(5), 387-395. https://doi.org/10.1093/jb/mvs105
Que, Q. (2018, March 23). Are there dangers for scientists working on genetically engineered plants, if so what are they? GMO Answers. https://gmoanswers.com/production-genetically-engineered-plants-aka-plant-transformation-probably-one-safest-techniques
Riederer, M., & Schreiber, L. (2001). Protecting against water loss: Analysis of the barrier properties of plant cuticles. Journal of Experimental Botany, 52(363), 2023-2032. https://doi.org/10.1093/jexbot/52.363.2023
Schiefelbein, M. (2022, August 20). Scorching heat in China wilts crops as farmers struggle with shrinking water supplies. PBS. https://www.pbs.org/newshour/world/scorching-heat-in-china-wilts-crops-as-farmers-struggle-with-shrinking-water-supplies
Shepherd, T., & Wynne Griffiths, D. (2006). The effects of stress on plant cuticular waxes. New Phytologist, 171(3), 469-499. https://doi.org/10.1111/j.1469-8137.2006.01826.x
Taiz, L., Zeiger, E., Møller, I. M., & Murphy, A. (2014). Plant physiology and development (6th ed.). Sinauer Associates.
Tsegai, D., Medel, M., Augenstein, P., & Huang, Z. (2022). Drought in numbers 2022 – Restoration for readiness and resilience (Report No. 15). United Nations. https://www.unccd.int/sites/default/files/2022-06/Drought%20in%20Numbers%20%28English%29.pdf
Vicente-serrano, S. M., Peña-angulo, D., Beguería, S., Domínguez-castro, F., Tomás-burguera, M., Noguera, I., Gimeno-sotelo, L., & El kenawy, A. (2022). Global drought trends and future projections. Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences, 380(2238). https://doi.org/10.1098/rsta.2021.0285
Vilonen, L., Ross, M., & Smith, M. D. (2022). What happens after drought ends: Synthesizing terms and definitions. New Phytologist, 235(2), 420-431. https://doi.org/10.1111/nph.18137
Winkler, K., Gessner, U., & Hochschild, V. (2017). Identifying droughts affecting agriculture in Africa based on remote sensing time series between 2000–2016: Rainfall anomalies and vegetation condition in the context of ENSO. Remote Sensing, 9(8), 381. https://doi.org/10.3390/rs9080831
Xue, D., Zhang, X., Lu, X., Chen, G., & Chen, Z. H. (2017). Molecular and evolutionary mechanisms of cuticular wax for plant drought tolerance. Frontiers in Plant Science, 8, 621. https://doi.org/10.3389/fpls.2017.00621 Yamaguchi-Shinozaki, K., Koizumi, M., Urao, S., & Shinozaki, K. (1992). Molecular cloning and characterization of 9 cDNAs for genes that are responsive to desiccation in Arabidopsis thaliana: Sequence analysis of one cDNA clone that encodes a putative transmembrane channel protein. Plant and Cell Physiology, 33(3), 217-224. https://doi.org/10.1093/oxfordjournals.pcp.a078243


Be the first to comment